TYMLOS Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Tymlos, and what generic alternatives are available?
Tymlos is a drug marketed by Radius and is included in one NDA. There are nine patents protecting this drug and one Paragraph IV challenge.
This drug has fifty-six patent family members in twenty-nine countries.
The generic ingredient in TYMLOS is abaloparatide. One supplier is listed for this compound. Additional details are available on the abaloparatide profile page.
DrugPatentWatch® Generic Entry Outlook for Tymlos
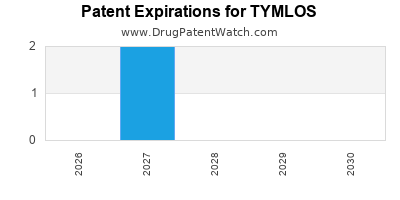
Tymlos was eligible for patent challenges on April 28, 2021.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be October 3, 2027. This may change due to patent challenges or generic licensing.
There have been two patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for TYMLOS?
- What are the global sales for TYMLOS?
- What is Average Wholesale Price for TYMLOS?
Summary for TYMLOS
| International Patents: | 56 |
| US Patents: | 9 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 6 |
| Drug Prices: | Drug price information for TYMLOS |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for TYMLOS |
| What excipients (inactive ingredients) are in TYMLOS? | TYMLOS excipients list |
| DailyMed Link: | TYMLOS at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for TYMLOS
Generic Entry Date for TYMLOS*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
SOLUTION;SUBCUTANEOUS |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for TYMLOS
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| The Affiliated Hospital of Qingdao University | PHASE1 |
| University of Vermont | Phase 2 |
| Crozer-Keystone Health System | Phase 4 |
Pharmacology for TYMLOS
Paragraph IV (Patent) Challenges for TYMLOS
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| TYMLOS | Subcutaneous Injection | abaloparatide | 3.12 mg/1.56 mL | 208743 | 1 | 2022-06-21 |
US Patents and Regulatory Information for TYMLOS
TYMLOS is protected by twelve US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of TYMLOS is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Radius | TYMLOS | abaloparatide | SOLUTION;SUBCUTANEOUS | 208743-001 | Apr 28, 2017 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Radius | TYMLOS | abaloparatide | SOLUTION;SUBCUTANEOUS | 208743-001 | Apr 28, 2017 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Radius | TYMLOS | abaloparatide | SOLUTION;SUBCUTANEOUS | 208743-001 | Apr 28, 2017 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Radius | TYMLOS | abaloparatide | SOLUTION;SUBCUTANEOUS | 208743-001 | Apr 28, 2017 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Radius | TYMLOS | abaloparatide | SOLUTION;SUBCUTANEOUS | 208743-001 | Apr 28, 2017 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Radius | TYMLOS | abaloparatide | SOLUTION;SUBCUTANEOUS | 208743-001 | Apr 28, 2017 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Radius | TYMLOS | abaloparatide | SOLUTION;SUBCUTANEOUS | 208743-001 | Apr 28, 2017 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for TYMLOS
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Radius Health Ireland Ltd | Eladynos | abaloparatide | EMEA/H/C/004157Treatment of osteoporosis. | Refused | no | no | no | 2019-01-07 | |
| Theramex Ireland Limited | Eladynos | abaloparatide | EMEA/H/C/005928Treatment of osteoporosis in postmenopausal women at increased risk of fracture. | Authorised | no | no | no | 2022-12-12 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for TYMLOS
When does loss-of-exclusivity occur for TYMLOS?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 07322334
Patent: Method of drug delivery for bone anabolic protein
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0719821
Patent: composiÇço estÁvel na armazenagem apropriada para administraÇço a pacientes e mÉtodo de tratamento de osteoporose nestes
Estimated Expiration: ⤷ Start Trial
Patent: 0722428
Patent: USO DE COMPOSIÇÃO ESTÁVEL NO ARMAZENAMENTO
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 64734
Patent: PROCEDE D'ADMINISTRATION PHARMACOLOGIQUE D'UNE PROTEINE ANABOLIQUE OSSEUSE (METHOD OF DRUG DELIVERY FOR BONE ANABOLIC PROTEIN)
Estimated Expiration: ⤷ Start Trial
China
Patent: 1578093
Patent: Method of drug delivery for bone anabolic protein
Estimated Expiration: ⤷ Start Trial
Patent: 2274492
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0171217
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 19198
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 73789
Estimated Expiration: ⤷ Start Trial
Patent: 57278
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 73789
Patent: COMPOSITION STABLE D'UNE PROTÉINE ANABOLIQUE OSSEUSE QUI EST UN ANALOGUE DE PTHRP, ET SES EMPLOIS (A STABLE COMPOSITION COMPRISING A BONE ANABOLIC PROTEIN, NAMELY A PTHRP ANALOGUE, AND USES THEREOF)
Estimated Expiration: ⤷ Start Trial
Patent: 57278
Patent: COMPOSITIONS STABLES CONTENANT PTHRP ET LEUR EMPLOI (A STABLE COMPOSITION COMPRISING PTHRP AND USES THEREOF)
Estimated Expiration: ⤷ Start Trial
France
Patent: C1024
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 14181
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 7926
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 75611
Estimated Expiration: ⤷ Start Trial
Patent: 10505835
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 57278
Estimated Expiration: ⤷ Start Trial
Luxembourg
Patent: 0309
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 09003569
Patent: METODO DE ADMINISTRACION DE FARMACOS PARA LA PROTEINA ANABOLICA OSEA. (METHOD OF DRUG DELIVERY FOR BONE ANABOLIC PROTEIN.)
Estimated Expiration: ⤷ Start Trial
Netherlands
Patent: 1235
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 6682
Patent: METHOD OF DRUG DELIVERY FOR BONE ANABOLIC PROTEIN PARATHYROID HORMONE-RELATED PROTEIN (PTHrP)
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 4885
Estimated Expiration: ⤷ Start Trial
Patent: 091545
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 57278
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 73789
Estimated Expiration: ⤷ Start Trial
Patent: 57278
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 06070
Patent: METHOD OF TREATING OSTEOPOROSIS AND COMPOSITION USED THEREIN
Estimated Expiration: ⤷ Start Trial
Patent: 09116531
Patent: СПОСОБ ЛЕЧЕНИЯ ОСТЕОПОРОЗА И ИСПОЛЬЗУЕМАЯ В НЕМ КОМПОЗИЦИЯ (METHOD OF TREATING OSTEOPOROSIS AND COMPOSITION USED THEREIN)
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 164
Patent: STABILNA KOMPOZICIJA KOJA SADRŽI PTHRP I NJENE UPOTREBE (A STABLE COMPOSITION COMPRISING PTHRP AND USES THEREOF)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 5580
Patent: METHOD OF DRUG DELIVERY FOR BONE ANABOLIC PROTEIN
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 57278
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1512377
Estimated Expiration: ⤷ Start Trial
Patent: 090083350
Patent: METHOD OF DRUG DELIVERY FOR BONE ANABOLIC PROTEIN
Estimated Expiration: ⤷ Start Trial
Patent: 150020289
Patent: METHOD OF DRUG DELIVERY FOR BONE ANABOLIC PROTEIN
Estimated Expiration: ⤷ Start Trial
Patent: 170067906
Patent: 골 아나볼릭 단백질을 위한 약물 전달 방법 (METHOD OF DRUG DELIVERY FOR BONE ANABOLIC PROTEIN)
Estimated Expiration: ⤷ Start Trial
Patent: 180117738
Patent: 골 아나볼릭 단백질을 위한 약물 전달 방법 (METHOD OF DRUG DELIVERY FOR BONE ANABOLIC PROTEIN)
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 37283
Estimated Expiration: ⤷ Start Trial
Patent: 39459
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 776
Patent: СПОСОБ ДОСТАВКИ лекарственного ПРЕПАРАТА ДЛЯ УСИЛЕНИЯ СИНТЕЗА БЕЛКА В КОСТЯХ (METHOD OF DRUG DELIVERY FOR BONE ANABOLIC PROTEIN)
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering TYMLOS around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| South Korea | 20210120009 | ⤷ Start Trial | |
| Israel | 197926 | ⤷ Start Trial | |
| Norway | 344885 | ⤷ Start Trial | |
| China | 101578093 | ⤷ Start Trial | |
| Canada | 3122231 | PROCEDES DE DETECTION D'ANTICORPS DE NEUTRALISATION DIRIGES CONTRE L'HORMONE PARATHYROIDE (PTH) ET ANALOGUE DE PEPTIDE LIE A L'HORMONE PARATHYROIDE (PTHRP) (METHODS FOR DETECTING NEUTRALIZING ANTIBODIES TO PARATHYROID HORMONE (PTH) AND PARATHYROID HORMONE-RELATED PEPTIDE (PTHRP) ANALOG) | ⤷ Start Trial |
| Israel | 197926 | תכשיר יציב המכיל חלבון עצם אנבולי, דהיינו אנלוג ל–pthrp, ושימושיו (Stable composition comprising a bone anabolic protein, namely a pthrp analogue, and uses thereof) | ⤷ Start Trial |
| South Korea | 101512377 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for TYMLOS
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2073789 | 2023C/523 | Belgium | ⤷ Start Trial | DETAILS ASSIGNMENT: CHANGE OF OWNER(S), OTHER |
| 2073789 | 2390018-6 | Sweden | ⤷ Start Trial | PRODUCT NAME: ABALOPARATIDE; REG. NO/DATE: EU/1/22/1706 20221213 |
| 2073789 | CA 2023 00019 | Denmark | ⤷ Start Trial | PRODUCT NAME: ABALOPARATID; REG. NO/DATE: EU/1/22/1706 20221213 |
| 2073789 | C202330026 | Spain | ⤷ Start Trial | PRODUCT NAME: ABALOPARATIDA; NATIONAL AUTHORISATION NUMBER: EU/1/22/1706; DATE OF AUTHORISATION: 20221212; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/22/1706; DATE OF FIRST AUTHORISATION IN EEA: 20221212 |
| 2073789 | 23C1024 | France | ⤷ Start Trial | PRODUCT NAME: ABALOPARATIDE; REGISTRATION NO/DATE: EU/1/22/1706 20221213 |
| 2073789 | LUC00309 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: ABALOPARATIDE; AUTHORISATION NUMBER AND DATE: EU/1/22/1706 20221213 |
| 2073789 | 301235 | Netherlands | ⤷ Start Trial | PRODUCT NAME: ABALOPARATIDE; REGISTRATION NO/DATE: EU/1/22/1706 20221213 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
TYMLOS (Abaloparatide) Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.