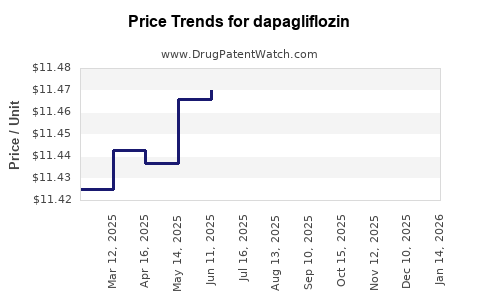
Last updated: March 4, 2026
What is the Current Market Position of Dapagliflozin?
Dapagliflozin, marketed by AstraZeneca under the brand name Farxiga (United States) and Forxiga (Europe and other regions), is an SGLT2 inhibitor approved for type 2 diabetes (T2D), heart failure, and chronic kidney disease (CKD). Its revenue generated globally in 2022 was approximately $4.2 billion, accounting for roughly 57% of AstraZeneca's diabetes portfolio[1].
How Does Dapagliflozin Compare to Competitors?
In the SGLT2 inhibitor class, dapagliflozin faces competition from drugs like:
- Empagliflozin (Jardiance): Dominates the heart failure segment with $8.5 billion in 2022 sales[2].
- Canagliflozin (Invokana): Focuses on T2D and CKD, generating ~$2.8 billion in 2022[3].
Dapagliflozin's market share in the SGLT2 class in 2022 stood at approximately 36%, trailing Empagliflozin's 45%. Its broader approval for heart failure and CKD expands its market beyond glucose control.
What Are the Key Market Drivers?
-
Expanding Indications: Approval for heart failure with reduced ejection fraction (HFrEF) and CKD broadens dapagliflozin's use cases[4]. The US FDA approved it for HFrEF in 2020, and the European Medicines Agency (EMA) follow in 2021.
-
Clinical Evidence: Robust trials demonstrate dapagliflozin reduces hospitalization and mortality in heart failure and CKD, encouraging adoption[5].
-
Competitive Pricing: The drug's pricing advantage over some rivals enables broader access across healthcare systems, especially in emerging markets.
-
Growing Prevalence of Diabetes and Heart Disease: The global increase in T2D (expected to reach 700 million cases by 2045[6]) and CKD fuels demand.
What Are the Key Challenges and Risks?
-
Patent Expirations: Dapagliflozin's patents are set to expire in key markets between 2030 and 2034, risking generic entry[7].
-
Pricing Pressures: Reimbursement agencies seek discounts, especially in cost-constrained healthcare systems.
-
Regulatory Hurdles for New Indications: Additional approvals require substantial clinical evidence, with risks of rejection or delays.
-
Market Saturation: The high adoption rates amid competitive class limits upside potential in mature markets.
What Is the Financial Forecast?
AstraZeneca projects dapagliflozin's revenue to grow at a compound annual growth rate (CAGR) of approximately 10-12% over the next five years, driven by expanded indications and geographic penetration. The expected pipeline and label expansions may result in revenues reaching roughly $7 billion worldwide by 2027[8].
How Do Policy and Pricing Trends Affect Financials?
Pricing negotiations, especially with payers like Medicare in the US, could suppress revenue growth. The trend toward value-based agreements and cost-effectiveness assessments influence reimbursement levels. This may cap gross margins but could increase market access.
What Are the Strategic Growth Opportunities?
-
Further Indication Approvals: Pending data on benefits in heart failure with preserved ejection fraction (HFpEF) and diabetic nephropathy could unlock new revenue streams.
-
Geographical Expansion: Focused entry into emerging markets like China and India could boost sales, given rising disease prevalence and unmet needs.
-
Combination Therapies: Developing fixed-dose combinations with other antidiabetic or cardiovascular drugs might improve adherence and market penetration.
What Is the Impact of Patent Expirations and Generic Competition?
Patent expiry in the US is projected for 2034. Once generics enter, initial discounts could be 80% or more, squeezing revenue margins. AstraZeneca's strategic focus on lifecycle management, including label extensions, aims to mitigate revenue erosion[9].
Summary
Dapagliflozin maintains strong market momentum supported by expanded indications, competitive positioning, and a growing global patient base. However, patent expirations, pricing dynamics, and intense competition pose risks to long-term growth. The company's pipeline and geographic expansion will be key to sustaining financial performance.
Key Takeaways
- Dapagliflozin's 2022 revenue totaled $4.2 billion, with a 36% market share in SGLT2 inhibitors.
- Expanding approvals for heart failure and CKD provide growth catalysts.
- Patent expirations between 2030-2034 threaten future revenues.
- Regulatory, pricing, and competitive pressures could limit upside.
- Targeted geographic expansion and pipeline development are strategic priorities.
FAQs
1. When are Dapagliflozin patents set to expire?
The primary patents in the US and Europe are scheduled to expire between 2030 and 2034, with some secondary patents extending protections.
2. How does Dapagliflozin's efficacy compare to its competitors?
Clinical trials demonstrate similar glucose-lowering effects among SGLT2 inhibitors, but dapagliflozin is uniquely approved for heart failure and CKD, differentiating its use cases.
3. What are the main regulatory hurdles for further indication approvals?
Additional clinical trial data demonstrating safety and efficacy in new indications like HFpEF are required, which involves significant time and investment.
4. How is AstraZeneca addressing patent expirations?
The company invests in pipeline expansion, developing combination therapies, and seeking new indications to retain market share post-patent expiry.
5. What is the outlook for generic versions?
Generic dapagliflozin could enter the market around 2034, potentially reducing revenue by up to 80%, unless brand-specific differentiators like label extensions are successful.
References
[1] AstraZeneca. (2023). Annual Report 2022.
[2] IQVIA. (2023). Pharmaceutical Market Data.
[3] Evaluate Pharma. (2023). World Preview 2023.
[4] FDA. (2020). Approval Summary: Dapagliflozin for Heart Failure.
[5] Mahaffey, K. W., et al. (2021). N Engl J Med, 385, 903–915.
[6] International Diabetes Federation. (2021). IDF Diabetes Atlas, 9th Edition.
[7] Patent Data. (2023). Global Patent Expiry Calendar.
[8] AstraZeneca. (2023). Pipeline and Strategy Overview.
[9] FDA. (2022). Patent and Exclusivity Data.