Expiring Drug Patents Cheat Sheet
We analyse the patents covering drugs in 134 countries and quickly give you the likely loss-of-exclusivity/generic entry date
Canada: These 77 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "Canada: These 77 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027" DrugPatentWatch.com thinkBiotech, 2026 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
When can TRACLEER (bosentan) generic drug versions launch?
Generic name: bosentan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 15, 2026
Generic Entry Controlled by: Canada Patent 2,607,098
Patent Title: COMPRIME DISPERSIBLE (DISPERSIBLE BOSERTAN TABLET)
TRACLEER is a drug marketed by Actelion. There are two patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has twenty-eight patent family members in twenty-three countries. There has been litigation on patents covering TRACLEER
See drug price trends for TRACLEER.
The generic ingredient in TRACLEER is bosentan. There are nineteen drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the bosentan profile page.
When can ZOLINZA (vorinostat) generic drug versions launch?
Generic name: vorinostat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 16, 2026
Generic Entry Controlled by: Canada Patent 2,580,367
Patent Title: FORMULATIONS DE L'ACIDE SUBEROYLANILIDE HYDROXAMIQUE ET LEURS PROCEDES DE PRODUCTION (FORMULATIONS OF SUBEROYLANILIDE HYDROXAMIC ACID AND METHODS FOR PRODUCING SAME)
ZOLINZA is a drug marketed by Msd Sub Merck. There are three patents protecting this drug.
This drug has one hundred and thirty-one patent family members in thirty-five countries.
See drug price trends for ZOLINZA.
The generic ingredient in ZOLINZA is vorinostat. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the vorinostat profile page.
When can LATUDA (lurasidone hydrochloride) generic drug versions launch?
Generic name: lurasidone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 26, 2026
Generic Entry Controlled by: Canada Patent 2,606,510
Patent Title: COMPOSITION PHARMACEUTIQUE (PHARMACEUTICAL COMPOSITION)
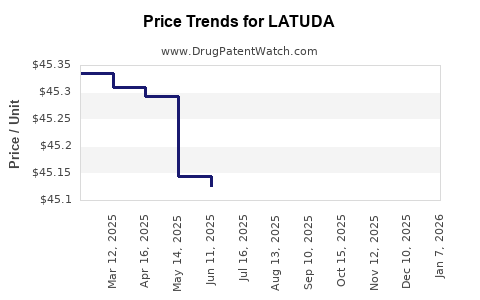
This drug has forty-four patent family members in twenty-two countries. There has been litigation on patents covering LATUDA
See drug price trends for LATUDA.
The generic ingredient in LATUDA is lurasidone hydrochloride. There are twenty-six drug master file entries for this API. Twenty-nine suppliers are listed for this generic product. Additional details are available on the lurasidone hydrochloride profile page.
When can VICTRELIS (boceprevir) generic drug versions launch?
Generic name: boceprevir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 31, 2026
Generic Entry Controlled by: Canada Patent 2,610,167
Patent Title: ADMINISTRATION D'INHIBITEURS DE PROTEASE DU VHC EN COMBINAISON AVEC DES ALIMENTS POUR AMELIORER LA BIODISPONIBILITE (HCV PROTEASE INHIBITORS IN COMBINATION WITH FOOD)
VICTRELIS is a drug marketed by Merck Sharp Dohme. There are two patents protecting this drug.
This drug has twenty-seven patent family members in seventeen countries.
The generic ingredient in VICTRELIS is boceprevir. Additional details are available on the boceprevir profile page.
When can OMONTYS (peginesatide acetate) generic drug versions launch?
Generic name: peginesatide acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 05, 2026
Generic Entry Controlled by: Canada Patent 2,609,401
Patent Title: PREPARATIONS DE PEPTIDES AGONISTES DU RECEPTEUR DE L'ERYTHROPOIETINE ET UTILISATIONS (ERYTHROPOIETIN RECEPTOR PEPTIDE FORMULATIONS AND USES)
OMONTYS is a drug marketed by Takeda Pharms Usa. There are two patents protecting this drug.
This drug has twenty-seven patent family members in eighteen countries.
The generic ingredient in OMONTYS is peginesatide acetate. Additional details are available on the peginesatide acetate profile page.
When can FINACEA (azelaic acid) generic drug versions launch?
Generic name: azelaic acid
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 07, 2026
Generic Entry Controlled by: Canada Patent 2,611,577
Patent Title: KIT ET COMPOSITION ANTIBIOTIQUES ET LEURS UTILISATIONS (ANTIBIOTIC KIT AND COMPOSITION AND USES THEREOF)

This drug has one hundred and thirty-seven patent family members in twenty countries. There has been litigation on patents covering FINACEA
See drug price trends for FINACEA .
The generic ingredient in FINACEA is azelaic acid. There are eight drug master file entries for this API. Eight suppliers are listed for this generic product. Additional details are available on the azelaic acid profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 12, 2026
Generic Entry Controlled by: Canada Patent 2,650,263
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 16, 2026
Generic Entry Controlled by: Canada Patent 2,684,571
Patent Title: NOUVELLES UTILISATIONS THERAPEUTIQUES DE 1-[2-(2,4-DIMETHYLPHENYLSULFANYL)PHENYL]PIPERAZINE (NOVEL THERAPEUTIC USES OF 1-[2-(2,4-DIMETHYLPHENYLSULFANYL)PHENYL]-PIPE RAZINE)

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Canada Patent 2,655,835
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Canada Patent 2,653,012

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can JESDUVROQ (daprodustat) generic drug versions launch?
Generic name: daprodustat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: Canada Patent 2,655,491
JESDUVROQ is a drug marketed by Glaxosmithkline. There are six patents protecting this drug.
This drug has forty-four patent family members in thirty-one countries.
See drug price trends for JESDUVROQ.
The generic ingredient in JESDUVROQ is daprodustat. One supplier is listed for this generic product. Additional details are available on the daprodustat profile page.
When can TEKTURNA HCT (aliskiren hemifumarate; hydrochlorothiazide) generic drug versions launch?
Generic name: aliskiren hemifumarate; hydrochlorothiazide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: Canada Patent 2,654,872

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering TEKTURNA HCT
See drug price trends for TEKTURNA HCT.
The generic ingredient in TEKTURNA HCT is aliskiren hemifumarate; hydrochlorothiazide. There are four drug master file entries for this API. Additional details are available on the aliskiren hemifumarate; hydrochlorothiazide profile page.
When can APLENZIN (bupropion hydrobromide) generic drug versions launch?
Generic name: bupropion hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 27, 2026
Generic Entry Controlled by: Canada Patent 2,578,626
Patent Title: FORMULATIONS A LIBERATION MODIFIEE D'UN SEL DE BUPROPION (MODIFIED-RELEASE FORMULATIONS OF A BUPROPION SALT)
APLENZIN is a drug marketed by Bausch. There are eight patents protecting this drug and three Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has fifty-two patent family members in eighteen countries. There has been litigation on patents covering APLENZIN
See drug price trends for APLENZIN.
The generic ingredient in APLENZIN is bupropion hydrobromide. There are thirty-eight drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the bupropion hydrobromide profile page.
When can EXFORGE HCT (amlodipine besylate; hydrochlorothiazide; valsartan) generic drug versions launch?
Generic name: amlodipine besylate; hydrochlorothiazide; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 27, 2026
Generic Entry Controlled by: Canada Patent 2,654,986

This drug has fifty-two patent family members in eighteen countries. There has been litigation on patents covering EXFORGE HCT
See drug price trends for EXFORGE HCT.
The generic ingredient in EXFORGE HCT is amlodipine besylate; hydrochlorothiazide; valsartan. There are fifty drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the amlodipine besylate; hydrochlorothiazide; valsartan profile page.
When can BOSULIF (bosutinib monohydrate) generic drug versions launch?
Generic name: bosutinib monohydrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,613,053
Patent Title: FORMES CRISTALLINES DE 4-[(2,4-DICHLORO-5-METHOXYPHENYL)AMINO]-6-METHOXY-7-[3-(4-METHYL-1-PIPERAZINYL)PROPOXY]-3-QUINOLINECARB-ONITRILE,ET LEURS PROCEDES DE PREPARATION (CRYSTALLINE FORMS OF 4-[(2,4-DICHLORO-5-METHOXYPHENYL)AMINO]-6-METHOXY-7-[3-(4-METHYL-1-PIPERAZINYL)PROPOXY]-3-QUINOLINECARBONITRILE ANDMETHODS OF PREPARING THE SAME)
BOSULIF is a drug marketed by Pf Prism Cv. There are four patents protecting this drug and two Paragraph IV challenges. Three tentatively approved generics are ready to enter the market.
This drug has eighty-one patent family members in thirty countries. There has been litigation on patents covering BOSULIF
See drug price trends for BOSULIF.
The generic ingredient in BOSULIF is bosutinib monohydrate. There are five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the bosutinib monohydrate profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,653,344

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,924,318

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,985,797

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,653,344

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,924,318

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,985,797

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,653,344

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,924,318

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,985,797

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Canada Patent 2,653,374
Patent Title: MODULATEURS DE PROPRIETES PHARMACOCINETIQUES D'AGENTS THERAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Canada Patent 2,678,907
Patent Title: MODULATEURS DE PROPRIETES PHARMACOCINETIQUES DE PRODUITS THERAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Canada Patent 2,692,331
Patent Title: MODULATEURS DES PROPRIETES PHARMACOCINETIQUES DE SUBSTANCES THERAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can BYFAVO (remimazolam besylate) generic drug versions launch?
Generic name: remimazolam besylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 10, 2026
Generic Entry Controlled by: Canada Patent 2,657,347
Patent Title: SELS DE BENZODIAZEPINE ET LEURS FORMES POLYMORPHES A ACTION BREVE (SHORT-ACTING BENZODIAZEPINE SALTS AND THEIR POLYMORPHIC FORMS)
BYFAVO is a drug marketed by Acacia. There are eleven patents protecting this drug.
This drug has fifty-three patent family members in twenty-four countries. There has been litigation on patents covering BYFAVO
See drug price trends for BYFAVO.
The generic ingredient in BYFAVO is remimazolam besylate. One supplier is listed for this generic product. Additional details are available on the remimazolam besylate profile page.
When can REZDIFFRA (resmetirom) generic drug versions launch?
Generic name: resmetirom
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 11, 2026
Generic Entry Controlled by: Canada Patent 2,614,529
Patent Title: DERIVES DE PYRIDAZINONE UTILISES COMME AGONISTES DU RECEPTEUR DE L'HORMONE THYROIDIENNE (PYRIDAZINONE DERIVATIVES AS THYROID HORMONE RECEPTOR AGONISTS)
REZDIFFRA is a drug marketed by Madrigal. There are six patents protecting this drug.
This drug has one hundred and twenty-six patent family members in thirty-seven countries.
See drug price trends for REZDIFFRA.
The generic ingredient in REZDIFFRA is resmetirom. One supplier is listed for this generic product. Additional details are available on the resmetirom profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Canada Patent 2,656,456
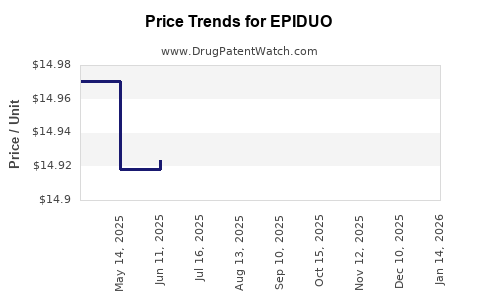
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Canada Patent 2,656,456
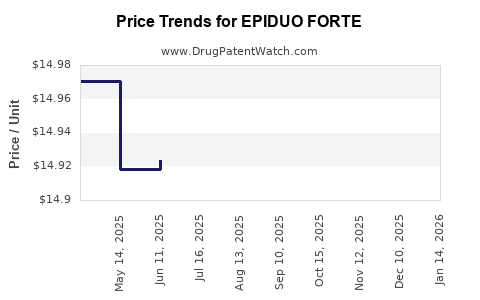
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO FORTE
See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can RASUVO (methotrexate) generic drug versions launch?
Generic name: methotrexate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 21, 2026
Generic Entry Controlled by: Canada Patent 2,869,671
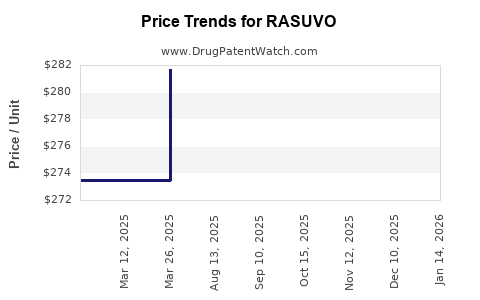
This drug has twenty-nine patent family members in twenty-one countries. There has been litigation on patents covering RASUVO
See drug price trends for RASUVO.
The generic ingredient in RASUVO is methotrexate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the methotrexate profile page.
When can OLYSIO (simeprevir sodium) generic drug versions launch?
Generic name: simeprevir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 28, 2026
Generic Entry Controlled by: Canada Patent 2,616,580
Patent Title: INHIBITEURS MACROCYCLIQUES DU VIRUS DE L'HEPATITE C (MACROCYCLIC INHIBITORS OF HEPATITIS C VIRUS)
OLYSIO is a drug marketed by Janssen Prods. There are eight patents protecting this drug.
This drug has sixty-four patent family members in forty-one countries.
See drug price trends for OLYSIO.
The generic ingredient in OLYSIO is simeprevir sodium. There is one drug master file entry for this API. Additional details are available on the simeprevir sodium profile page.
When can EPSOLAY (benzoyl peroxide) generic drug versions launch?
Generic name: benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 02, 2026
Generic Entry Controlled by: Canada Patent 2,617,681
Patent Title: REVETEMENT PAR UN OXYDE METALLIQUE D'INGREDIENTS HYDROINSOLUBLES (METAL OXIDE COATING OF WATER INSOLUBLE INGREDIENTS)
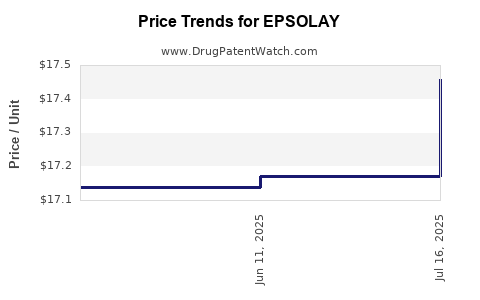
This drug has fifty-one patent family members in fifteen countries. There has been litigation on patents covering EPSOLAY
See drug price trends for EPSOLAY.
The generic ingredient in EPSOLAY is benzoyl peroxide. There are seventeen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the benzoyl peroxide profile page.
When can SALONPAS (menthol; methyl salicylate) generic drug versions launch?
Generic name: menthol; methyl salicylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 04, 2026
Generic Entry Controlled by: Canada Patent 2,652,220
Patent Title: PREPARATION ADHESIVE COMPORTANT DU SALICYLATE DE METHYLE POUR ABSORPTION PERCUTANEE (ADHESIVE PREPARATION COMPRISING METHYL SALICYLATE FOR PERCUTANEOUS ABSORPTION)
SALONPAS is a drug marketed by Hisamitsu Pharm Co. There are two patents protecting this drug.
This drug has twenty patent family members in sixteen countries.
The generic ingredient in SALONPAS is menthol; methyl salicylate. There are eighteen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the menthol; methyl salicylate profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: Canada Patent 2,660,520

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 15, 2026
Generic Entry Controlled by: Canada Patent 2,619,475
Patent Title: COMPOSITIONS PHARMACEUTIQUES A LIBERATION CONTROLEE DESTINEES A DES MEDICAMENTS ACIDO-LABILES (CONTROLLED RELEASE PHARMACEUTICAL COMPOSITIONS FOR ACID LABILE DRUGS)
CREON is a drug marketed by
This drug has ninety-three patent family members in thirty-one countries.
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 15, 2026
Generic Entry Controlled by: Canada Patent 2,619,477
Patent Title: NOYAUX CONSTITUES DE MICROGRANULES DE PANCREATINE ADAPTES A UN ENROBAGE ENTERIQUE (PANCREATIN MICROPELLET CORES SUITABLE FOR ENTERIC COATING)
CREON is a drug marketed by
This drug has ninety-three patent family members in thirty-one countries.
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can EUCRISA (crisaborole) generic drug versions launch?
Generic name: crisaborole
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 16, 2026
Generic Entry Controlled by: Canada Patent 2,635,680
Patent Title: PETITES MOLECULES CONTENANT DU BORE (BORON-CONTAINING SMALL MOLECULES)
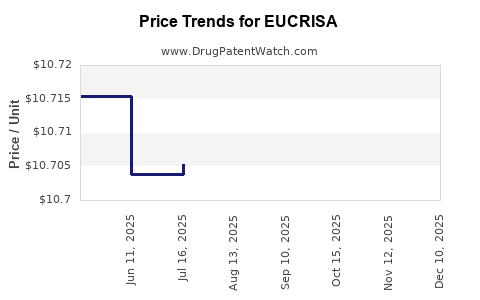
This drug has one hundred and forty-eight patent family members in twenty-eight countries. There has been litigation on patents covering EUCRISA
See drug price trends for EUCRISA.
The generic ingredient in EUCRISA is crisaborole. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the crisaborole profile page.
When can KERYDIN (tavaborole) generic drug versions launch?
Generic name: tavaborole
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 16, 2026
Generic Entry Controlled by: Canada Patent 2,635,680
Patent Title: PETITES MOLECULES CONTENANT DU BORE (BORON-CONTAINING SMALL MOLECULES)

This drug has one hundred and forty-eight patent family members in twenty-eight countries. There has been litigation on patents covering KERYDIN
See drug price trends for KERYDIN.
The generic ingredient in KERYDIN is tavaborole. There are six drug master file entries for this API. Seven suppliers are listed for this generic product. Additional details are available on the tavaborole profile page.
When can AURYXIA (ferric citrate) generic drug versions launch?
Generic name: ferric citrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 18, 2026
Generic Entry Controlled by: Canada Patent 2,619,591
Patent Title: COMPOSES ORGANIQUES FERRIQUES DE QUALITE PHARMACEUTIQUE AINSI QU'UTILISATION DE CEUX-CI ET PROCEDES DE FABRICATION DE CEUX-CI (PHARMACEUTICAL-GRADE FERRIC ORGANIC COMPOUNDS, USES THEREOF AND METHODS OF MAKING SAME)
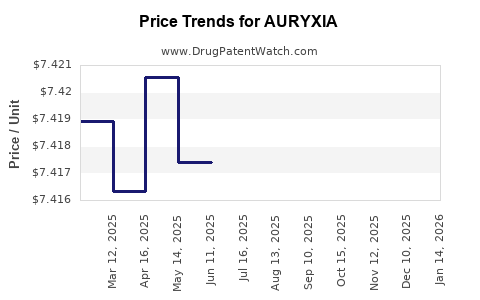
This drug has one hundred and twenty-two patent family members in twenty-three countries. There has been litigation on patents covering AURYXIA
See drug price trends for AURYXIA.
The generic ingredient in AURYXIA is ferric citrate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the ferric citrate profile page.
When can AURYXIA (ferric citrate) generic drug versions launch?
Generic name: ferric citrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 18, 2026
Generic Entry Controlled by: Canada Patent 3,050,453
Patent Title: COMPOSES ORGANIQUES FERRIQUES DE QUALITE PHARMACEUTIQUE AINSI QU'UTILISATION DE CEUX-CI ET PROCEDES DE FABRICATION DE CEUX-CI (PHARMACEUTICAL-GRADE FERRIC ORGANIC COMPOUNDS, USES THEREOF AND METHODS OF MAKING SAME)

This drug has one hundred and twenty-two patent family members in twenty-three countries. There has been litigation on patents covering AURYXIA
See drug price trends for AURYXIA.
The generic ingredient in AURYXIA is ferric citrate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the ferric citrate profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 21, 2026
Generic Entry Controlled by: Canada Patent 2,659,328
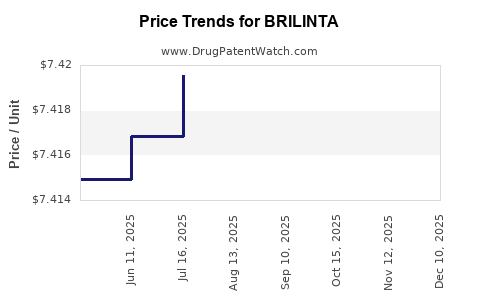
This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 29, 2026
Generic Entry Controlled by: Canada Patent 2,659,770
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the macitentan profile page.
When can LIVDELZI (seladelpar lysine) generic drug versions launch?
Generic name: seladelpar lysine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 13, 2026
Generic Entry Controlled by: Canada Patent 2,622,722
LIVDELZI is a drug marketed by Gilead Sciences Inc. There are six patents protecting this drug.
This drug has one hundred and thirty-eight patent family members in forty-six countries.
The generic ingredient in LIVDELZI is seladelpar lysine. One supplier is listed for this generic product. Additional details are available on the seladelpar lysine profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 15, 2026
Generic Entry Controlled by: Canada Patent 2,663,172
Patent Title: PREPARATION MEDICAMENTEUSE EMPECHANT L'UTILISATION ABUSIVE (ABUSE RESISTANT DRUG FORMULATION)
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 15, 2026
Generic Entry Controlled by: Canada Patent 2,699,142
Patent Title: FORMULATION MEDICAMENTEUSE CONTRE L'ABUS (ABUSE RESISTANT DRUG FORMULATION)
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 22, 2026
Generic Entry Controlled by: Canada Patent 2,620,380
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can ZUNVEYL (benzgalantamine gluconate) generic drug versions launch?
Generic name: benzgalantamine gluconate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 22, 2026
Generic Entry Controlled by: Canada Patent 2,623,114
Patent Title: AMPLIFICATEURS CHOLINERGIQUES DE PERMEABILITE DE LA BARRIERE SANG-CERVEAU AMELIOREE POUR LE TRAITEMENT DE MALADIES ACCOMPAGNEES D'UNE DEFICIENCE COGNITIVE (CHOLINERGIC ENHANCERS WITH IMPROVED BLOOD-BRAIN BARRIER PERMEABILITY FOR THE TREATMENT OF DISEASES ACCOMPANIED BY COGNITIVE IMPAIRMENT)
ZUNVEYL is a drug marketed by Alpha Cognition. There are four patents protecting this drug.
This drug has twenty-six patent family members in seventeen countries. There has been litigation on patents covering ZUNVEYL
The generic ingredient in ZUNVEYL is benzgalantamine gluconate. One supplier is listed for this generic product. Additional details are available on the benzgalantamine gluconate profile page.
When can REVLIMID (lenalidomide) generic drug versions launch?
Generic name: lenalidomide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 03, 2026
Generic Entry Controlled by: Canada Patent 2,570,755
Patent Title: METHODES DE TRAITEMENT DE CERTAINES FORMES DE LEUCEMIE AU MOYEN DE 3-(4-AMINO-1-OXO-1,3-DIHYDRO-ISOINDOL-2-YL)-PIPERIDIN-2,6-DIONE (METHODS USING 3-(4-AMINO-1-OXO-1,3-DIHYDRO-ISOINDOL-2-YL)-PIPERIDINE-2,6-DIONE FOR TREATMENT OF CERTAIN LEUKEMIAS)
REVLIMID is a drug marketed by Bristol Myers Squibb. There are two patents protecting this drug and three Paragraph IV challenges.
This drug has three hundred and thirty-one patent family members in forty-one countries. There has been litigation on patents covering REVLIMID
See drug price trends for REVLIMID.
The generic ingredient in REVLIMID is lenalidomide. There are fourteen drug master file entries for this API. Nineteen suppliers are listed for this generic product. Additional details are available on the lenalidomide profile page.
When can REVLIMID (lenalidomide) generic drug versions launch?
Generic name: lenalidomide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 03, 2026
Generic Entry Controlled by: Canada Patent 2,972,299
Patent Title: METHODES DE TRAITEMENT DE CERTAINES FORMES DE LEUCEMIE AU MOYEN DE 3-(4-AMINO-1-OXO-1,3-DIHYDRO-ISOINDOL-2-YL)-PIPERIDIN-2,6-DIONE (METHODS USING 3-(4-AMINO-1-OXO-1,3-DIHYDRO-ISOINDOL-2-YL)-PIPERIDINE-2,6-DIONE FOR TREATMENT OF CERTAIN LEUKEMIAS)
REVLIMID is a drug marketed by Bristol Myers Squibb. There are two patents protecting this drug and three Paragraph IV challenges.
This drug has three hundred and thirty-one patent family members in forty-one countries. There has been litigation on patents covering REVLIMID
See drug price trends for REVLIMID.
The generic ingredient in REVLIMID is lenalidomide. There are fourteen drug master file entries for this API. Nineteen suppliers are listed for this generic product. Additional details are available on the lenalidomide profile page.
When can TYMLOS (abaloparatide) generic drug versions launch?
Generic name: abaloparatide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 03, 2026
Generic Entry Controlled by: Canada Patent 2,664,734
Patent Title: PROCEDE D'ADMINISTRATION PHARMACOLOGIQUE D'UNE PROTEINE ANABOLIQUE OSSEUSE (METHOD OF DRUG DELIVERY FOR BONE ANABOLIC PROTEIN)

This drug has fifty-six patent family members in twenty-nine countries. There has been litigation on patents covering TYMLOS
See drug price trends for TYMLOS.
The generic ingredient in TYMLOS is abaloparatide. One supplier is listed for this generic product. Additional details are available on the abaloparatide profile page.
When can VOCABRIA (cabotegravir sodium) generic drug versions launch?
Generic name: cabotegravir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: Canada Patent 2,626,956
Patent Title: DERIVE DE CARBAMOYLPYRIDONE POLYCYCLIQUE AYANT UNE ACTIVITE D'INHIBITION SUR L'INTEGRASE DU VIH (POLYCYCLIC CARBAMOYLPYRIDONE DERIVATIVE HAVING INHIBITORY ACTIVITY ON HIV INTEGRASE)
VOCABRIA is a drug marketed by Viiv Hlthcare. There are two patents protecting this drug.
This drug has one hundred and twenty-six patent family members in thirty-three countries.
The generic ingredient in VOCABRIA is cabotegravir sodium. One supplier is listed for this generic product. Additional details are available on the cabotegravir sodium profile page.
When can BEVYXXA (betrixaban) generic drug versions launch?
Generic name: betrixaban
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 07, 2026
Generic Entry Controlled by: Canada Patent 2,627,086
Patent Title: NOUVEAUX SELS PHARMACEUTIQUES ET POLYMORPHES D'UN INHIBITEUR DU FACTEUR XA (PHARMACEUTICAL SALTS AND POLYMORPHS OF N- (5-CHL0R0-2-PYRIDINYL) -2- [ [4- [ (DIMETHYLAMINO) IMINOMETHYL] BENZOYL] AMINO] -5-METH OXY-BENZAMIDE, A FACTOR XA INHIBITOR)
BEVYXXA is a drug marketed by Portola Pharms Inc. There are five patents protecting this drug.
This drug has sixty-seven patent family members in twenty-six countries. There has been litigation on patents covering BEVYXXA
See drug price trends for BEVYXXA.
The generic ingredient in BEVYXXA is betrixaban. Additional details are available on the betrixaban profile page.
When can ENTRESTO SPRINKLE (sacubitril; valsartan) generic drug versions launch?
Generic name: sacubitril; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 08, 2026
Generic Entry Controlled by: Canada Patent 2,590,511
Patent Title: COMBINAISON PHARMACEUTIQUES D'UN ANTAGONISTE DE RECEPTEUR D'ANGIOTENSINE ET D'UN INHIBITEUR DE NEP (PHARMACEUTICAL COMBINATIONS OF AN ANGIOTENSIN RECEPTOR ANTAGONIST AND AN NEP INHIBITOR)
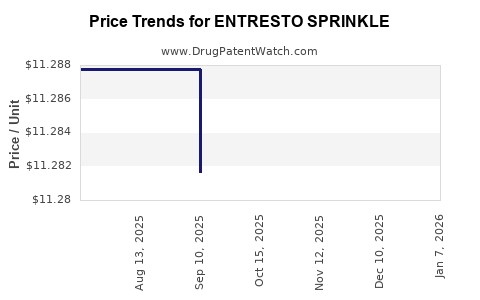
This drug has one hundred and eight patent family members in thirty-nine countries. There has been litigation on patents covering ENTRESTO SPRINKLE
See drug price trends for ENTRESTO SPRINKLE.
The generic ingredient in ENTRESTO SPRINKLE is sacubitril; valsartan. There are eleven drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the sacubitril; valsartan profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 10, 2026
Generic Entry Controlled by: Canada Patent 2,667,155
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 10, 2026
Generic Entry Controlled by: Canada Patent 2,667,460
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can SOFDRA (sofpironium bromide) generic drug versions launch?
Generic name: sofpironium bromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 13, 2026
Generic Entry Controlled by: Canada Patent 2,627,982
SOFDRA is a drug marketed by Botanix Sb. There are twenty patents protecting this drug.
This drug has one hundred and sixty patent family members in twenty-six countries. There has been litigation on patents covering SOFDRA
The generic ingredient in SOFDRA is sofpironium bromide. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the sofpironium bromide profile page.
When can AXUMIN (fluciclovine f-18) generic drug versions launch?
Generic name: fluciclovine f-18
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 28, 2026
Generic Entry Controlled by: Canada Patent 2,629,227
Patent Title: COMPOSE PRECURSEUR DE COMPOSE ORGANIQUE MARQUE A L'HALOGENE RADIOACTIF (PRECURSOR COMPOUND OF RADIOACTIVE HALOGEN LABELED ORGANIC COMPOUND)
AXUMIN is a drug marketed by Blue Earth. There are eight patents protecting this drug.
This drug has thirty patent family members in sixteen countries. There has been litigation on patents covering AXUMIN
The generic ingredient in AXUMIN is fluciclovine f-18. One supplier is listed for this generic product. Additional details are available on the fluciclovine f-18 profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 05, 2026
Generic Entry Controlled by: Canada Patent 2,668,512
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Canada Patent 2,671,744
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Canada Patent 2,696,824
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can LETAIRIS (ambrisentan) generic drug versions launch?
Generic name: ambrisentan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 12, 2026
Generic Entry Controlled by: Canada Patent 2,669,536
Patent Title: COMPOSITION DE TRAITEMENT DE L'HYPERTENSION PULMONAIRE (COMPOSITION FOR TREATING A PULMONARY HYPERTENSION)
LETAIRIS is a drug marketed by Gilead. There are three patents protecting this drug and one Paragraph IV challenge.
This drug has fifty-one patent family members in twenty-six countries. There has been litigation on patents covering LETAIRIS
See drug price trends for LETAIRIS.
The generic ingredient in LETAIRIS is ambrisentan. There are nine drug master file entries for this API. Thirteen suppliers are listed for this generic product. Additional details are available on the ambrisentan profile page.
When can XERMELO (telotristat etiprate) generic drug versions launch?
Generic name: telotristat etiprate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 12, 2026
Generic Entry Controlled by: Canada Patent 2,672,233
XERMELO is a drug marketed by Tersera. There are five patents protecting this drug.
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for XERMELO.
The generic ingredient in XERMELO is telotristat etiprate. One supplier is listed for this generic product. Additional details are available on the telotristat etiprate profile page.
When can PICATO (ingenol mebutate) generic drug versions launch?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 18, 2026
Generic Entry Controlled by: Canada Patent 2,634,073
Patent Title: COMPOSITIONS THERAPEUTIQUES (THERAPEUTIC COMPOSITIONS COMPRISING INGENOL-3-ANGELATE)

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can SEYSARA (sarecycline hydrochloride) generic drug versions launch?
Generic name: sarecycline hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 21, 2026
Generic Entry Controlled by: Canada Patent 2,673,486
Patent Title: UTILISATION DE COMPOSES A BASE DE TETRACYCLINE SUBSTITUEE DANS LE TRAITEMENT D'AFFECTIONS CUTANEES INFLAMMATOIRES (SUBSTITUTED TETRACYCLINE COMPOUNDS FOR TREATMENT OF INFLAMMATORY SKIN DISORDERS)
SEYSARA is a drug marketed by Almirall. There are four patents protecting this drug.
This drug has seventy-two patent family members in twenty countries.
See drug price trends for SEYSARA.
The generic ingredient in SEYSARA is sarecycline hydrochloride. One supplier is listed for this generic product. Additional details are available on the sarecycline hydrochloride profile page.
When can SEYSARA (sarecycline hydrochloride) generic drug versions launch?
Generic name: sarecycline hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 21, 2026
Generic Entry Controlled by: Canada Patent 2,892,739
Patent Title: UTILISATION DE COMPOSES A BASE DE TETRACYCLINE SUBSTITUEE DANS LE TRAITEMENT D'AFFECTIONS CUTANEES INFLAMMATOIRES (SUBSTITUTED TETRACYCLINE COMPOUNDS FOR TREATMENT OF INFLAMMATORY SKIN DISORDERS)
SEYSARA is a drug marketed by Almirall. There are four patents protecting this drug.
This drug has seventy-two patent family members in twenty countries.
See drug price trends for SEYSARA.
The generic ingredient in SEYSARA is sarecycline hydrochloride. One supplier is listed for this generic product. Additional details are available on the sarecycline hydrochloride profile page.
When can AURYXIA (ferric citrate) generic drug versions launch?
Generic name: ferric citrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 26, 2027
Generic Entry Controlled by: Canada Patent 2,640,763
Patent Title: METHODE PERMETTANT D'INVERSER, DE PREVENIR, DE RETARDER OU DE STABILISER LA CALCIFICATION DES TISSUS MOUS (METHOD OF REVERSING, PREVENTING, DELAYING OR STABILIZING SOFT TISSUE CALCIFICATION)

This drug has one hundred and twenty-two patent family members in twenty-three countries. There has been litigation on patents covering AURYXIA
See drug price trends for AURYXIA.
The generic ingredient in AURYXIA is ferric citrate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the ferric citrate profile page.
When can AURYXIA (ferric citrate) generic drug versions launch?
Generic name: ferric citrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 26, 2027
Generic Entry Controlled by: Canada Patent 2,640,974
Patent Title: METHODE DE TRAITEMENT DE NEPHROPATHIE CHRONIQUE (METHOD OF TREATING CHRONIC KIDNEY DISEASE)

This drug has one hundred and twenty-two patent family members in twenty-three countries. There has been litigation on patents covering AURYXIA
See drug price trends for AURYXIA.
The generic ingredient in AURYXIA is ferric citrate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the ferric citrate profile page.
When can NESINA (alogliptin benzoate) generic drug versions launch?
Generic name: alogliptin benzoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 01, 2027
Generic Entry Controlled by: Canada Patent 2,677,193
Patent Title: PREPARATION DE COMPRIME NE PROVOQUANT PAS DE PROBLEME DE PASTILLAGE (TABLET PREPARATION WITHOUT CAUSING A TABLETING TROUBLE)
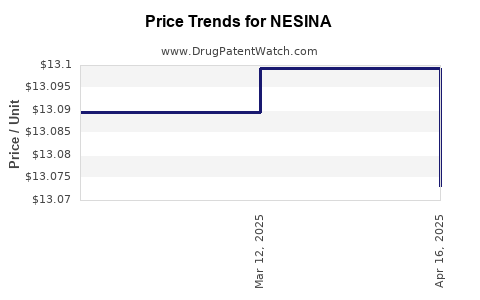
This drug has sixty-nine patent family members in thirty-seven countries. There has been litigation on patents covering NESINA
See drug price trends for NESINA.
The generic ingredient in NESINA is alogliptin benzoate. There are ten drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate profile page.
When can OSENI (alogliptin benzoate; pioglitazone hydrochloride) generic drug versions launch?
Generic name: alogliptin benzoate; pioglitazone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 01, 2027
Generic Entry Controlled by: Canada Patent 2,677,201

This drug has one hundred and one patent family members in forty-two countries. There has been litigation on patents covering OSENI
See drug price trends for OSENI.
The generic ingredient in OSENI is alogliptin benzoate; pioglitazone hydrochloride. There are ten drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate; pioglitazone hydrochloride profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Canada Patent 2,677,989
PANCREAZE is a drug marketed by
This drug has one hundred and one patent family members in forty-two countries.
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Canada Patent 2,677,989
ZENPEP is a drug marketed by
This drug has one hundred and one patent family members in forty-two countries.
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can XENLETA (lefamulin acetate) generic drug versions launch?
Generic name: lefamulin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 20, 2027
Generic Entry Controlled by: Canada Patent 2,678,795
XENLETA is a drug marketed by Hong Kong. There are four patents protecting this drug.
This drug has one hundred and twenty patent family members in thirty-six countries. There has been litigation on patents covering XENLETA
See drug price trends for XENLETA.
The generic ingredient in XENLETA is lefamulin acetate. Additional details are available on the lefamulin acetate profile page.
When can VARUBI (rolapitant hydrochloride) generic drug versions launch?
Generic name: rolapitant hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 22, 2027
Generic Entry Controlled by: Canada Patent 2,681,465
Patent Title: FORMULATIONS POUR COMPRIMES CONTENANT DES SELS DE 8-[{1-(3,5-BIS- (TRIFLUOROMETHYL)PHENYL)-ETHOXY}-METHYL]-8-PHENYL-1,7-DIAZA-SPIRO[4,5]- DECAN-2-ONE ET COMPRIMES (TABLET FORMULATIONS CONTAINING 8-[{1-(3,5-BIS-(TRIFLUOROMETHYL)PHENYL)-ETHOXY}-METHYL]-8-PHENYL-1,7-DIAZA-SPIRO[4.5]DECAN-2-ONE SALTS AND TABLETS MADE THEREFROM)
VARUBI is a drug marketed by Tersera. There are eight patents protecting this drug.
This drug has one hundred and fifty-eight patent family members in thirty-five countries.
The generic ingredient in VARUBI is rolapitant hydrochloride. One supplier is listed for this generic product. Additional details are available on the rolapitant hydrochloride profile page.
When can AMYVID (florbetapir f-18) generic drug versions launch?
Generic name: florbetapir f-18
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 26, 2027
Generic Entry Controlled by: Canada Patent 2,644,530
AMYVID is a drug marketed by Avid Radiopharms Inc. There are two patents protecting this drug.
This drug has fifty-one patent family members in thirty-three countries. There has been litigation on patents covering AMYVID
The generic ingredient in AMYVID is florbetapir f-18. One supplier is listed for this generic product. Additional details are available on the florbetapir f-18 profile page.
When can EDARBI (azilsartan kamedoxomil) generic drug versions launch?
Generic name: azilsartan kamedoxomil
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 28, 2027
Generic Entry Controlled by: Canada Patent 2,681,143
Patent Title: COMPOSITION PHARMACEUTIQUE SOLIDE COMPRENANT UN DERIVE DE BENZIMIDAZOLE-7-CARBOXYLATE ET UN AGENT DE CONTROLE DU PH (SOLID PHARMACEUTICAL COMPOSITION COMPRISING A BENZIMIDAZOLE-7-CARBOXYLATE DERIVATIVE AND A PH CONTROL AGENT)
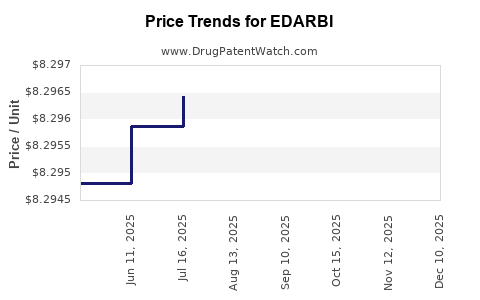
This drug has twenty-one patent family members in seventeen countries. There has been litigation on patents covering EDARBI
See drug price trends for EDARBI.
The generic ingredient in EDARBI is azilsartan kamedoxomil. There are six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the azilsartan kamedoxomil profile page.
When can XTANDI (enzalutamide) generic drug versions launch?
Generic name: enzalutamide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 29, 2027
Generic Entry Controlled by: Canada Patent 2,648,139
Patent Title: COMPOSES DE DIARYLTHIOHYDANTOINE ET LEUR UTILISATION DANS LE TRAITEMENT DE TROUBLES D'HYPERPROLIFERATION (DIARYL THIOHYDANTOIN COMPOUNDS AND THEIR USE IN THE TREATMENT OF HYPERPROLIFERATIVE DISORDERS)
XTANDI is a drug marketed by Astellas. There are seven patents protecting this drug and one Paragraph IV challenge. Three tentatively approved generics are ready to enter the market.
This drug has one hundred and ninety-two patent family members in thirty-six countries. There has been litigation on patents covering XTANDI
See drug price trends for XTANDI.
The generic ingredient in XTANDI is enzalutamide. There are nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the enzalutamide profile page.
When can VARUBI (rolapitant hydrochloride) generic drug versions launch?
Generic name: rolapitant hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 04, 2027
Generic Entry Controlled by: Canada Patent 2,648,604
VARUBI is a drug marketed by Tersera. There are eight patents protecting this drug.
This drug has one hundred and fifty-eight patent family members in thirty-five countries.
The generic ingredient in VARUBI is rolapitant hydrochloride. One supplier is listed for this generic product. Additional details are available on the rolapitant hydrochloride profile page.
When can INLYTA (axitinib) generic drug versions launch?
Generic name: axitinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 05, 2027
Generic Entry Controlled by: Canada Patent 2,682,859
INLYTA is a drug marketed by Pf Prism Cv. There are four patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has ninety-seven patent family members in thirty countries. There has been litigation on patents covering INLYTA
See drug price trends for INLYTA.
The generic ingredient in INLYTA is axitinib. There are four drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the axitinib profile page.
When can SYMDEKO (COPACKAGED) (ivacaftor; ivacaftor, tezacaftor) generic drug versions launch?
Generic name: ivacaftor; ivacaftor, tezacaftor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 09, 2027
Generic Entry Controlled by: Canada Patent 2,648,719
Patent Title: MODULATEURS DES TRANSPORTEURS DE CASSETTES DE LIAISON DE L'ATP (MODULATORS OF ATP-BINDING CASSETTE TRANSPORTERS)
SYMDEKO (COPACKAGED) is a drug marketed by Vertex Pharms Inc. There are twenty-six patents protecting this drug.
This drug has four hundred and twenty-eight patent family members in forty-one countries. There has been litigation on patents covering SYMDEKO (COPACKAGED)
The generic ingredient in SYMDEKO (COPACKAGED) is ivacaftor; ivacaftor, tezacaftor. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivacaftor; ivacaftor, tezacaftor profile page.
When can SYMDEKO (COPACKAGED) (ivacaftor; ivacaftor, tezacaftor) generic drug versions launch?
Generic name: ivacaftor; ivacaftor, tezacaftor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 09, 2027
Generic Entry Controlled by: Canada Patent 2,869,945
Patent Title: MODULATEURS DES TRANSPORTEURS DE CASSETTES DE LIAISON DE L'ATP (MODULATORS OF ATP-BINDING CASSETTE TRANSPORTERS)
SYMDEKO (COPACKAGED) is a drug marketed by Vertex Pharms Inc. There are twenty-six patents protecting this drug.
This drug has four hundred and twenty-eight patent family members in forty-one countries. There has been litigation on patents covering SYMDEKO (COPACKAGED)
The generic ingredient in SYMDEKO (COPACKAGED) is ivacaftor; ivacaftor, tezacaftor. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivacaftor; ivacaftor, tezacaftor profile page.
When can QVAR REDIHALER (beclomethasone dipropionate) generic drug versions launch?
Generic name: beclomethasone dipropionate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 11, 2027
Generic Entry Controlled by: Canada Patent 2,682,528