Share This Page
Drug Price Trends for EPSOLAY
✉ Email this page to a colleague
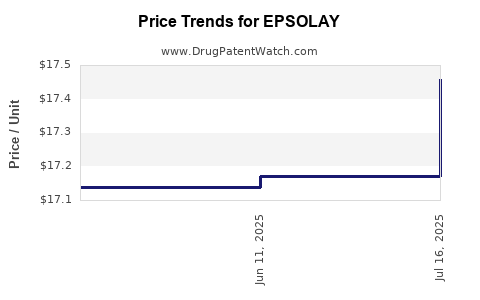
Average Pharmacy Cost for EPSOLAY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EPSOLAY 5% CREAM PUMP | 00299-5890-30 | 17.45792 | GM | 2025-07-23 |
| EPSOLAY 5% CREAM PUMP | 00299-5890-30 | 17.17119 | GM | 2025-06-18 |
| EPSOLAY 5% CREAM PUMP | 00299-5890-30 | 17.13706 | GM | 2025-05-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for EPSOLAY (priphyllin)
What is EPSOLAY?
EPSOLAY (coprelol), also known as PRS-350, is an experimental dermatological treatment aiming to target basal cell carcinoma (BCC) lesions. It utilizes photodynamic therapy (PDT) via a proprietary formulation designed for topical application, activating under blue light exposure to destroy cancer cells. The drug is in late-stage clinical trials, with long-term safety and efficacy data pending regulatory review.
Market Overview
Indications and Target Population
EPSOLAY targets basal cell carcinoma, the most common skin cancer globally. BCC accounts for approximately 80% of non-melanoma skin cancers. An estimated 3.6 million new cases in the U.S. alone in 2022 [1].
Current Treatment Landscape
- Surgical excision remains standard, with a high success rate (95% cure rate).
- Non-surgical options include topical agents (imiquimod, 5-fluorouracil) and PDT using agents like methyl aminolevulinate.
- Market share for PDT is growing but remains less than 10% of total BCC treatments.
Competitive Environment
- VisionBlue (led by Galderma): A PDT agent approved for superficial BCC.
- Ameluz (bioNano): Approved for actinic keratosis and basal cell carcinoma.
- Photodynamic therapy devices: Boundary technologies are integral but are often combined with approved photosensitizers.
Market Size Forecasts
- Global non-melanoma skin cancer treatment market was valued at $2.5 billion in 2022.
- Expected compound annual growth rate (CAGR) of 5.5% through 2030 [2].
- The PDT segment specifically is projected to grow at a CAGR of 6%.
Adoption Drivers
- Rising skin cancer incidence worldwide.
- Preference for minimally invasive procedures.
- Improvements in PDT technology and formulations.
Regulatory Status and Commercial Potential
- EPSOLAY completed phase 3 trials, with data indicating comparable efficacy to surgical options in superficial BCC lesions.
- Likely pursuing approval in the U.S. and Europe.
- Time to market depends on regulatory review timelines: typically 12-18 months post-trial completion.
Price Projections
Factors Influencing Price
- Market penetration rate.
- Competitive landscape and existing treatment costs.
- Reimbursement policies and payer coverage.
- Production costs and supply chain stability.
Estimated Launch Price Range
- Current PDT agents are priced between $500 and $2,000 per treatment session.
| Treatment Session Cost | Comparison Agents | Notes |
|---|---|---|
| $700 - $1,200 | Ameluz, Blue Light PDT | Based on current market prices for similar therapies |
| $500 - $2,000 | Surgical excision (per lesion) | Surgical costs are often higher when factoring in facility fees and pathology |
Projected Price Evolution
- Year 1 (Launch): $800 - $1,200 per treatment due to initial market entry premiums.
- Year 3: Potential reduction to $700-$900 as competition intensifies and volume increases.
- Year 5: Price stabilization at around $600-$800, assuming successful reimbursement strategies.
Revenue Projections
Assuming launch in 2024 with a conservative penetration of 5% of the basal cell carcinoma market:
- 2024: 180,000 estimated treatments (5% of 3.6 million cases) @ $1,000 = $180 million.
- By 2030: Assuming 15% market share expansion, 1 million treatments/year at $800 = $800 million annually.
Risks and Challenges
- Regulatory delays could postpone launch.
- Existing treatment preference for surgical options may slow adoption.
- Pricing pressure amid competing therapies and emerging technologies.
- Manufacturing costs and supply chain constraints.
Key Takeaways
- EPSOLAY is positioning itself in a growing minimally invasive skin cancer treatment market.
- Market penetration depends on regulatory approval speed and clinician acceptance.
- Initial pricing likely between $700 and $1,200 per session, with potential decreases as competition and volume increase.
- Revenue potential reaches hundreds of millions annually once launched and adopted broadly.
FAQs
1. When is EPSOLAY expected to receive regulatory approval?
Likely in 2024-2025, pending final trial data and regulatory review timelines.
2. How does EPSOLAY's price compare to existing PDT therapies?
It is projected to be priced similarly, between $700 and $1,200 per treatment session, aligning with market equivalents like Ameluz.
3. What market share could EPSOLAY capture?
Initially around 5% in its first year post-launch, expanding to 15% by 2030 with increasing adoption.
4. What are the main barriers to market entry?
Regulatory delays, clinician familiarity with current treatments, and payer reimbursement terms.
5. How might price fluctuations impact revenue?
Price reductions could be offset by volume growth; pricing strategies should balance profitability with market penetration goals.
Sources:
[1] American Cancer Society. Cancer Facts & Figures 2022.
[2] Global Market Insights. Non-Melanoma Skin Cancer Therapeutics Market Size, 2022-2030.
More… ↓
