SIRTURO Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Sirturo, and what generic alternatives are available?
Sirturo is a drug marketed by Janssen Therap and is included in one NDA. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this compound. One supplier is listed for this compound. Additional details are available on the bedaquiline fumarate profile page.
DrugPatentWatch® Generic Entry Outlook for Sirturo
Sirturo was eligible for patent challenges on December 28, 2016.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be March 19, 2029. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for SIRTURO?
- What are the global sales for SIRTURO?
- What is Average Wholesale Price for SIRTURO?
Summary for SIRTURO
| International Patents: | 97 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 54 |
| Clinical Trials: | 16 |
| Patent Applications: | 156 |
| Drug Prices: | Drug price information for SIRTURO |
| What excipients (inactive ingredients) are in SIRTURO? | SIRTURO excipients list |
| DailyMed Link: | SIRTURO at DailyMed |
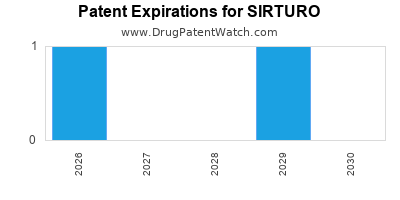

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for SIRTURO
Generic Entry Date for SIRTURO*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for SIRTURO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Tuberculosis Trials Consortium | Phase 2/Phase 3 |
| Centers for Disease Control and Prevention | Phase 2/Phase 3 |
| Beijing Chest Hospital | Phase 4 |
Pharmacology for SIRTURO
| Drug Class | Diarylquinoline Antimycobacterial |
US Patents and Regulatory Information for SIRTURO
SIRTURO is protected by two US patents and four FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of SIRTURO is ⤷ Start Trial.
This potential generic entry date is based on patent 8,546,428.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Janssen Therap | SIRTURO | bedaquiline fumarate | TABLET;ORAL | 204384-002 | May 27, 2020 | RX | Yes | No | 8,546,428 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Janssen Therap | SIRTURO | bedaquiline fumarate | TABLET;ORAL | 204384-001 | Dec 28, 2012 | RX | Yes | Yes | 8,546,428 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Janssen Therap | SIRTURO | bedaquiline fumarate | TABLET;ORAL | 204384-002 | May 27, 2020 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Janssen Therap | SIRTURO | bedaquiline fumarate | TABLET;ORAL | 204384-001 | Dec 28, 2012 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Janssen Therap | SIRTURO | bedaquiline fumarate | TABLET;ORAL | 204384-002 | May 27, 2020 | RX | Yes | No | 7,498,343 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for SIRTURO
When does loss-of-exclusivity occur for SIRTURO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
African Regional IP Organization (ARIPO)
Patent: 98
Estimated Expiration: ⤷ Start Trial
Argentina
Patent: 4149
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 07328945
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0719693
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 68512
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 07003472
Estimated Expiration: ⤷ Start Trial
China
Patent: 1547904
Estimated Expiration: ⤷ Start Trial
Patent: 5012303
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0120639
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 13594
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 86940
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 7091
Estimated Expiration: ⤷ Start Trial
Patent: 0970532
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 86940
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 14513
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 9077
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 94239
Estimated Expiration: ⤷ Start Trial
Patent: 10511663
Estimated Expiration: ⤷ Start Trial
Patent: 15028049
Patent: (アルファS,ベータR)−6−ブロモ−アルファ−[2−(ジメチルアミノ)エチル]−2−メトキシ−アルファ−1−ナフタレニル−ベータ−フェニル−3−キノリンエタノールのフマル酸塩 (FUMARATE SALT OF (ALPHA S, BETA R)-6-BROMO-ALPHA-[2-(DIMETHYLAMINO)ETHYL]-2-METHOXY-ALPHA-1-NAPHTHALENYL-BETA-PHENYL-3-QUINOLINEETHANOL)
Estimated Expiration: ⤷ Start Trial
Jordan
Patent: 73
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 8844
Patent: FUMARATE SALT OF (ALPHA S, BETA R)-6- BROMO-ALPHA-[2-(DIMETHYLAMINO)ETHYL]-2-METHOXY-ALPHA-1-NAPHTHALENYL-BETA-PHENYL-3-QUINOLINEETHANOL
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 09005909
Patent: SAL FUMARATO DE (ALFA S, BETA R)-6-BROMO-ALFA-[2-(DIMETILAMINO)ETI L]-2-METOXI-ALFA-1-NAFTALENIL-BETA-FENIL-3-QUINOLINAETANOL. (FUMARATE SALT OF (ALPHA S, BETA R)-6-BROMO-ALPHA-[2-(DIMETHYLAMIN O)ETHYL]-2-METHOXY-ALPHA-1-NAPHTHALENYL-BETA-PHENYL-3-QUINOLINEE THANOL.)
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 456
Patent: FUMARATNA SO (ALFA S, BETA R)-6-BROMO-ALFA-[2-(DIMETILAMINO)ETIL]-2-METOKSI-ALFA-1-NAFTALENIL-BETA-FENIL-3-KINOLINETANOLA (FUMARATE SALT OF (ALPHA S, BETA R)-6-BROMO-ALPHA-[2-(DIMETHYLAMINO)ETHYL]-2-METHOXY-ALPHA-1-NAPHTHALENYL-BETA-PHENYL-3-QUINOLINEETHANOL)
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 6485
Patent: FUMARATE SALT OF (ALPHA S, BETA R)-6-BROMO-ALPHA-[2-(DIMETHYLAMINO)ETHYL]-2-METHOXY-ALPHA-1-NAPHTHALENYL-BETA-PHENYL-3-QUINOLINEETHANOL
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 2773
Estimated Expiration: ⤷ Start Trial
Patent: 092535
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 081350
Patent: SAL FUMARATO DE (ALFAS S, BETA R)-6-BROMO-ALFA-[2(DIMETILAMINO)ETIL]-2-METOXI-ALFA-1-NAFTALENIL-BETA-FENIL-3-QUINOLINAETANOL
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 86940
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 86940
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 408
Patent: FUMARATNA SO (ALFA S, BETA R)-6-BROMO-ALFA-[2-(DIMETILAMINO)ETIL]-2-METOKSI-ALFA-1-NAFTALENIL-BETA-FENIL-3-KINOLINETANOLA (FUMARATE SALT OF (ALPHA S, BETA R)-6-BROMO-ALPHA-[2-(DIMETHYLAMINO)ETHYL]-2¬METHOXY-ALPHA-1-NAPHTHALENYL-BETA-PHENYL-3-QUlNOLINEETHANOL)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 86940
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 0903907
Patent: FUMARATE SALT OF (ALPHA S,BETA R)-6-BROMO-ALPHA-[2-(DIMETHYLAMINO)ETHYL]-2-METHOXY-ALPHA-1-NAPHTHALENYL-BETA-PHENYL-3-QUINOLINEETHANOL
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1514700
Estimated Expiration: ⤷ Start Trial
Patent: 090087020
Patent: FUMARATE SALT OF (ALPHA S, BETA R)-6-BROMO-ALPHA-[2-(DIMETHYLAMINO)ETHYL]-2-METHOXY-ALPHA-1-NAPHTHALENYL-BETA-PHENYL-3-QUINOLINEETHANOL
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 87923
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 17098
Estimated Expiration: ⤷ Start Trial
Patent: 0838527
Patent: Fumarate salt of (alpha S, beta R)-6-bromo-alpha-[2-(dimethylamino)ethyl]-2-methoxy-alpha-1-naphthalenyl-beta-phenyl-3-quinolineethanol
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 813
Patent: ФУМАРАТНАЯ СОЛЬ (АЛЬФА S, БЕТА R)-6-БРОМ-АЛЬФА-[2-(ДИМЕТИЛАМИНО)ЭТИЛ]-2-МЕТОКСИ-АЛЬФА-1-НАФТАЛЕНИЛ-БЕТА-ФЕНИЛ-3-ХИНОЛИНЭТАНОЛА;ФУМАРАТНА СІЛЬ (АЛЬФА S, БЕТА R)-6-БРОМ-АЛЬФА-[2-(ДИМЕТИЛАМІНО)ЕТИЛ]-2-МЕТОКСІ-АЛЬФА-1-НАФТАЛЕНІЛ-БЕТА-ФЕНІЛ-3-ХІНОЛІНЕТАНОЛУ (FUMARATE SALT OF (ALPHA S, BETA R)-6-BROMO-ALPHA-[2-(DIMETHYLAMINO)ETHYL]-2-METHOXY-ALPHA-1-NAPHTHALENYL-BETA-PHENYL-3-QUINOLINEETHANOL)
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 762
Patent: SAL FUMARATO DE (ALFA S, BETA R)-6-BROMO-ALFA-[2(DIMETILAMINO) ETIL]-2-METOXI-ALFA-1-NAFTALENIL-BETA-FENIL-3-QUINOLINAETANOL
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering SIRTURO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Luxembourg | 92520 | ⤷ Start Trial | |
| Eurasian Patent Organization | 200500257 | ПРОИЗВОДНЫЕ ХИНОЛИНА И ИХ ПРИМЕНЕНИЕ В КАЧЕСТВЕ МИКОБАКТЕРИАЛЬНЫХ ИНГИБИТОРОВ | ⤷ Start Trial |
| New Zealand | 576485 | FUMARATE SALT OF (ALPHA S, BETA R)-6-BROMO-ALPHA-[2-(DIMETHYLAMINO)ETHYL]-2-METHOXY-ALPHA-1-NAPHTHALENYL-BETA-PHENYL-3-QUINOLINEETHANOL | ⤷ Start Trial |
| Portugal | 1527050 | ⤷ Start Trial | |
| Mexico | 2009005909 | SAL FUMARATO DE (ALFA S, BETA R)-6-BROMO-ALFA-[2-(DIMETILAMINO)ETI L]-2-METOXI-ALFA-1-NAFTALENIL-BETA-FENIL-3-QUINOLINAETANOL. (FUMARATE SALT OF (ALPHA S, BETA R)-6-BROMO-ALPHA-[2-(DIMETHYLAMIN O)ETHYL]-2-METHOXY-ALPHA-1-NAPHTHALENYL-BETA-PHENYL-3-QUINOLINEE THANOL.) | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for SIRTURO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1527050 | 132014902288493 | Italy | ⤷ Start Trial | PRODUCT NAME: BEDAQUILINA O UN SUO SALE DI ADDIZIONE DI ACIDO O BASE FARMACEUTICAMENTE ACCETTABILE INCLUSO BEDAQUILINA FUMARATO(SIRTURO); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/13/901, 20140305 |
| 1527050 | 92520 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: BEDAQUILINE, OU SON SEL D ADDITION D ACIDE OU DE BASE PHARMACEUTIQUEMENT ACCEPTABLE, Y COMPRIS LE FUMARATE DE BEDAQUILINE. FIRST REGISTRATION: 20140307 |
| 1527050 | C300684 | Netherlands | ⤷ Start Trial | PRODUCT NAME: BEDAQUILINE, OF EEN FARMACEUTISCH AANVAARDBAAR ZUUR- OF BASE-ADDITIEZOUT DAARVAN, WAARONDER BEDAQUILINEFUMARAAT; REGISTRATION NO/DATE: EU/1/13/901 20140307 |
| 1527050 | 50/2014 | Austria | ⤷ Start Trial | PRODUCT NAME: BEDAQUILIN ODER EIN PHARMAZEUTISCH ANNEHMBARES SAEURE- ODER BASENADDITIONSSALZ DAVON, EINSCHLIESSLICH BEDAQUILINFUMARAT; REGISTRATION NO/DATE: EU/1/13/901 20140307 |
| 1527050 | 14C0060 | France | ⤷ Start Trial | PRODUCT NAME: BEDAQUILINE OU UN DE SES SELS D'ADDITION D'ACIDE OU DE BASE PHARMACEUTIQUEMENT ACCEPTABLE, Y COMPRIS LE FUMARATE DE BEDAQUILINE; REGISTRATION NO/DATE: EU/1/13/901 20140307 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for SIRTURO (bedaquiline)
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
