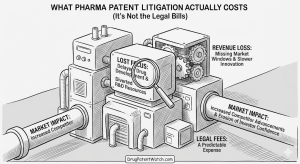
What Pharma Patent Litigation Actually Costs (It’s Not the Legal Bills)
How drug companies, insurers, payers, and patients absorb billions in collateral damage that never appears on a litigation docket “The […]
What Pharma Patent Litigation Actually Costs (It’s Not the Legal Bills) Read Post »