Expiring Drug Patents Cheat Sheet
We analyse the patents covering drugs in 134 countries and quickly give you the likely loss-of-exclusivity/generic entry date
Mexico: These 43 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "Mexico: These 43 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027" DrugPatentWatch.com thinkBiotech, 2026 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 12, 2026
Generic Entry Controlled by: Mexico Patent 2,008,015,900
Patent Title: POLIMORFOS DE N-HIDROXI-3-[4-[[[2-(2-METIL-1H-INDOL-3-IL)ETIL]AMIN O]METIL]FENIL]-2E-2-PROPENAMIDA. (POLYMORPHS OF N-HYDROXY-3-[4-[[[2-(2-METHYL-1H-INDOL-3-YL)ETHYL]A MINO]METHYL]PHENYL]-2E-2-PROPENAMIDE.)
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 15, 2026
Generic Entry Controlled by: Mexico Patent 2,009,002,757
Patent Title: FORMULACION DE FARMACO RESISTENTE AL ABUSO. (ABUSE RESISTANT DRUG FORMULATION.)
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 15, 2026
Generic Entry Controlled by: Mexico Patent 2,010,002,780
Patent Title: FORMULACION DE FARMACO RESISTENTE AL ABUSO. (ABUSE RESISTANT DRUG FORMULATION.)
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 15, 2026
Generic Entry Controlled by: Mexico Patent 336,861
Patent Title: FORMULACION DE FARMACO RESISTENTE AL ABUSO. (ABUSE RESISTANT DRUG FORMULATION.)
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Mexico Patent 2,008,016,372
Patent Title: COMPOSICIONES FARMACEUTICAS. (PHARMACEUTICAL COMPOSITIONS.)
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Mexico Patent 2,008,015,704
Patent Title: PROCESO PARA LA PRODUCCION DE 2-[4-(3-Y 2-FLUORO BENCILOXI) BENCILAMINO]PROPANAMIDAS. (PROCESS FOR THE PRODUCTION OF 2- [4 - ( 3- AND 2-FLU0R0BENZYL0XY) BENZYLAMIN0] PROPAN AMIDES.)

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Mexico Patent 342,698
Patent Title: PROCESO PARA LA PRODUCCION DE 2-[4-(3- Y 2-FLUOROBENCILOXI) BENCILAMINO] PROPANAMIDAS. (PROCESS FOR THE PRODUCTION OF 2- [4 - ( 3- AND 2-FLU0R0BENZYL0XY) BENZYLAMIN0] PROPAN AMIDES.)

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can JESDUVROQ (daprodustat) generic drug versions launch?
Generic name: daprodustat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: Mexico Patent 2,009,000,165
Patent Title: INHIBIDORES DE PROLIL HIDROXILASA. (PROLYL HYDROXYLASE INHIBITORS.)
JESDUVROQ is a drug marketed by Glaxosmithkline. There are six patents protecting this drug.
This drug has forty-four patent family members in thirty-one countries.
See drug price trends for JESDUVROQ.
The generic ingredient in JESDUVROQ is daprodustat. One supplier is listed for this generic product. Additional details are available on the daprodustat profile page.
When can TEKTURNA HCT (aliskiren hemifumarate; hydrochlorothiazide) generic drug versions launch?
Generic name: aliskiren hemifumarate; hydrochlorothiazide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: Mexico Patent 2,008,016,533
Patent Title: FORMULACIONES GALENICAS DE ALISQUIRENO E HIDROCLOROTIAZIDA. (GALENICAL FORMULATIONS OF ALISKIREN AND HYDROCHLOROTHIAZIDE.)

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering TEKTURNA HCT
See drug price trends for TEKTURNA HCT.
The generic ingredient in TEKTURNA HCT is aliskiren hemifumarate; hydrochlorothiazide. There are four drug master file entries for this API. Additional details are available on the aliskiren hemifumarate; hydrochlorothiazide profile page.
When can EXFORGE HCT (amlodipine besylate; hydrochlorothiazide; valsartan) generic drug versions launch?
Generic name: amlodipine besylate; hydrochlorothiazide; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 27, 2026
Generic Entry Controlled by: Mexico Patent 2,008,016,532

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering EXFORGE HCT
See drug price trends for EXFORGE HCT.
The generic ingredient in EXFORGE HCT is amlodipine besylate; hydrochlorothiazide; valsartan. There are fifty drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the amlodipine besylate; hydrochlorothiazide; valsartan profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Mexico Patent 2,008,015,377

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Mexico Patent 339,143

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Mexico Patent 367,155

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Mexico Patent 2,008,015,377

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Mexico Patent 339,143

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Mexico Patent 367,155

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Mexico Patent 2,008,015,377

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Mexico Patent 339,143

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Mexico Patent 367,155

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Mexico Patent 2,009,008,935
Patent Title: MODULADORES DE LAS PROPIEDADES FARMACOCINETICAS DE PRODUCTOS TERAPEUTICOS. (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS.)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can BYFAVO (remimazolam besylate) generic drug versions launch?
Generic name: remimazolam besylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 10, 2026
Generic Entry Controlled by: Mexico Patent 2,009,000,404
Patent Title: SALES DE BENZODIAZEPINA DE ACCION CORTA Y SUS FORMAS POLIMORFICAS. (SHORT-ACTING BENZODIAZEPINE SALTS AND THEIR POLYMORPHIC FORMS.)
BYFAVO is a drug marketed by Acacia. There are eleven patents protecting this drug.
This drug has fifty-three patent family members in twenty-four countries. There has been litigation on patents covering BYFAVO
See drug price trends for BYFAVO.
The generic ingredient in BYFAVO is remimazolam besylate. One supplier is listed for this generic product. Additional details are available on the remimazolam besylate profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Mexico Patent 2,009,000,319
Patent Title: COMBINACION DE ADAPALENO Y PEROXIDO DE BENZOILO PARA TRATAMIENTO DE LESIONES DEL ACNE. (COMBINATION OF ADAPALENE AND BENZOYL PEROXIDE FOR TREATING ACNE LESIONS.)
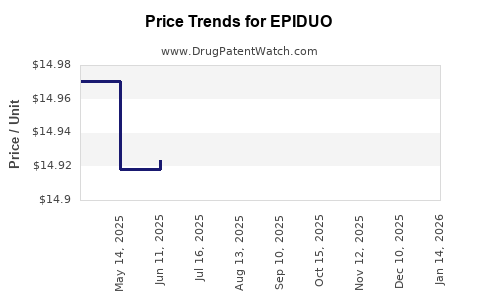
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Mexico Patent 2,009,000,319
Patent Title: COMBINACION DE ADAPALENO Y PEROXIDO DE BENZOILO PARA TRATAMIENTO DE LESIONES DEL ACNE. (COMBINATION OF ADAPALENE AND BENZOYL PEROXIDE FOR TREATING ACNE LESIONS.)
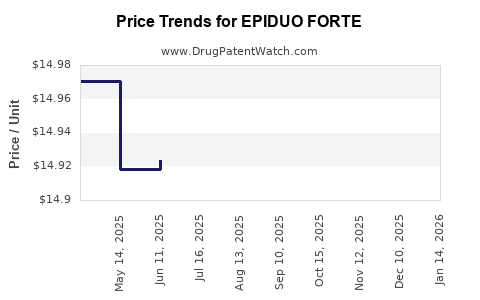
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO FORTE
See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can RASUVO (methotrexate) generic drug versions launch?
Generic name: methotrexate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 21, 2026
Generic Entry Controlled by: Mexico Patent 2,009,000,812
Patent Title: SOLUCIONES DE METOTREXATO CONCENTRADAS. (CONCENTRATED METHOTREXATE SOLUTIONS.)
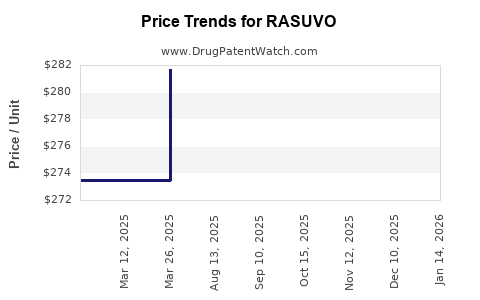
This drug has twenty-nine patent family members in twenty-one countries. There has been litigation on patents covering RASUVO
See drug price trends for RASUVO.
The generic ingredient in RASUVO is methotrexate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the methotrexate profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: Mexico Patent 2,009,001,426

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 15, 2026
Generic Entry Controlled by: Mexico Patent 2,008,001,557
Patent Title: NUCLEOS DE MICROGLOBULOS DE PANCREATIVA ADECUADOS PARA REVESTIMIENTO ENTERICO. (PANCREATIN MICROPELLET CORES SUITABLE FOR ENTERIC COATING.)
CREON is a drug marketed by
This drug has ninety-three patent family members in thirty-one countries.
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 15, 2026
Generic Entry Controlled by: Mexico Patent 2,008,001,558
Patent Title: COMPOSICIONES FARMACEUTICAS DE DESPRENDIMIENTO CONTROLADO PARA FARMACOS LABILES EN MEDIO ACIDO. (CONTROLLED RELEASE PHARMACEUTICAL COMPOSITIONS FOR ACID LABILE DRUGS.)
CREON is a drug marketed by
This drug has ninety-three patent family members in thirty-one countries.
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 21, 2026
Generic Entry Controlled by: Mexico Patent 340,403
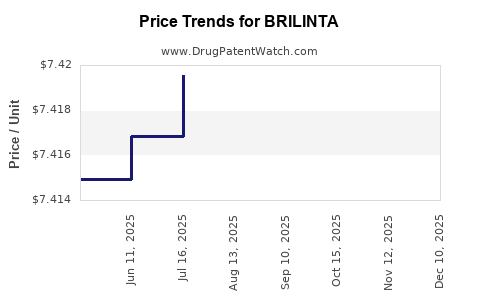
This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 29, 2026
Generic Entry Controlled by: Mexico Patent 2,009,002,057
Patent Title: COMPOSICIONES TERAPEUTICAS QUE COMPRENDEN UN ANTAGONISTA DEL RECEPTOR DE ENDOTELINA ESPECIFICO Y UN INHIBIDOR PDE5. (THERAPEUTIC COMPOSITIONS.)
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the macitentan profile page.
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 22, 2026
Generic Entry Controlled by: Mexico Patent 2,008,003,882
Patent Title: FORMULACION DE CAPSULA DE PIRFENIDONA Y EXCIPIENTES FARMACEUTICAMENTE ACEPTABLES. (CAPSULE FORMULATION OF PIRFENIDONE AND PHARMACEUTICALLY ACCEPTABLE EXCIPIENTS.)
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can INREBIC (fedratinib hydrochloride) generic drug versions launch?
Generic name: fedratinib hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: Mexico Patent 391,408
INREBIC is a drug marketed by Bristol-myers. There are five patents protecting this drug.
This drug has one hundred and nineteen patent family members in forty-two countries. There has been litigation on patents covering INREBIC
See drug price trends for INREBIC.
The generic ingredient in INREBIC is fedratinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the fedratinib hydrochloride profile page.
When can ENTRESTO SPRINKLE (sacubitril; valsartan) generic drug versions launch?
Generic name: sacubitril; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 08, 2026
Generic Entry Controlled by: Mexico Patent 2,007,008,075
Patent Title: COMBINACIONES FARMACEUTICAS DE UN ANTAGONISTA DEL RECEPTOR DE ANGIOTENSINA Y UN INHIBIDOR DE ENDOPEPTIDASA NEUTRAL (NEP). (PHARMACEUTICAL COMBINATIONS OF AN ANGIOTENSIN RECEPTOR ANTAGONIST AND AN NEP INHIBITOR.)
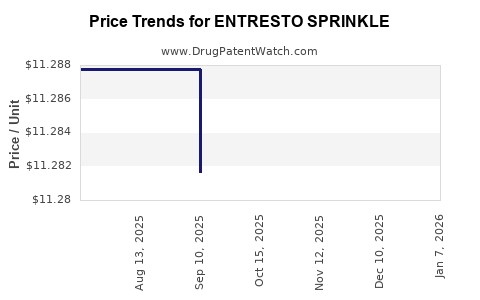
This drug has one hundred and eight patent family members in thirty-nine countries. There has been litigation on patents covering ENTRESTO SPRINKLE
See drug price trends for ENTRESTO SPRINKLE.
The generic ingredient in ENTRESTO SPRINKLE is sacubitril; valsartan. There are eleven drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the sacubitril; valsartan profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 10, 2026
Generic Entry Controlled by: Mexico Patent 2,009,005,000
Patent Title: AMIDAS DE PEPTIDOS SINTETICOS. (SYNTHETIC PEPTIDE AMIDES.)
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can ZTALMY (ganaxolone) generic drug versions launch?
Generic name: ganaxolone
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 28, 2026
Generic Entry Controlled by: Mexico Patent 2,008,006,888
Patent Title: FORMULAS Y METODOS PARA LA MANUFACTURA Y USO DE LA GANAXOLONA. (GANAXOLONE FORMULATIONS AND METHODS FOR THE MAKING AND USE THEREOF.)
ZTALMY is a drug marketed by Immedica Pharma. There are eleven patents protecting this drug.
This drug has forty-nine patent family members in sixteen countries. There has been litigation on patents covering ZTALMY
See drug price trends for ZTALMY.
The generic ingredient in ZTALMY is ganaxolone. One supplier is listed for this generic product. Additional details are available on the ganaxolone profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 05, 2026
Generic Entry Controlled by: Mexico Patent 2,009,005,909
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Mexico Patent 2,009,006,081
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Mexico Patent 2,010,002,336
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can XERMELO (telotristat etiprate) generic drug versions launch?
Generic name: telotristat etiprate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 12, 2026
Generic Entry Controlled by: Mexico Patent 2,009,006,195
Patent Title: COMPUESTOS BASADOS EN 4-FENIL-6-(2,2,2-TRIFLUORO-1-FENILETOXI)PIRI MIDINA Y METODOS DE SU USO. (4-PHENYL-6-(2,2,2-TRIFLUORO-1-PHENYLETHOXY)PYRIMIDINE-BASED COMPOUNDS AND METHODS OF THEIR USE.)
XERMELO is a drug marketed by Tersera. There are five patents protecting this drug.
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for XERMELO.
The generic ingredient in XERMELO is telotristat etiprate. One supplier is listed for this generic product. Additional details are available on the telotristat etiprate profile page.
When can PICATO (ingenol mebutate) generic drug versions launch?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 18, 2026
Generic Entry Controlled by: Mexico Patent 2,008,007,685
Patent Title: COMPOSICIONES TERAPEUTICAS. (THERAPEUTIC COMPOSITIONS COMPRISING INGENOL-3-ANGELATE.)

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can OSENI (alogliptin benzoate; pioglitazone hydrochloride) generic drug versions launch?
Generic name: alogliptin benzoate; pioglitazone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 01, 2027
Generic Entry Controlled by: Mexico Patent 2,009,008,100
Patent Title: PREPARACION SOLIDA QUE COMPRENDE ALOGLIPTINA Y PIOGLITAZONA. (SOLID PREPARATION COMPRISING ALOGLIPTIN AND PIOGLITAZONE.)

This drug has one hundred and one patent family members in forty-two countries. There has been litigation on patents covering OSENI
See drug price trends for OSENI.
The generic ingredient in OSENI is alogliptin benzoate; pioglitazone hydrochloride. There are ten drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate; pioglitazone hydrochloride profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Mexico Patent 2,009,008,828
Patent Title: COMPOSICIONES DE ENZIMAS DIGESTIVAS ESTABLES. (STABLE DIGESTIVE ENZYME COMPOSITIONS.)
PANCREAZE is a drug marketed by
This drug has one hundred and one patent family members in forty-two countries.
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Mexico Patent 2,009,008,828
Patent Title: COMPOSICIONES DE ENZIMAS DIGESTIVAS ESTABLES. (STABLE DIGESTIVE ENZYME COMPOSITIONS.)
ZENPEP is a drug marketed by
This drug has one hundred and one patent family members in forty-two countries.
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can VARUBI (rolapitant hydrochloride) generic drug versions launch?
Generic name: rolapitant hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 22, 2027
Generic Entry Controlled by: Mexico Patent 2,009,010,210
Patent Title: FORMULACIONES DE COMPRIMIDOS QUE CONTIENEN SALES DE 8-[{1-(3,5-BIS-(TRIFLUOROMETIL)FENIL)-ETOXI}-METIL]-8-FENIL-1,7-D IAZA-SPIRO[4.5]DECAN-2-ONA Y COMPRIMIDOS ELABORADOS A PARTIR DE ESTAS. (TABLET FORMULATIONS CONTAINING 8-[{1-(3,5-BIS-(TRIFLUOROMETHYL)PH ENYL)-ETHOXY}-METHYL]-8-PHENYL-1,7-DIAZA-SPIRO[4.5]DECAN-2-ONE SALTS AND TABLETS MADE THEREFROM.)
VARUBI is a drug marketed by Tersera. There are eight patents protecting this drug.
This drug has one hundred and fifty-eight patent family members in thirty-five countries.
The generic ingredient in VARUBI is rolapitant hydrochloride. One supplier is listed for this generic product. Additional details are available on the rolapitant hydrochloride profile page.
When can AMYVID (florbetapir f-18) generic drug versions launch?
Generic name: florbetapir f-18
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 26, 2027
Generic Entry Controlled by: Mexico Patent 2,008,012,527
AMYVID is a drug marketed by Avid Radiopharms Inc. There are two patents protecting this drug.
This drug has fifty-one patent family members in thirty-three countries. There has been litigation on patents covering AMYVID
The generic ingredient in AMYVID is florbetapir f-18. One supplier is listed for this generic product. Additional details are available on the florbetapir f-18 profile page.
When can SYMDEKO (COPACKAGED) (ivacaftor; ivacaftor, tezacaftor) generic drug versions launch?
Generic name: ivacaftor; ivacaftor, tezacaftor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 09, 2027
Generic Entry Controlled by: Mexico Patent 2,008,012,945
Patent Title: MODULADORES DE TRANSPORTADORES DE CARTUCHO DE UNION A ADENOSINA-TRIFOSFATO CELULAR (ATP). (MODULATORS OF ATP-BINDING CASSETTE TRANSPORTERS.)
SYMDEKO (COPACKAGED) is a drug marketed by Vertex Pharms Inc. There are twenty-six patents protecting this drug.
This drug has four hundred and twenty-eight patent family members in forty-one countries. There has been litigation on patents covering SYMDEKO (COPACKAGED)
The generic ingredient in SYMDEKO (COPACKAGED) is ivacaftor; ivacaftor, tezacaftor. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivacaftor; ivacaftor, tezacaftor profile page.
When can QVAR REDIHALER (beclomethasone dipropionate) generic drug versions launch?
Generic name: beclomethasone dipropionate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 11, 2027
Generic Entry Controlled by: Mexico Patent 2,009,010,600
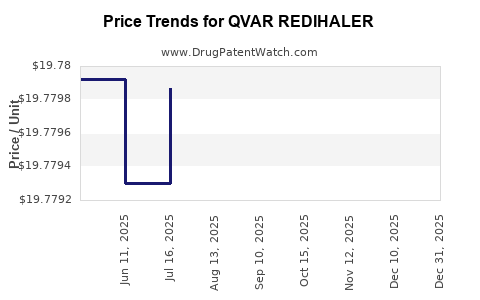
This drug has two hundred and seventy-eight patent family members in twenty-seven countries. There has been litigation on patents covering QVAR REDIHALER
See drug price trends for QVAR REDIHALER.
The generic ingredient in QVAR REDIHALER is beclomethasone dipropionate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the beclomethasone dipropionate profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 03, 2027
Generic Entry Controlled by: Mexico Patent 2,009,011,881
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can TRELSTAR (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 06, 2027
Generic Entry Controlled by: Mexico Patent 2,009,012,856
Patent Title: COMPOSICION FARMACEUTICA DE LIBERACION LENTA HECHA DE MICROPARTICULAS. (SLOW RELEASE PHARMACEUTICAL COMPOSITION MADE OF MICROPARTICLES.)
TRELSTAR is a drug marketed by Verity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
See drug price trends for TRELSTAR.
The generic ingredient in TRELSTAR is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can TRIPTODUR KIT (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 06, 2027
Generic Entry Controlled by: Mexico Patent 2,009,012,856
Patent Title: COMPOSICION FARMACEUTICA DE LIBERACION LENTA HECHA DE MICROPARTICULAS. (SLOW RELEASE PHARMACEUTICAL COMPOSITION MADE OF MICROPARTICLES.)
TRIPTODUR KIT is a drug marketed by Azurity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
The generic ingredient in TRIPTODUR KIT is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can JAKAFI (ruxolitinib phosphate) generic drug versions launch?
Generic name: ruxolitinib phosphate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 13, 2027
Generic Entry Controlled by: Mexico Patent 2,009,013,402
Patent Title: SALES DE INHIBIDOR DE JANUS CINASA (R)-3-(4-(7H-PIRROLO[2,3-D]PIRI MIDIN-4-IL)-1H-PIRAZOL-1-IL)-3-CICLOPENTILPROPANITRILO. (SALTS OF THE JANUS KINASE INHIBITOR (R)-3-(4-(7H-PYRROLO[2,3-D]PY RIMIDIN-4-YL)-1H-PYRAZOL-1-YL)-3-CYCLOPENTYLPROPANENITRILE.)
JAKAFI is a drug marketed by Incyte Corp. There are eight patents protecting this drug and one Paragraph IV challenge.
This drug has two hundred and thirty-six patent family members in forty-six countries. There has been litigation on patents covering JAKAFI
See drug price trends for JAKAFI.
The generic ingredient in JAKAFI is ruxolitinib phosphate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ruxolitinib phosphate profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 15, 2027
Generic Entry Controlled by: Mexico Patent 2,008,016,141
Patent Title: 1-[2-(2,4-DIMETILFENILSULFANIL)-FENIL] PIPERAZINA COMO UN COMPUESTO CON ACTIVIDAD COMBINADA DE RECAPTACIÓN DE SEROTONINA, 5-HT3 Y 5-HT1A.PARA EL TRATAMIENTO DE DAÑO COGNITIVO. (1- [2- (2, 4-DIMETHYLPHENYLSULFANYL) -PHENYL] PIPERAZINE AS A COMPOUND WITH COMBINED SEROTONIN REUPTAKE, 5-HT3 AND 5-HT1A ACTIVITY FOR THE TREATMENT OF COGNITIVE IMPAIRMENT.)

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2027
Generic Entry Controlled by: Mexico Patent 2,009,013,664
Patent Title: COMPOSICION, METODO Y PREPARACION DEL ACIDO BILIAR SINTETICO. (SYNTHETIC BILE ACID COMPOSITION, METHOD, AND PREPARATION.)
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2027
Generic Entry Controlled by: Mexico Patent 360,094
Patent Title: COMPOSICIÓN, MÉTODO Y PREPARACIÓN DEL ÁCIDO BILIAR SINTÉTICO. (SYNTHETIC BILE ACID COMPOSITION, METHOD, AND PREPARATION.)
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can CARDAMYST (etripamil) generic drug versions launch?
Generic name: etripamil
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 20, 2027
Generic Entry Controlled by: Mexico Patent 347,325
Patent Title: BLOQUEADORES DEL CANAL DE CALCIO DE FENILALQUILAMINA DE ACCION CORTA Y SUS USOS. (SHORT ACTING PHENYLALKYLAMINE CALCIUM CHANNEL BLOCKERS AND USES THEREOF.)
CARDAMYST is a drug marketed by Milestone Pharms Usa. There are six patents protecting this drug.
This drug has sixty-six patent family members in twenty-nine countries.
The generic ingredient in CARDAMYST is etripamil. One supplier is listed for this generic product. Additional details are available on the etripamil profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 06, 2027
Generic Entry Controlled by: Mexico Patent 2,009,000,234
Patent Title: MODULADORES DE LAS PROPIEDADES FARMACOCINETICAS DE PRODUCTOS TERAPEUTICOS. (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS.)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
Mexico Drug Market Analysis: Regulatory Landscape and Market Dynamics
More… ↓
DrugPatentWatch cited by CNN, NEJM, Nature Journals, and more …

Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
