Expiring Drug Patents Cheat Sheet
We analyse the patents covering drugs in 134 countries and quickly give you the likely loss-of-exclusivity/generic entry date
Eurasian Patent Organization: These 31 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "Eurasian Patent Organization: These 31 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027" DrugPatentWatch.com thinkBiotech, 2026 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
When can FARYDAK (panobinostat lactate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 12, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 17,984
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 12, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,802,383
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch in the Eurasian Patent Organization?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 16, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 15,287
Patent Title: 1-[2-(2,4-ДИМЕТИЛФЕНИЛСУЛЬФАНИЛ)ФЕНИЛ]ПИПЕРАЗИН В КАЧЕСТВЕ СОЕДИНЕНИЯ С СОЧЕТАНИЕМ АКТИВНОСТИ В ОТНОШЕНИИ ПОВТОРНОГО ЗАХВАТА СЕРОТОНИНА, 5-НТИ 5-НТДЛЯ ЛЕЧЕНИЯ КОГНИТИВНОГО НАРУШЕНИЯ (1- [2- (2, 4-DIMETHYLPHENYLSULFANYL) -PHENYL] PIPERAZINE AS A COMPOUND WITH COMBINED SEROTONIN REUPTAKE, 5-HTAND 5-HTACTIVITY FOR THE TREATMENT OF COGNITIVE IMPAIRMENT)

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch in the Eurasian Patent Organization?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 16, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 17,058
Patent Title: НОВОЕ ТЕРАПЕВТИЧЕСКОЕ ПРИМЕНЕНИЕ 1-[2-(2,4-ДИМЕТИЛФЕНИЛСУЛЬФАНИЛ)ФЕНИЛ]ПИПЕРАЗИНА (NOVEL THERAPEUTIC USES OF 1-[2-(2,4-DIMETHYLPHENYLSULFANYL)PHENYL]PIPERAZINE)

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch in the Eurasian Patent Organization?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 16, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,970,018
Patent Title: 1-[2-(2,4-ДИМЕТИЛФЕНИЛСУЛЬФАНИЛ)ФЕНИЛ]ПИПЕРАЗИН В КАЧЕСТВЕ СОЕДИНЕНИЯ С СОЧЕТАНИЕМ АКТИВНОСТИ В ОТНОШЕНИИ ПОВТОРНОГО ЗАХВАТА СЕРОТОНИНА, 5-НТ3 И 5-НТ1А ДЛЯ ЛЕЧЕНИЯ КОГНИТИВНОГО НАРУШЕНИЯ

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch in the Eurasian Patent Organization?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 16, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,970,870
Patent Title: НОВОЕ ТЕРАПЕВТИЧЕСКОЕ ПРИМЕНЕНИЕ 1-[2-(2,4-ДИМЕТИЛФЕНИЛСУЛЬФАНИЛ)ФЕНИЛ]ПИПЕРАЗИНА

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 17,123

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 25,326

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,970,028

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,101,308

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can JESDUVROQ (daprodustat) generic drug versions launch in the Eurasian Patent Organization?
Generic name: daprodustat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 18,220
JESDUVROQ is a drug marketed by Glaxosmithkline. There are six patents protecting this drug.
This drug has forty-four patent family members in thirty-one countries.
See drug price trends for JESDUVROQ.
The generic ingredient in JESDUVROQ is daprodustat. One supplier is listed for this generic product. Additional details are available on the daprodustat profile page.
When can JESDUVROQ (daprodustat) generic drug versions launch in the Eurasian Patent Organization?
Generic name: daprodustat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,970,050
JESDUVROQ is a drug marketed by Glaxosmithkline. There are six patents protecting this drug.
This drug has forty-four patent family members in thirty-one countries.
See drug price trends for JESDUVROQ.
The generic ingredient in JESDUVROQ is daprodustat. One supplier is listed for this generic product. Additional details are available on the daprodustat profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch in the Eurasian Patent Organization?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 18,229

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch in the Eurasian Patent Organization?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 20,428

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch in the Eurasian Patent Organization?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 28,259

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch in the Eurasian Patent Organization?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 35,999

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch in the Eurasian Patent Organization?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,900,066

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch in the Eurasian Patent Organization?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,171,333

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch in the Eurasian Patent Organization?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,490,902

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch in the Eurasian Patent Organization?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,791,254

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch in the Eurasian Patent Organization?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 202,091,391

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 18,229

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 20,428

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 28,259

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 35,999

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,900,066

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,171,333

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,490,902

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,791,254

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 202,091,391

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 18,229

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 20,428

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 28,259

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 35,999

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,900,066

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,171,333

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,490,902

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,791,254

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 202,091,391

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch in the Eurasian Patent Organization?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 19,893
Patent Title: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ И СПОСОБ ЛЕЧЕНИЯ ВИЧ-ИНФЕКЦИИ (PHARMACEUTICAL COMPOSITION AND METHOD FOR TREATING HIV INFECTION)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch in the Eurasian Patent Organization?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 20,489
Patent Title: МОДУЛЯТОРЫ ФАРМАКОКИНЕТИЧЕСКИХ СВОЙСТВ ЛЕКАРСТВЕННЫХ СРЕДСТВ (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch in the Eurasian Patent Organization?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 25,845
Patent Title: МОДУЛЯТОРЫ ФАРМАКОКИНЕТИЧЕСКИХ СВОЙСТВ ЛЕКАРСТВЕННЫХ СРЕДСТВ (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch in the Eurasian Patent Organization?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,900,155
Patent Title: МОДУЛЯТОРЫ ФАРМАКОКИНЕТИЧЕСКИХ СВОЙСТВ ЛЕКАРСТВЕННЫХ СРЕДСТВ

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch in the Eurasian Patent Organization?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,901,155
Patent Title: МОДУЛЯТОРЫ ФАРМАКОКИНЕТИЧЕСКИХ СВОЙСТВ ТЕРАПЕВТИЧЕСКИХ ПРЕПАРАТОВ (ВАРИАНТЫ) И ИХ ИСПОЛЬЗОВАНИЕ В ТЕРАПИИ

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch in the Eurasian Patent Organization?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,270,738
Patent Title: МОДУЛЯТОРЫ ФАРМАКОКИНЕТИЧЕСКИХ СВОЙСТВ ЛЕКАРСТВЕННЫХ СРЕДСТВ

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can OLYSIO (simeprevir sodium) generic drug versions launch in the Eurasian Patent Organization?
Generic name: simeprevir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 15,131
Patent Title: МАКРОЦИКЛИЧЕСКИЕ ИНГИБИТОРЫ ВИРУСА ГЕПАТИТА С (MACROCYCLIC INHIBITORS OF HEPATITIS C VIRUS)
OLYSIO is a drug marketed by Janssen Prods. There are eight patents protecting this drug.
This drug has sixty-four patent family members in forty-one countries.
See drug price trends for OLYSIO.
The generic ingredient in OLYSIO is simeprevir sodium. There is one drug master file entry for this API. Additional details are available on the simeprevir sodium profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 15,756

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,900,298

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can ESBRIET (pirfenidone) generic drug versions launch in the Eurasian Patent Organization?
Generic name: pirfenidone
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 22, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,800,881
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can COTELLIC (cobimetinib fumarate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: cobimetinib fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 05, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 19,983
COTELLIC is a drug marketed by Genentech Inc. There are seven patents protecting this drug.
This drug has two hundred and seven patent family members in forty-six countries.
See drug price trends for COTELLIC.
The generic ingredient in COTELLIC is cobimetinib fumarate. One supplier is listed for this generic product. Additional details are available on the cobimetinib fumarate profile page.
When can COTELLIC (cobimetinib fumarate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: cobimetinib fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 05, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 32,466
COTELLIC is a drug marketed by Genentech Inc. There are seven patents protecting this drug.
This drug has two hundred and seven patent family members in forty-six countries.
See drug price trends for COTELLIC.
The generic ingredient in COTELLIC is cobimetinib fumarate. One supplier is listed for this generic product. Additional details are available on the cobimetinib fumarate profile page.
When can COTELLIC (cobimetinib fumarate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: cobimetinib fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 05, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,801,041
COTELLIC is a drug marketed by Genentech Inc. There are seven patents protecting this drug.
This drug has two hundred and seven patent family members in forty-six countries.
See drug price trends for COTELLIC.
The generic ingredient in COTELLIC is cobimetinib fumarate. One supplier is listed for this generic product. Additional details are available on the cobimetinib fumarate profile page.
When can COTELLIC (cobimetinib fumarate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: cobimetinib fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 05, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,400,111
COTELLIC is a drug marketed by Genentech Inc. There are seven patents protecting this drug.
This drug has two hundred and seven patent family members in forty-six countries.
See drug price trends for COTELLIC.
The generic ingredient in COTELLIC is cobimetinib fumarate. One supplier is listed for this generic product. Additional details are available on the cobimetinib fumarate profile page.
When can COTELLIC (cobimetinib fumarate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: cobimetinib fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 05, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,691,142
COTELLIC is a drug marketed by Genentech Inc. There are seven patents protecting this drug.
This drug has two hundred and seven patent family members in forty-six countries.
See drug price trends for COTELLIC.
The generic ingredient in COTELLIC is cobimetinib fumarate. One supplier is listed for this generic product. Additional details are available on the cobimetinib fumarate profile page.
When can COTELLIC (cobimetinib fumarate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: cobimetinib fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 05, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,990,219
COTELLIC is a drug marketed by Genentech Inc. There are seven patents protecting this drug.
This drug has two hundred and seven patent family members in forty-six countries.
See drug price trends for COTELLIC.
The generic ingredient in COTELLIC is cobimetinib fumarate. One supplier is listed for this generic product. Additional details are available on the cobimetinib fumarate profile page.
When can ZTALMY (ganaxolone) generic drug versions launch in the Eurasian Patent Organization?
Generic name: ganaxolone
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 28, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 17,290
Patent Title: КОМПОЗИЦИИ НА ОСНОВЕ ГАНАКСОЛОНА (GANAXOLONE-BASED FORMULATIONS)
ZTALMY is a drug marketed by Immedica Pharma. There are eleven patents protecting this drug.
This drug has forty-nine patent family members in sixteen countries. There has been litigation on patents covering ZTALMY
See drug price trends for ZTALMY.
The generic ingredient in ZTALMY is ganaxolone. One supplier is listed for this generic product. Additional details are available on the ganaxolone profile page.
When can ALIQOPA (copanlisib dihydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: copanlisib dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 05, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,900,733
Patent Title: ЗАМЕЩЁННЫЕ ПРОИЗВОДНЫЕ 2,3-ДИГИДРОИМИДАЗО[1,2-c]ХИНАЗОЛИНА, ПОЛЕЗНЫЕ ДЛЯ ЛЕЧЕНИЯ ГИПЕРПРОЛИФЕРАТИВНЫХ НАРУШЕНИЙ И БОЛЕЗНЕЙ, СВЯЗАННЫХ С АНГИОГЕНЕЗОМ
ALIQOPA is a drug marketed by Bayer Healthcare. There are three patents protecting this drug.
This drug has one hundred and five patent family members in forty-eight countries.
See drug price trends for ALIQOPA.
The generic ingredient in ALIQOPA is copanlisib dihydrochloride. Additional details are available on the copanlisib dihydrochloride profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 05, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 17,091
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 05, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,970,532
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can ZYKADIA (ceritinib) generic drug versions launch in the Eurasian Patent Organization?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 17,252
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch in the Eurasian Patent Organization?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 17,405
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch in the Eurasian Patent Organization?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 19,941
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch in the Eurasian Patent Organization?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 19,966
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch in the Eurasian Patent Organization?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,970,557
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch in the Eurasian Patent Organization?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,000,366
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch in the Eurasian Patent Organization?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,100,687
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch in the Eurasian Patent Organization?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,100,688
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can XERMELO (telotristat etiprate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: telotristat etiprate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 12, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 18,150
XERMELO is a drug marketed by Tersera. There are five patents protecting this drug.
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for XERMELO.
The generic ingredient in XERMELO is telotristat etiprate. One supplier is listed for this generic product. Additional details are available on the telotristat etiprate profile page.
When can XERMELO (telotristat etiprate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: telotristat etiprate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 12, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,970,575
XERMELO is a drug marketed by Tersera. There are five patents protecting this drug.
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for XERMELO.
The generic ingredient in XERMELO is telotristat etiprate. One supplier is listed for this generic product. Additional details are available on the telotristat etiprate profile page.
When can PICATO (ingenol mebutate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 18, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 18,545
Patent Title: ТЕРАПЕВТИЧЕСКИЕ КОМПОЗИЦИИ (THERAPEUTIC COMPOSITIONS)

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can PICATO (ingenol mebutate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 18, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 24,152
Patent Title: КОМПОЗИЦИЯ ДЛЯ МЕСТНОГО ВВЕДЕНИЯ ДЛЯ ЛЕЧЕНИЯ ИЛИ ПРОФИЛАКТИКИ РАКА КОЖИ (TOPICAL COMPOSITION FOR TREATING OR PREVENTING SKIN CANCER)

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can PICATO (ingenol mebutate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 18, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,870,063
Patent Title: ТЕРАПЕВТИЧЕСКИЕ КОМПОЗИЦИИ

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can PICATO (ingenol mebutate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 18, 2026
Generic Entry Controlled by: Eurasian Patent Organization Patent 201,201,452
Patent Title: ТЕРАПЕВТИЧЕСКИЕ КОМПОЗИЦИИ

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can EPSOLAY (benzoyl peroxide) generic drug versions launch in the Eurasian Patent Organization?
Generic name: benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 01, 2027
Generic Entry Controlled by: Eurasian Patent Organization Patent 18,275
Patent Title: СПОСОБ ПОЛУЧЕНИЯ ЧАСТИЦ, СОДЕРЖАЩИХ ПОКРЫТИЕ ИЗ ОКСИДА МЕТАЛЛА, И ЧАСТИЦЫ С ПОКРЫТИЕМ ИЗ ОКСИДА МЕТАЛЛА (METHOD FOR PREPARING PARTICLES COMPRISING METAL OXIDE COATING AND PARTICLES WITH METAL OXIDE COATING)
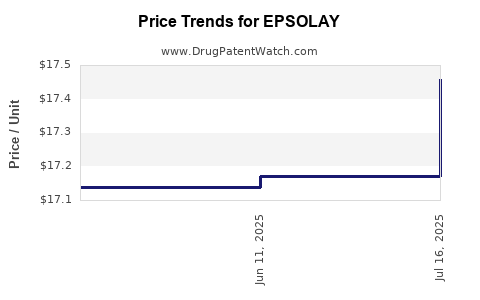
This drug has fifty-one patent family members in fifteen countries. There has been litigation on patents covering EPSOLAY
See drug price trends for EPSOLAY.
The generic ingredient in EPSOLAY is benzoyl peroxide. There are seventeen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the benzoyl peroxide profile page.
When can OSENI (alogliptin benzoate; pioglitazone hydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: alogliptin benzoate; pioglitazone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 01, 2027
Generic Entry Controlled by: Eurasian Patent Organization Patent 15,180

This drug has one hundred and one patent family members in forty-two countries. There has been litigation on patents covering OSENI
See drug price trends for OSENI.
The generic ingredient in OSENI is alogliptin benzoate; pioglitazone hydrochloride. There are ten drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate; pioglitazone hydrochloride profile page.
When can OSENI (alogliptin benzoate; pioglitazone hydrochloride) generic drug versions launch in the Eurasian Patent Organization?
Generic name: alogliptin benzoate; pioglitazone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 01, 2027
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,970,726

This drug has one hundred and one patent family members in forty-two countries. There has been litigation on patents covering OSENI
See drug price trends for OSENI.
The generic ingredient in OSENI is alogliptin benzoate; pioglitazone hydrochloride. There are ten drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate; pioglitazone hydrochloride profile page.
When can XENLETA (lefamulin acetate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: lefamulin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 20, 2027
Generic Entry Controlled by: Eurasian Patent Organization Patent 18,707
XENLETA is a drug marketed by Hong Kong. There are four patents protecting this drug.
This drug has one hundred and twenty patent family members in thirty-six countries. There has been litigation on patents covering XENLETA
See drug price trends for XENLETA.
The generic ingredient in XENLETA is lefamulin acetate. Additional details are available on the lefamulin acetate profile page.
When can XENLETA (lefamulin acetate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: lefamulin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 20, 2027
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,970,868
XENLETA is a drug marketed by Hong Kong. There are four patents protecting this drug.
This drug has one hundred and twenty patent family members in thirty-six countries. There has been litigation on patents covering XENLETA
See drug price trends for XENLETA.
The generic ingredient in XENLETA is lefamulin acetate. Additional details are available on the lefamulin acetate profile page.
When can QVAR REDIHALER (beclomethasone dipropionate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: beclomethasone dipropionate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 11, 2027
Generic Entry Controlled by: Eurasian Patent Organization Patent 16,831
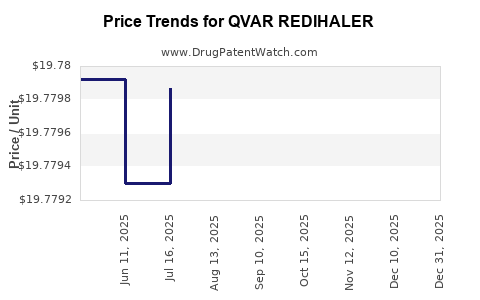
This drug has two hundred and seventy-eight patent family members in twenty-seven countries. There has been litigation on patents covering QVAR REDIHALER
See drug price trends for QVAR REDIHALER.
The generic ingredient in QVAR REDIHALER is beclomethasone dipropionate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the beclomethasone dipropionate profile page.
When can QVAR REDIHALER (beclomethasone dipropionate) generic drug versions launch in the Eurasian Patent Organization?
Generic name: beclomethasone dipropionate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 11, 2027
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,970,907

This drug has two hundred and seventy-eight patent family members in twenty-seven countries. There has been litigation on patents covering QVAR REDIHALER
See drug price trends for QVAR REDIHALER.
The generic ingredient in QVAR REDIHALER is beclomethasone dipropionate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the beclomethasone dipropionate profile page.
When can TRADJENTA (linagliptin) generic drug versions launch in the Eurasian Patent Organization?
Generic name: linagliptin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 30, 2027
Generic Entry Controlled by: Eurasian Patent Organization Patent 29,890
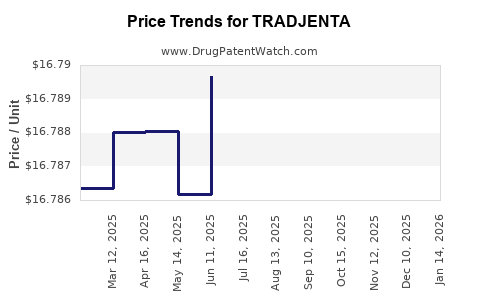
This drug has three hundred and eighty-one patent family members in forty-one countries. There has been litigation on patents covering TRADJENTA
See drug price trends for TRADJENTA.
The generic ingredient in TRADJENTA is linagliptin. There are nineteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the linagliptin profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch in the Eurasian Patent Organization?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 03, 2027
Generic Entry Controlled by: Eurasian Patent Organization Patent 34,294
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch in the Eurasian Patent Organization?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 03, 2027
Generic Entry Controlled by: Eurasian Patent Organization Patent 200,971,018
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.