Glaxosmithkline Company Profile
✉ Email this page to a colleague
What is the competitive landscape for GLAXOSMITHKLINE
GLAXOSMITHKLINE has one hundred and fifty-two approved drugs.
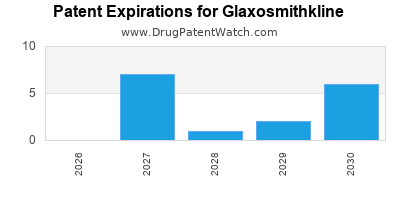
There are thirty-seven US patents protecting GLAXOSMITHKLINE drugs.
There are eight hundred and five patent family members on GLAXOSMITHKLINE drugs in sixty-two countries and one hundred and eighty-one supplementary protection certificates in twenty-one countries.
Summary for Glaxosmithkline
| International Patents: | 805 |
| US Patents: | 37 |
| Tradenames: | 107 |
| Ingredients: | 93 |
| NDAs: | 152 |
| Drug Master File Entries: | 8 |
| Patent Litigation for Glaxosmithkline: | See patent lawsuits for Glaxosmithkline |
| PTAB Cases with Glaxosmithkline as petitioner: | See PTAB cases with Glaxosmithkline as petitioner |
Drugs and US Patents for Glaxosmithkline
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glaxosmithkline | ANTEPAR | piperazine citrate | SYRUP;ORAL | 009102-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Glaxosmithkline | CEFTIN | cefuroxime axetil | FOR SUSPENSION;ORAL | 050672-001 | Jun 30, 1994 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Glaxosmithkline Llc | LAMICTAL XR | lamotrigine | TABLET, EXTENDED RELEASE;ORAL | 022115-005 | Apr 14, 2010 | AB | RX | Yes | No | 8,637,512 | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Glaxosmithkline Llc | LAMICTAL ODT | lamotrigine | TABLET, ORALLY DISINTEGRATING;ORAL | 022251-001 | May 8, 2009 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ||||
| Glaxosmithkline Llc | REQUIP | ropinirole hydrochloride | TABLET;ORAL | 020658-002 | Sep 19, 1997 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Glaxosmithkline | CLOXAPEN | cloxacillin sodium | CAPSULE;ORAL | 062233-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Glaxosmithkline
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Glaxosmithkline | RELENZA | zanamivir | POWDER;INHALATION | 021036-001 | Jul 26, 1999 | 5,648,379 | ⤷ Start Trial |
| Glaxosmithkline | BECONASE | beclomethasone dipropionate | AEROSOL, METERED;NASAL | 018584-001 | Approved Prior to Jan 1, 1982 | 4,414,209 | ⤷ Start Trial |
| Glaxosmithkline | WELLBUTRIN | bupropion hydrochloride | TABLET;ORAL | 018644-002 | Dec 30, 1985 | 4,347,257 | ⤷ Start Trial |
| Glaxosmithkline | TRANDATE HCT | hydrochlorothiazide; labetalol hydrochloride | TABLET;ORAL | 019174-002 | Apr 10, 1987 | 4,066,755 | ⤷ Start Trial |
| Glaxosmithkline Llc | LAMICTAL XR | lamotrigine | TABLET, EXTENDED RELEASE;ORAL | 022115-005 | Apr 14, 2010 | 9,144,547 | ⤷ Start Trial |
| Glaxosmithkline | ZYBAN | bupropion hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 020711-003 | May 14, 1997 | 5,731,000 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for GLAXOSMITHKLINE drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Extended-release Tablets | 3 mg | ➤ Subscribe | 2009-01-08 |
| ➤ Subscribe | Injection | 6 mg/0.5 mL, 0.5 mL vials | ➤ Subscribe | 2004-10-25 |
| ➤ Subscribe | Tablets | 0.25 mg, 0.5 mg, 1 mg and 2 mg | ➤ Subscribe | 2004-12-22 |
| ➤ Subscribe | Tablets | 100 mg | ➤ Subscribe | 2007-10-31 |
| ➤ Subscribe | Tablets | 62.5 mg/25 mg | ➤ Subscribe | 2010-09-14 |
| ➤ Subscribe | Extended-release Capsules | 225 mg and 425 mg | ➤ Subscribe | 2006-10-11 |
| ➤ Subscribe | Extended-release Tablets | 25 mg, 50 mg, 100 mg, 200 mg, 250 mg, and 300 mg | ➤ Subscribe | 2014-02-12 |
| ➤ Subscribe | Extended-release Tablets | 8 mg | ➤ Subscribe | 2008-11-03 |
| ➤ Subscribe | Extended-release Tablets | 12 mg | ➤ Subscribe | 2009-02-05 |
| ➤ Subscribe | Extended-release Tablets | 6 mg | ➤ Subscribe | 2009-07-22 |
| ➤ Subscribe | Tablets | 150 mg | ➤ Subscribe | 2007-10-30 |
| ➤ Subscribe | Orally Disintegrating Tablets | 25 mg, 50 mg, 100 mg, and 200 mg | ➤ Subscribe | 2009-12-21 |
| ➤ Subscribe | Injection | 6 mg/0.5 mL, 0.5 mL (prefilled syringes) | ➤ Subscribe | 2006-05-09 |
| ➤ Subscribe | Tablets | 3 mg, 4 mg and 5 mg | ➤ Subscribe | 2005-02-04 |
| ➤ Subscribe | Tablets | 250 mg/100 mg | ➤ Subscribe | 2009-04-03 |
| ➤ Subscribe | Extended-release Capsules | 325 mg | ➤ Subscribe | 2006-11-07 |
| ➤ Subscribe | Oral Suspension | 750 mg/5 mL | ➤ Subscribe | 2009-10-20 |
| ➤ Subscribe | Extended-release Tablets | 4 mg | ➤ Subscribe | 2008-10-31 |
| ➤ Subscribe | Extended-release Tablets | 3 mg | ➤ Subscribe | 2009-01-08 |
| ➤ Subscribe | Extended-release Tablets | 2 mg | ➤ Subscribe | 2008-10-14 |
International Patents for Glaxosmithkline Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Portugal | 1633724 | ⤷ Start Trial |
| Mexico | PA06012405 | ⤷ Start Trial |
| European Patent Office | 1960021 | ⤷ Start Trial |
| Malaysia | 147789 | ⤷ Start Trial |
| Brazil | PI0412899 | ⤷ Start Trial |
| Japan | 2005533583 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Glaxosmithkline Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0933372 | 13/2008 | Austria | ⤷ Start Trial | PRODUCT NAME: FOSAMPRENAVIR ALS FOSAMPRENAVIR-CALCIUM; REGISTRATION NO/DATE: EU/1/04/282/001 - EU/1/04/282/002 (MITTEILUNG VOM 14.07.2004) 20040713 |
| 0809498 | 10C0038 | France | ⤷ Start Trial | PRODUCT NAME: ACYCLOVIR ET HYDROCORTISONE; NAT. REGISTRATION NO/DATE: NL 36 826 20100420; FIRST REGISTRATION: SK - 2108/08467-R 20091026 |
| 2240466 | C201830027 | Spain | ⤷ Start Trial | PRODUCT NAME: NIRAPARIB; NATIONAL AUTHORISATION NUMBER: EU/1/17/1235; DATE OF AUTHORISATION: 20171116; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/17/1235; DATE OF FIRST AUTHORISATION IN EEA: 20171116 |
| 0208846 | 99C0032 | Belgium | ⤷ Start Trial | PRODUCT NAME: ZANAMIVIR; NAT. REGISTRATION NO/DATE: NL 24 551 19990726; FIRST REGISTRATION: SE - 14 997 19990209 |
| 0526543 | SPC/GB99/030 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: ZANAMIVIR, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT OR DERIVATIVE; REGISTERED: SE 14997 19990209; UK PL10949/0327 19990625 |
| 2109608 | 2018/019 | Ireland | ⤷ Start Trial | PRODUCT NAME: NIRAPARIB OR A PHARMACEUTICALLY ACCEPTABLE SALT, STEREOISOMER OR TAUTOMER THEREOF, PARTICULARLY THE TOSYLATE OR A HYDRATE, ESPECIALLY THE TOSYLATE MONOHYDRATE.; REGISTRATION NO/DATE: EU/1/17/1235 20171116 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.