Janssen Therap Company Profile
✉ Email this page to a colleague
What is the competitive landscape for JANSSEN THERAP
JANSSEN THERAP has one approved drug.
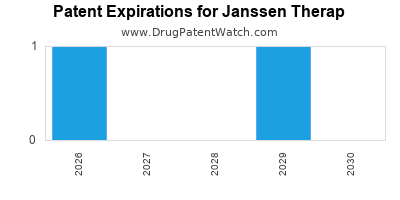
There are two US patents protecting JANSSEN THERAP drugs.
There are eighty-seven patent family members on JANSSEN THERAP drugs in thirty-nine countries and eighteen supplementary protection certificates in fifteen countries.
Drugs and US Patents for Janssen Therap
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Janssen Therap | SIRTURO | bedaquiline fumarate | TABLET;ORAL | 204384-002 | May 27, 2020 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Janssen Therap | SIRTURO | bedaquiline fumarate | TABLET;ORAL | 204384-001 | Dec 28, 2012 | RX | Yes | Yes | 8,546,428 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Janssen Therap | SIRTURO | bedaquiline fumarate | TABLET;ORAL | 204384-002 | May 27, 2020 | RX | Yes | No | 7,498,343 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Janssen Therap | SIRTURO | bedaquiline fumarate | TABLET;ORAL | 204384-001 | Dec 28, 2012 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | |||||
| Janssen Therap | SIRTURO | bedaquiline fumarate | TABLET;ORAL | 204384-001 | Dec 28, 2012 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Janssen Therap Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| World Intellectual Property Organization (WIPO) | 2008068231 | ⤷ Start Trial |
| Montenegro | 00131 | ⤷ Start Trial |
| Peru | 20081350 | ⤷ Start Trial |
| South Korea | 100733577 | ⤷ Start Trial |
| Austria | 463482 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Janssen Therap Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1527050 | CA 2014 00044 | Denmark | ⤷ Start Trial | PRODUCT NAME: BEDAKILIN ELLER ET FARMACEUTISK ACCEPTABELT SYRE-ELLER BASEADDITIONSSALT DERAF, HERUNDER BEDAKILINFUMARAT; REG. NO/DATE: EU/1/13/901 20140305 |
| 1527050 | CR 2014 00044 | Denmark | ⤷ Start Trial | PRODUCT NAME: BEDAKILIN ELLER ET FARMACEUTISK ACCEPTABELT SYRE- ELLER BASEADDITIONSSALT DERAF, HERUNDER BEDAKILINFUMARAT; REG. NO/DATE: EU/1/13/901 20140305 |
| 1527050 | PA2014032 | Lithuania | ⤷ Start Trial | PRODUCT NAME: BEDAQUILINUM; REGISTRATION NO/DATE: EU/1/13/901 20140305 |
| 1527050 | 92520 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: BEDAQUILINE, OU SON SEL D ADDITION D ACIDE OU DE BASE PHARMACEUTIQUEMENT ACCEPTABLE, Y COMPRIS LE FUMARATE DE BEDAQUILINE. FIRST REGISTRATION: 20140307 |
| 1527050 | PA2014032,C1527050 | Lithuania | ⤷ Start Trial | PRODUCT NAME: BEDAQUILINUM; REGISTRATION NO/DATE: EU/1/13/901 20140305 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.