Last updated: February 19, 2026
Takeda Pharmaceuticals USA (Takeda USA) operates as a key subsidiary of Takeda Pharmaceutical Company Limited, a global, R&D-driven biopharmaceutical firm. Takeda USA focuses on the U.S. market, contributing significantly to the parent company's global revenue and pipeline development. The company's strategic priorities center on oncology, rare diseases, neuroscience, and gastroenterology, with a portfolio that includes both established blockbusters and emerging therapies.
What is Takeda USA's Current Market Position?
Takeda USA holds a significant position within the U.S. pharmaceutical market, characterized by a diversified product portfolio and a strategic focus on specialty therapeutics. The company’s market share is driven by its presence in therapeutic areas with high unmet medical needs and significant growth potential.
- Revenue Contribution: Takeda USA consistently contributes a substantial portion of Takeda Pharmaceutical Company Limited's global revenue. In fiscal year 2022, the U.S. market represented approximately 40% of Takeda's total net sales [1].
- Therapeutic Area Focus: The company's market presence is strongest in oncology, rare diseases, neuroscience, and gastroenterology. These areas are characterized by complex patient populations and a demand for innovative treatments.
- Key Product Performance: Takeda USA markets several blockbuster drugs that underpin its market position. For instance, Entyvio (vedolizumab) for inflammatory bowel disease (IBD) and Adcetris (brentuximab vedotin) for lymphoma have demonstrated robust sales growth [2]. Vyvanse (lisdexamfetamine dimesylate) for ADHD, although facing some market shifts, remains a significant product.
- Competitive Landscape: Takeda USA competes with other large multinational pharmaceutical companies, including Pfizer, Novartis, Roche, and Bristol Myers Squibb, particularly within its core therapeutic areas. The competitive environment is marked by patent cliffs, the emergence of biosimilars, and the ongoing quest for novel drug development.
- Pipeline Strength: The company's future market position is influenced by its R&D pipeline, which includes investigational therapies in areas like gene therapy and precision medicine. Early-stage assets in areas such as Alzheimer's disease and novel oncology targets are critical for long-term growth.
What Are Takeda USA's Core Strengths?
Takeda USA's competitive advantage is built upon several key strengths that enable its sustained performance and strategic growth within the dynamic U.S. pharmaceutical landscape.
What is the strength of Takeda USA's R&D Capabilities?
Takeda USA leverages significant R&D investment and a robust global research infrastructure to drive innovation.
- Global R&D Network: Takeda operates a global network of research and development centers, including significant facilities and collaborations within the U.S. This network facilitates access to leading scientific talent and cutting-edge research institutions [3].
- Focus on Modalities: The company emphasizes innovation across various therapeutic modalities, including small molecules, biologics, and gene therapies. This diversified approach allows for tackling complex diseases from multiple angles.
- Strategic Acquisitions and Partnerships: Takeda USA actively pursues strategic acquisitions and licensing agreements to augment its pipeline. The acquisition of Shire in 2019 was a transformative event, significantly strengthening Takeda's rare disease and plasma-derived therapies portfolio, along with its neuroscience and gastroenterology franchises [4].
- Investment in Emerging Technologies: The company invests in cutting-edge research areas such as AI-driven drug discovery and novel delivery systems, aiming to accelerate the development of next-generation therapies.
How do Takeda USA's Therapeutic Areas Contribute to its Market Standing?
Takeda USA's focused approach on specific therapeutic areas allows for deep expertise and market penetration.
- Oncology: Takeda USA has a strong presence in oncology with key products like Adcetris and Imbruvica (ibrutinib), contributing to its position as a significant player in hematological malignancies and solid tumors [5].
- Rare Diseases: Following the Shire acquisition, Takeda USA became a leader in rare diseases, offering treatments for conditions such as hereditary angioedema (HAE) with products like Takhzyro (lanadelumab), and lysosomal storage disorders. This segment is characterized by high unmet needs and premium pricing [6].
- Neuroscience: The company maintains a portfolio in neuroscience, including treatments for ADHD with Vyvanse. Research efforts are ongoing to address neurodegenerative diseases and psychiatric disorders.
- Gastroenterology: Entyvio has established Takeda USA as a dominant force in the treatment of inflammatory bowel disease, including Crohn's disease and ulcerative colitis.
What is the Impact of Takeda USA's Product Portfolio and Pipeline?
The breadth and depth of Takeda USA's product portfolio, coupled with a promising pipeline, are critical to its market standing.
- Established Blockbusters: Products like Entyvio, Adcetris, and historically Vyvanse generate substantial revenue and provide a stable financial base.
- Pipeline Advancements: Takeda USA's pipeline includes several promising candidates in late-stage development and early-stage research. Notable programs include investigational therapies for Alzheimer's disease, multiple sclerosis, and various oncology indications [7].
- Life Cycle Management: The company actively engages in life cycle management for its key products, seeking new indications and formulations to extend their market exclusivity and utility.
What is Takeda USA's Operational and Commercial Excellence?
Takeda USA demonstrates strong commercial execution and operational efficiency in navigating the complex U.S. healthcare system.
- Market Access and Reimbursement: The company possesses considerable expertise in securing market access and favorable reimbursement for its specialty pharmaceuticals, a crucial factor in the U.S. market.
- Sales and Marketing Infrastructure: Takeda USA maintains a robust sales force and marketing infrastructure tailored to reach prescribers and patients in its specialized therapeutic areas.
- Supply Chain Management: Efficient supply chain operations ensure the consistent availability of its products, particularly for therapies requiring specialized handling or distribution.
What are Takeda USA's Strategic Imperatives and Future Outlook?
Takeda USA's strategic direction is shaped by its commitment to innovation, portfolio optimization, and adapting to evolving market dynamics.
How is Takeda USA Evolving its Therapeutic Focus?
The company is continually refining its therapeutic focus to align with emerging scientific opportunities and market needs.
- Continued Investment in Core Areas: Takeda USA will maintain its strong focus on oncology, rare diseases, neuroscience, and gastroenterology, where it has established leadership and possesses significant pipeline assets.
- Expansion into New Modalities: Increasing emphasis is placed on advanced modalities like gene therapy, cell therapy, and RNA-based therapeutics, aiming to address previously intractable diseases.
- Precision Medicine Initiatives: The integration of genomic data and patient stratification is a key strategy to develop more targeted and effective therapies, particularly in oncology.
What are Takeda USA's Strategies for Portfolio Management?
Takeda USA employs a proactive approach to portfolio management to maximize value and manage lifecycle risks.
- Divestitures and Acquisitions: The company has a history of strategic divestitures of non-core assets and targeted acquisitions to bolster its pipeline and expand into new therapeutic areas or technologies. For example, the divestiture of its Shire neuroscience drug, Xiidra (lifitegrast ophthalmic solution) 5%, to Bausch Health in 2023 for up to $1.75 billion, aims to streamline its portfolio and focus resources [8].
- Life Cycle Management: Strategies include seeking new indications for existing drugs, developing new formulations, and exploring combination therapies to enhance efficacy and extend product lifecycles.
- Pipeline Prioritization: Rigorous prioritization of R&D pipeline assets based on scientific merit, market potential, and strategic fit is essential to optimize resource allocation.
How is Takeda USA Addressing Market Access and Reimbursement Challenges?
Navigating the U.S. payer landscape is a critical strategic imperative.
- Value-Based Healthcare Approaches: Takeda USA is exploring and implementing value-based agreements and outcomes-based contracts to demonstrate the economic and clinical value of its therapies to payers.
- Engagement with Stakeholders: Proactive engagement with payers, providers, and patient advocacy groups is essential to foster understanding and secure access for innovative treatments.
- Health Economics and Outcomes Research (HEOR): Robust HEOR data generation is crucial to support market access and reimbursement negotiations, demonstrating the long-term benefits and cost-effectiveness of Takeda's medicines.
What are Takeda USA's Geographic and Operational Strategies?
While its primary focus is the U.S., Takeda USA's operations are integrated within Takeda's global strategy.
- Leveraging Global Capabilities: Takeda USA benefits from Takeda's global manufacturing, supply chain, and R&D network, enabling efficient operations and broad market reach.
- U.S. as a Key Market: The U.S. remains a primary market for Takeda, serving as a launch pad for many of its innovative therapies due to its size, regulatory environment, and reimbursement structures that often support novel treatments.
Key Takeaways
Takeda USA is a significant player in the U.S. pharmaceutical market, driven by a strong portfolio in oncology, rare diseases, neuroscience, and gastroenterology. Its strengths lie in robust R&D capabilities, strategic acquisitions (notably Shire), established blockbuster products, and effective market access strategies. The company's future outlook is focused on continued innovation in advanced modalities, precision medicine, and proactive portfolio management, including strategic divestitures and lifecycle management. Takeda USA's ability to navigate complex reimbursement landscapes and demonstrate the value of its innovative therapies will be critical for sustained growth.
Frequently Asked Questions
1. Which therapeutic areas are Takeda USA's largest revenue drivers?
Takeda USA's largest revenue drivers are primarily in oncology, driven by products like Adcetris, and gastroenterology, with Entyvio as a significant contributor. The rare disease portfolio, significantly bolstered by the Shire acquisition, also represents a substantial and growing revenue stream.
2. What is Takeda USA's approach to gene therapy development?
Takeda USA is actively investing in gene therapy research and development, leveraging its expertise in rare diseases. This includes both internal research programs and strategic partnerships with academic institutions and biotechnology companies specializing in gene editing and delivery technologies.
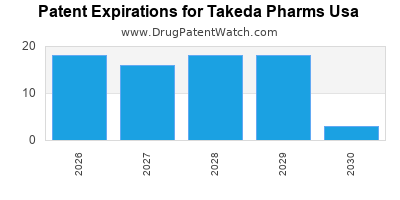
3. How does Takeda USA manage patent expirations for its key products?
Takeda USA employs a multifaceted strategy for managing patent expirations. This includes robust life cycle management efforts to extend product utility through new indications or formulations, and a strong focus on developing and launching next-generation therapies to fill pipeline gaps. The company also engages in legal strategies to defend its intellectual property where appropriate.
4. What impact did the Shire acquisition have on Takeda USA's market position?
The acquisition of Shire significantly transformed Takeda USA's market position by substantially expanding its presence in rare diseases and plasma-derived therapies. It also strengthened its portfolios in neuroscience and gastroenterology, creating a more diversified and robust business.
5. What are Takeda USA's primary R&D collaborations in the United States?
Takeda USA collaborates with numerous U.S.-based academic institutions, research hospitals, and biotechnology firms. These collaborations are focused on areas such as early-stage drug discovery, novel target identification, and clinical trial development across its core therapeutic areas, including oncology, neuroscience, and rare diseases.
Citations
[1] Takeda Pharmaceutical Company Limited. (2023). Integrated Report 2023. Retrieved from [Takeda's Investor Relations website or similar official source]
[2] Takeda Pharmaceutical Company Limited. (2023). Fourth Quarter and Fiscal Year 2022 Earnings Presentation. Retrieved from [Takeda's Investor Relations website or similar official source]
[3] Takeda Pharmaceutical Company Limited. (n.d.). Research & Development. Retrieved from Takeda's official website.
[4] Takeda Pharmaceutical Company Limited. (2019, January 8). Takeda Completes Acquisition of Shire. Press Release.
[5] Takeda Pharmaceutical Company Limited. (2023). Our Portfolio. Retrieved from Takeda's official website.
[6] Takeda Pharmaceutical Company Limited. (2023). Rare Disease Patient Stories. Retrieved from Takeda's official website.
[7] Takeda Pharmaceutical Company Limited. (n.d.). Pipeline. Retrieved from Takeda's official website.
[8] Takeda Pharmaceutical Company Limited. (2023, February 22). Takeda to Divest Xiidra® to Bausch Health. Press Release.