Expiring Drug Patents Cheat Sheet
We analyse the patents covering drugs in 134 countries and quickly give you the likely loss-of-exclusivity/generic entry date
South Africa: These 28 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "South Africa: These 28 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027" DrugPatentWatch.com thinkBiotech, 2026 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 12, 2026
Generic Entry Controlled by: South Africa Patent 200,809,094
Patent Title: Polymorphs of N-hydroxy-3-[4[[[2-(2-methyl-1H-indol-3-yl)ethyl]amino]methyl]pheny]-2E-2-propenamide
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: South Africa Patent 200,810,501
Patent Title: PHARMACEUTICAL COMPOSITIONS
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can JESDUVROQ (daprodustat) generic drug versions launch?
Generic name: daprodustat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: South Africa Patent 200,810,602
Patent Title: Prolyl hydroxylase inhibitors
JESDUVROQ is a drug marketed by Glaxosmithkline. There are six patents protecting this drug.
This drug has forty-four patent family members in thirty-one countries.
See drug price trends for JESDUVROQ.
The generic ingredient in JESDUVROQ is daprodustat. One supplier is listed for this generic product. Additional details are available on the daprodustat profile page.
When can EXFORGE HCT (amlodipine besylate; hydrochlorothiazide; valsartan) generic drug versions launch?
Generic name: amlodipine besylate; hydrochlorothiazide; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 27, 2026
Generic Entry Controlled by: South Africa Patent 200,810,053

This drug has forty-four patent family members in thirty-one countries. There has been litigation on patents covering EXFORGE HCT
See drug price trends for EXFORGE HCT.
The generic ingredient in EXFORGE HCT is amlodipine besylate; hydrochlorothiazide; valsartan. There are fifty drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the amlodipine besylate; hydrochlorothiazide; valsartan profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: South Africa Patent 200,810,475
Patent Title: CRYSTALLINE SOLVATES AND COMPLEXES OF (IS)-1,5-ANHYDRO-L-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: South Africa Patent 200,810,475
Patent Title: CRYSTALLINE SOLVATES AND COMPLEXES OF (IS)-1,5-ANHYDRO-L-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: South Africa Patent 200,810,475
Patent Title: CRYSTALLINE SOLVATES AND COMPLEXES OF (IS)-1,5-ANHYDRO-L-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: South Africa Patent 200,905,882
Patent Title: MODULATOR OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: South Africa Patent 200,900,962
Patent Title: HEPATITIS C VIRUS INHIBITORS

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 21, 2026
Generic Entry Controlled by: South Africa Patent 200,900,991
Patent Title: COMPOSITIONS,SUITABLE FOR ORAL ADMINISTRATION,COMPRISING A TRIAZOLO [4,5-D]PYRIMIDIN DERIVATE
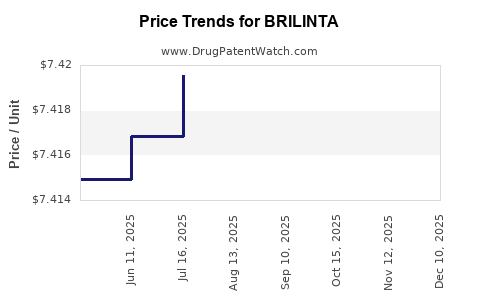
This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 29, 2026
Generic Entry Controlled by: South Africa Patent 200,902,164
Patent Title: Therapeutic compositions comprising a specific endothelin receptor antagonist and a PDE5 inhibitor
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the macitentan profile page.
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 22, 2026
Generic Entry Controlled by: South Africa Patent 200,802,237
Patent Title: Capsule formulation of pirfenidone and pharmaceutically acceptable excipients
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can INREBIC (fedratinib hydrochloride) generic drug versions launch?
Generic name: fedratinib hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: South Africa Patent 200,804,083
Patent Title: Bi-aryl meta-pyrimidine inhibitors of kinases
INREBIC is a drug marketed by Bristol-myers. There are five patents protecting this drug.
This drug has one hundred and nineteen patent family members in forty-two countries. There has been litigation on patents covering INREBIC
See drug price trends for INREBIC.
The generic ingredient in INREBIC is fedratinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the fedratinib hydrochloride profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 10, 2026
Generic Entry Controlled by: South Africa Patent 200,903,053
Patent Title: SYNTHETIC PEPTIDE AMIDES AND DIMERS THEREOF
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 05, 2026
Generic Entry Controlled by: South Africa Patent 200,903,907
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: South Africa Patent 200,903,601
Patent Title: COMPOUNDS AND COMPOSITIONS AS PROTEIN KINASE INHIBITORS
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can XERMELO (telotristat etiprate) generic drug versions launch?
Generic name: telotristat etiprate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 12, 2026
Generic Entry Controlled by: South Africa Patent 200,903,367
Patent Title: 4-Phenyl-6-(2,2,2-trifluoro-1-phenylethoxy) pyrimidine-based compounds and methods of their use
XERMELO is a drug marketed by Tersera. There are five patents protecting this drug.
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for XERMELO.
The generic ingredient in XERMELO is telotristat etiprate. One supplier is listed for this generic product. Additional details are available on the telotristat etiprate profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: South Africa Patent 200,905,630
Patent Title: STABLE DIGESTIVE ENZYME COMPOSITIONS
PANCREAZE is a drug marketed by
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: South Africa Patent 200,905,630
Patent Title: STABLE DIGESTIVE ENZYME COMPOSITIONS
ZENPEP is a drug marketed by
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can XENLETA (lefamulin acetate) generic drug versions launch?
Generic name: lefamulin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 20, 2027
Generic Entry Controlled by: South Africa Patent 200,905,590
Patent Title: Pleuromutilin derivatives for the treatment of diseases mediated by microbes
XENLETA is a drug marketed by Hong Kong. There are four patents protecting this drug.
This drug has one hundred and twenty patent family members in thirty-six countries. There has been litigation on patents covering XENLETA
See drug price trends for XENLETA.
The generic ingredient in XENLETA is lefamulin acetate. Additional details are available on the lefamulin acetate profile page.
When can INLYTA (axitinib) generic drug versions launch?
Generic name: axitinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 05, 2027
Generic Entry Controlled by: South Africa Patent 200,906,990
Patent Title: Crystalline forms of 6-[2-(methylcarbamoyl)phenylsulfanyl]-3-E-[2-(pyridin-2-yl) ethenyl] indazole suitable for the treatment of abnormal cell growth in mammals
INLYTA is a drug marketed by Pf Prism Cv. There are four patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has ninety-seven patent family members in thirty countries. There has been litigation on patents covering INLYTA
See drug price trends for INLYTA.
The generic ingredient in INLYTA is axitinib. There are four drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the axitinib profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 03, 2027
Generic Entry Controlled by: South Africa Patent 200,907,710
Patent Title: NOVEL PHARMACEUICAL COMPOSITION
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can TRELSTAR (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 06, 2027
Generic Entry Controlled by: South Africa Patent 200,907,940
Patent Title: Slow release pharmaceutical composition made of microparticles
TRELSTAR is a drug marketed by Verity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
See drug price trends for TRELSTAR.
The generic ingredient in TRELSTAR is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can TRIPTODUR KIT (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 06, 2027
Generic Entry Controlled by: South Africa Patent 200,907,940
Patent Title: Slow release pharmaceutical composition made of microparticles
TRIPTODUR KIT is a drug marketed by Azurity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
The generic ingredient in TRIPTODUR KIT is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can JAKAFI (ruxolitinib phosphate) generic drug versions launch?
Generic name: ruxolitinib phosphate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 13, 2027
Generic Entry Controlled by: South Africa Patent 200,908,826
Patent Title: SALTS OF THE JANUS KINASE INHIBITOR (R)-3-(4-(7H-PYRROLO[2,3-D]PYRIMIDIN-4-YL)-1H-PYRAZOL-1-YL)-3-CYCLOPENTHYLPROPANEITRILE
JAKAFI is a drug marketed by Incyte Corp. There are eight patents protecting this drug and one Paragraph IV challenge.
This drug has two hundred and thirty-six patent family members in forty-six countries. There has been litigation on patents covering JAKAFI
See drug price trends for JAKAFI.
The generic ingredient in JAKAFI is ruxolitinib phosphate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ruxolitinib phosphate profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 15, 2027
Generic Entry Controlled by: South Africa Patent 200,810,017
Patent Title: 1- [2- (2, 4-Dimethylphenylsulfanyl) -phenyl] piperazine as a compound with combined serotonin Reuptake, 5-HT3 and 5-HT1A activity for the treatment of cognitive impairment

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2027
Generic Entry Controlled by: South Africa Patent 200,907,928
Patent Title: SYNETHOC BILE ACID COMPOSITION, METHOD, AND PREPARATION
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can OJEMDA (tovorafenib) generic drug versions launch?
Generic name: tovorafenib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 29, 2027
Generic Entry Controlled by: South Africa Patent 200,909,223
OJEMDA is a drug marketed by Day One Biopharms. There are two patents protecting this drug.
This drug has fifty patent family members in twenty-seven countries.
The generic ingredient in OJEMDA is tovorafenib. One supplier is listed for this generic product. Additional details are available on the tovorafenib profile page.
South Africa Branded and Generic Drug Markets Assessment and Regulatory Opportunities and Challenges
More… ↓
DrugPatentWatch cited by CNN, NEJM, Nature Journals, and more …

Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
