Expiring Drug Patents Cheat Sheet
We analyse the patents covering drugs in 134 countries and quickly give you the likely loss-of-exclusivity/generic entry date
United States: These 64 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "United States: These 64 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027" DrugPatentWatch.com thinkBiotech, 2026 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
When can NATAZIA (dienogest; estradiol valerate) generic drug versions launch?
Generic name: dienogest; estradiol valerate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2026
Generic Entry Controlled by: United States Patent Patent 8,071,577
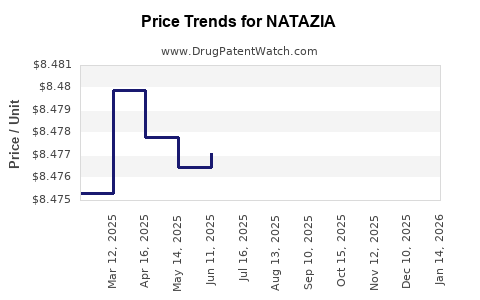
This drug has ninety patent family members in thirty-nine countries. There has been litigation on patents covering NATAZIA
See drug price trends for NATAZIA.
The generic ingredient in NATAZIA is dienogest; estradiol valerate. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the dienogest; estradiol valerate profile page.
When can GATTEX KIT (teduglutide recombinant) generic drug versions launch?
Generic name: teduglutide recombinant
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 16, 2026
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
GATTEX KIT is a drug marketed by Takeda Pharms Usa. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has eight patent family members in seven countries. There has been litigation on patents covering GATTEX KIT
The generic ingredient in GATTEX KIT is teduglutide recombinant. There is one drug master file entry for this API. Two suppliers are listed for this generic product. Additional details are available on the teduglutide recombinant profile page.
When can ZOLINZA (vorinostat) generic drug versions launch?
Generic name: vorinostat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 16, 2026
Generic Entry Controlled by: United States Patent Patent 8,093,295
ZOLINZA is a drug marketed by Msd Sub Merck. There are three patents protecting this drug.
This drug has one hundred and thirty-one patent family members in thirty-five countries.
See drug price trends for ZOLINZA.
The generic ingredient in ZOLINZA is vorinostat. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the vorinostat profile page.
When can OMONTYS (peginesatide acetate) generic drug versions launch?
Generic name: peginesatide acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 02, 2026
Generic Entry Controlled by: United States Patent Patent 7,550,433
OMONTYS is a drug marketed by Takeda Pharms Usa. There are two patents protecting this drug.
This drug has twenty-seven patent family members in eighteen countries.
The generic ingredient in OMONTYS is peginesatide acetate. Additional details are available on the peginesatide acetate profile page.
When can SUPPRELIN LA (histrelin acetate) generic drug versions launch?
Generic name: histrelin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 16, 2026
Generic Entry Controlled by: United States Patent Patent 8,062,652
SUPPRELIN LA is a drug marketed by Endo Operations. There is one patent protecting this drug.
This drug has three patent family members in three countries.
See drug price trends for SUPPRELIN LA.
The generic ingredient in SUPPRELIN LA is histrelin acetate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the histrelin acetate profile page.
When can APLENZIN (bupropion hydrobromide) generic drug versions launch?
Generic name: bupropion hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 27, 2026
Generic Entry Controlled by: United States Patent Patent 7,572,935
APLENZIN is a drug marketed by Bausch. There are eight patents protecting this drug and three Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has fifty-two patent family members in eighteen countries. There has been litigation on patents covering APLENZIN
See drug price trends for APLENZIN.
The generic ingredient in APLENZIN is bupropion hydrobromide. There are thirty-eight drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the bupropion hydrobromide profile page.
When can VALTURNA (aliskiren hemifumarate; valsartan) generic drug versions launch?
Generic name: aliskiren hemifumarate; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 03, 2026
Generic Entry Controlled by: United States Patent Patent 8,168,616
VALTURNA is a drug marketed by Novartis. There is one patent protecting this drug.
This drug has ninety-three patent family members in thirty-one countries.
The generic ingredient in VALTURNA is aliskiren hemifumarate; valsartan. There are four drug master file entries for this API. Additional details are available on the aliskiren hemifumarate; valsartan profile page.
When can OPILL (norgestrel) generic drug versions launch?
Generic name: norgestrel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
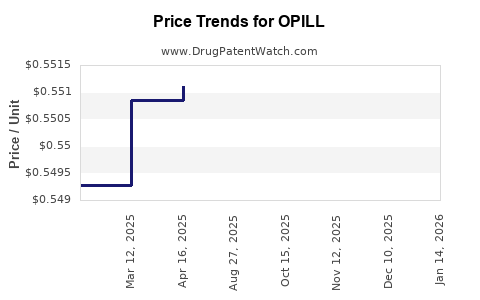
This drug has ninety-three patent family members in thirty-one countries.
See drug price trends for OPILL.
The generic ingredient in OPILL is norgestrel. There are twenty-five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the norgestrel profile page.
When can KAZANO (alogliptin benzoate; metformin hydrochloride) generic drug versions launch?
Generic name: alogliptin benzoate; metformin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 27, 2026
Generic Entry Controlled by: United States FDA Regulatory Exclusivity

This drug has ninety patent family members in forty-one countries. There has been litigation on patents covering KAZANO
See drug price trends for KAZANO.
The generic ingredient in KAZANO is alogliptin benzoate; metformin hydrochloride. There are ten drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate; metformin hydrochloride profile page.
When can NESINA (alogliptin benzoate) generic drug versions launch?
Generic name: alogliptin benzoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 27, 2026
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
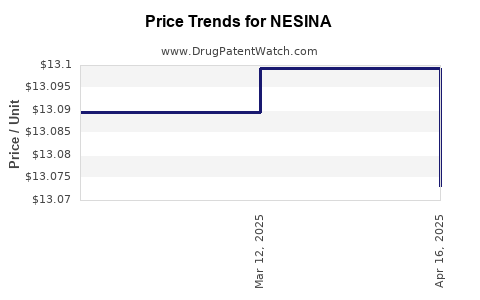
This drug has sixty-nine patent family members in thirty-seven countries. There has been litigation on patents covering NESINA
See drug price trends for NESINA.
The generic ingredient in NESINA is alogliptin benzoate. There are ten drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate profile page.
When can OLYSIO (simeprevir sodium) generic drug versions launch?
Generic name: simeprevir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 28, 2026
Generic Entry Controlled by: United States Patent Patent 8,349,869
OLYSIO is a drug marketed by Janssen Prods. There are eight patents protecting this drug.
This drug has sixty-four patent family members in forty-one countries.
See drug price trends for OLYSIO.
The generic ingredient in OLYSIO is simeprevir sodium. There is one drug master file entry for this API. Additional details are available on the simeprevir sodium profile page.
When can OMEGAVEN (fish oil triglycerides) generic drug versions launch?
Generic name: fish oil triglycerides
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 30, 2026
Generic Entry Controlled by: United States Patent Patent 9,566,260
OMEGAVEN is a drug marketed by Fresenius Kabi Usa. There is one patent protecting this drug.
This drug has five patent family members in four countries. There has been litigation on patents covering OMEGAVEN
See drug price trends for OMEGAVEN.
The generic ingredient in OMEGAVEN is fish oil triglycerides. There are six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the fish oil triglycerides profile page.
When can GALLIUM DOTATOC GA 68 (gallium dotatoc ga-68) generic drug versions launch?
Generic name: gallium dotatoc ga-68
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 21, 2026
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
GALLIUM DOTATOC GA 68 is a drug marketed by
This drug has five patent family members in four countries.
The generic ingredient in GALLIUM DOTATOC GA 68 is gallium dotatoc ga-68. There are sixteen drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the gallium dotatoc ga-68 profile page.
When can TECHNEGAS KIT (technetium tc-99m labeled carbon) generic drug versions launch?
Generic name: technetium tc-99m labeled carbon
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 29, 2026
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
TECHNEGAS KIT is a drug marketed by Cyclomedica. There is one patent protecting this drug.
This drug has fourteen patent family members in ten countries.
The generic ingredient in TECHNEGAS KIT is technetium tc-99m labeled carbon. There are four drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the technetium tc-99m labeled carbon profile page.
When can VEREGEN (sinecatechins) generic drug versions launch?
Generic name: sinecatechins
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 02, 2026
Generic Entry Controlled by: United States Patent Patent 5,795,911
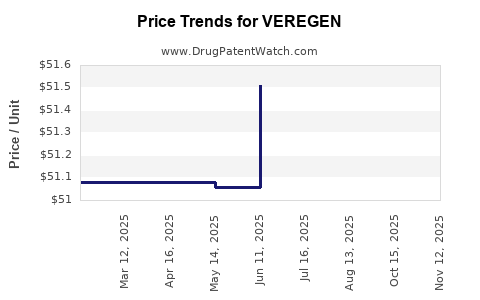
This drug has thirty-one patent family members in twenty countries. There has been litigation on patents covering VEREGEN
See drug price trends for VEREGEN.
The generic ingredient in VEREGEN is sinecatechins. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the sinecatechins profile page.
When can SCENESSE (afamelanotide) generic drug versions launch?
Generic name: afamelanotide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 08, 2026
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
SCENESSE is a drug marketed by Clivunel Inc. There is one patent protecting this drug.
This drug has twenty-three patent family members in sixteen countries.
See drug price trends for SCENESSE.
The generic ingredient in SCENESSE is afamelanotide. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the afamelanotide profile page.
When can ADASUVE (loxapine) generic drug versions launch?
Generic name: loxapine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 23, 2026
Generic Entry Controlled by: United States Patent Patent 8,387,612
ADASUVE is a drug marketed by Nova Pneuma. There is one patent protecting this drug.
This drug has twenty-one patent family members in six countries. There has been litigation on patents covering ADASUVE
See drug price trends for ADASUVE.
The generic ingredient in ADASUVE is loxapine. There are eight drug master file entries for this API. Additional details are available on the loxapine profile page.
When can SYNRIBO (omacetaxine mepesuccinate) generic drug versions launch?
Generic name: omacetaxine mepesuccinate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: United States Patent Patent 6,987,103
SYNRIBO is a drug marketed by Teva Pharms Intl. There is one patent protecting this drug and one Paragraph IV challenge.
This drug has twenty-one patent family members in twelve countries.
See drug price trends for SYNRIBO.
The generic ingredient in SYNRIBO is omacetaxine mepesuccinate. There are two drug master file entries for this API. Additional details are available on the omacetaxine mepesuccinate profile page.
When can XEGLYZE (abametapir) generic drug versions launch?
Generic name: abametapir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 28, 2026
Generic Entry Controlled by: United States Patent Patent 7,812,163
XEGLYZE is a drug marketed by Hatchtech. There are two patents protecting this drug.
This drug has thirty-six patent family members in fifteen countries.
The generic ingredient in XEGLYZE is abametapir. Additional details are available on the abametapir profile page.
When can BEVYXXA (betrixaban) generic drug versions launch?
Generic name: betrixaban
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 08, 2026
Generic Entry Controlled by: United States Patent Patent 7,598,276
BEVYXXA is a drug marketed by Portola Pharms Inc. There are five patents protecting this drug.
This drug has sixty-seven patent family members in twenty-six countries. There has been litigation on patents covering BEVYXXA
See drug price trends for BEVYXXA.
The generic ingredient in BEVYXXA is betrixaban. Additional details are available on the betrixaban profile page.
When can HORIZANT (gabapentin enacarbil) generic drug versions launch?
Generic name: gabapentin enacarbil
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 10, 2026
Generic Entry Controlled by: United States Patent Patent 8,026,279
HORIZANT is a drug marketed by Azurity. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has seventy-three patent family members in twenty-four countries.
See drug price trends for HORIZANT.
The generic ingredient in HORIZANT is gabapentin enacarbil. There are twenty-nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the gabapentin enacarbil profile page.
When can IZERVAY (avacincaptad pegol sodium) generic drug versions launch?
Generic name: avacincaptad pegol sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 11, 2026
Generic Entry Controlled by: United States Patent Patent 8,236,773
IZERVAY is a drug marketed by Astellas. There are six patents protecting this drug.
This drug has one hundred and thirteen patent family members in thirty countries.
See drug price trends for IZERVAY.
The generic ingredient in IZERVAY is avacincaptad pegol sodium. One supplier is listed for this generic product. Additional details are available on the avacincaptad pegol sodium profile page.
When can AXUMIN (fluciclovine f-18) generic drug versions launch?
Generic name: fluciclovine f-18
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 28, 2026
Generic Entry Controlled by: United States Patent Patent 10,010,632
AXUMIN is a drug marketed by Blue Earth. There are eight patents protecting this drug.
This drug has thirty patent family members in sixteen countries. There has been litigation on patents covering AXUMIN
The generic ingredient in AXUMIN is fluciclovine f-18. One supplier is listed for this generic product. Additional details are available on the fluciclovine f-18 profile page.
When can ZUNVEYL (benzgalantamine gluconate) generic drug versions launch?
Generic name: benzgalantamine gluconate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 01, 2026
Generic Entry Controlled by: United States Patent Patent 9,763,953
ZUNVEYL is a drug marketed by Alpha Cognition. There are four patents protecting this drug.
This drug has twenty-six patent family members in seventeen countries. There has been litigation on patents covering ZUNVEYL
The generic ingredient in ZUNVEYL is benzgalantamine gluconate. One supplier is listed for this generic product. Additional details are available on the benzgalantamine gluconate profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 12, 2026
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There is one patent protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can SIGNIFOR (pasireotide diaspartate) generic drug versions launch?
Generic name: pasireotide diaspartate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 14, 2026
Generic Entry Controlled by: United States Patent Patent 7,473,761
SIGNIFOR is a drug marketed by Recordati Rare. There are three patents protecting this drug.
This drug has forty-five patent family members in thirty-three countries.
See drug price trends for SIGNIFOR.
The generic ingredient in SIGNIFOR is pasireotide diaspartate. One supplier is listed for this generic product. Additional details are available on the pasireotide diaspartate profile page.
When can VYNDAMAX (tafamidis) generic drug versions launch?
Generic name: tafamidis
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 19, 2026
Generic Entry Controlled by: United States Patent Patent 7,214,695
VYNDAMAX is a drug marketed by Foldrx Pharms. There are three patents protecting this drug.
This drug has sixty-six patent family members in twenty-seven countries. There has been litigation on patents covering VYNDAMAX
See drug price trends for VYNDAMAX.
The generic ingredient in VYNDAMAX is tafamidis. One supplier is listed for this generic product. Additional details are available on the tafamidis profile page.
When can VYNDAQEL (tafamidis meglumine) generic drug versions launch?
Generic name: tafamidis meglumine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 19, 2026
Generic Entry Controlled by: United States Patent Patent 7,214,695
VYNDAQEL is a drug marketed by Foldrx Pharms. There are two patents protecting this drug.
This drug has thirty-five patent family members in seventeen countries. There has been litigation on patents covering VYNDAQEL
See drug price trends for VYNDAQEL.
The generic ingredient in VYNDAQEL is tafamidis meglumine. One supplier is listed for this generic product. Additional details are available on the tafamidis meglumine profile page.
When can EPANOVA (omega-3-carboxylic acids) generic drug versions launch?
Generic name: omega-3-carboxylic acids
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 20, 2026
Generic Entry Controlled by: United States Patent Patent 7,960,370
EPANOVA is a drug marketed by Astrazeneca. There are four patents protecting this drug.
This drug has eighty-five patent family members in forty-one countries.
The generic ingredient in EPANOVA is omega-3-carboxylic acids. There is one drug master file entry for this API. Additional details are available on the omega-3-carboxylic acids profile page.
When can TISSUEBLUE (brilliant blue g) generic drug versions launch?
Generic name: brilliant blue g
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 20, 2026
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
TISSUEBLUE is a drug marketed by Dutch Ophthalmic.
This drug has eighty-five patent family members in forty-one countries.
The generic ingredient in TISSUEBLUE is brilliant blue g. One supplier is listed for this generic product. Additional details are available on the brilliant blue g profile page.
When can SYMTUZA (cobicistat; darunavir; emtricitabine; tenofovir alafenamide fumarate) generic drug versions launch?
Generic name: cobicistat; darunavir; emtricitabine; tenofovir alafenamide fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 26, 2026
Generic Entry Controlled by: United States Patent Patent 7,700,645
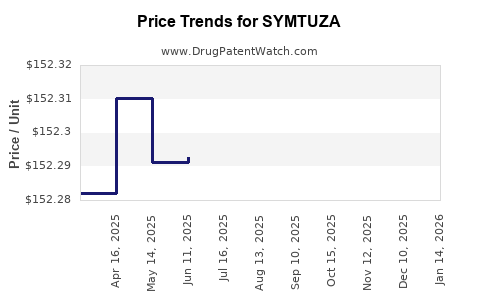
This drug has four hundred and twenty-three patent family members in forty-six countries. There has been litigation on patents covering SYMTUZA
See drug price trends for SYMTUZA.
The generic ingredient in SYMTUZA is cobicistat; darunavir; emtricitabine; tenofovir alafenamide fumarate. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the cobicistat; darunavir; emtricitabine; tenofovir alafenamide fumarate profile page.
When can VIBATIV (telavancin hydrochloride) generic drug versions launch?
Generic name: telavancin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 01, 2027
Generic Entry Controlled by: United States Patent Patent 7,531,623
VIBATIV is a drug marketed by Cumberland. There is one patent protecting this drug.
This drug has thirty-three patent family members in eighteen countries.
See drug price trends for VIBATIV.
The generic ingredient in VIBATIV is telavancin hydrochloride. One supplier is listed for this generic product. Additional details are available on the telavancin hydrochloride profile page.
When can PIQRAY (alpelisib) generic drug versions launch?
Generic name: alpelisib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 18, 2027
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
PIQRAY is a drug marketed by Novartis. There are two patents protecting this drug.
This drug has sixty patent family members in fifty countries.
See drug price trends for PIQRAY.
The generic ingredient in PIQRAY is alpelisib. One supplier is listed for this generic product. Additional details are available on the alpelisib profile page.
When can STIOLTO RESPIMAT (olodaterol hydrochloride; tiotropium bromide) generic drug versions launch?
Generic name: olodaterol hydrochloride; tiotropium bromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 19, 2027
Generic Entry Controlled by: United States Patent Patent 7,727,984
STIOLTO RESPIMAT is a drug marketed by Boehringer Ingelheim. There are five patents protecting this drug.
This drug has one hundred and twenty-three patent family members in forty countries. There has been litigation on patents covering STIOLTO RESPIMAT
See drug price trends for STIOLTO RESPIMAT.
The generic ingredient in STIOLTO RESPIMAT is olodaterol hydrochloride; tiotropium bromide. One supplier is listed for this generic product. Additional details are available on the olodaterol hydrochloride; tiotropium bromide profile page.
When can STRIVERDI RESPIMAT (olodaterol hydrochloride) generic drug versions launch?
Generic name: olodaterol hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 19, 2027
Generic Entry Controlled by: United States Patent Patent 7,727,984
STRIVERDI RESPIMAT is a drug marketed by Boehringer Ingelheim. There is one patent protecting this drug.
This drug has fifty-one patent family members in thirty-six countries. There has been litigation on patents covering STRIVERDI RESPIMAT
See drug price trends for STRIVERDI RESPIMAT.
The generic ingredient in STRIVERDI RESPIMAT is olodaterol hydrochloride. One supplier is listed for this generic product. Additional details are available on the olodaterol hydrochloride profile page.
When can KYNAMRO (mipomersen sodium) generic drug versions launch?
Generic name: mipomersen sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 29, 2027
Generic Entry Controlled by: United States Patent Patent 7,511,131
KYNAMRO is a drug marketed by Kastle Theraps Llc. There is one patent protecting this drug.
This drug has forty-five patent family members in eleven countries. There has been litigation on patents covering KYNAMRO
See drug price trends for KYNAMRO.
The generic ingredient in KYNAMRO is mipomersen sodium. Additional details are available on the mipomersen sodium profile page.
When can NEVANAC (nepafenac) generic drug versions launch?
Generic name: nepafenac
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 31, 2027
Generic Entry Controlled by: United States Patent Patent 7,834,059
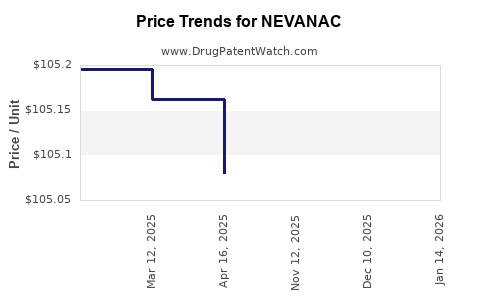
This drug has twenty-seven patent family members in twenty-three countries.
See drug price trends for NEVANAC.
The generic ingredient in NEVANAC is nepafenac. There are eight drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the nepafenac profile page.
When can INJECTAFER (ferric carboxymaltose) generic drug versions launch?
Generic name: ferric carboxymaltose
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 05, 2027
Generic Entry Controlled by: United States Patent Patent 7,612,109
INJECTAFER is a drug marketed by Am Regent. There are six patents protecting this drug.
This drug has seventy-one patent family members in thirty-two countries. There has been litigation on patents covering INJECTAFER
See drug price trends for INJECTAFER.
The generic ingredient in INJECTAFER is ferric carboxymaltose. There are twenty drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ferric carboxymaltose profile page.
When can RAPIVAB (peramivir) generic drug versions launch?
Generic name: peramivir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 12, 2027
Generic Entry Controlled by: United States Patent Patent 10,391,075
RAPIVAB is a drug marketed by Biocryst. There are two patents protecting this drug.
This drug has forty-three patent family members in fourteen countries.
See drug price trends for RAPIVAB.
The generic ingredient in RAPIVAB is peramivir. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the peramivir profile page.
When can LEGUBETI (acetylcysteine lysine) generic drug versions launch?
Generic name: acetylcysteine lysine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 13, 2027
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
LEGUBETI is a drug marketed by Galephar.
This drug has forty-three patent family members in fourteen countries.
The generic ingredient in LEGUBETI is acetylcysteine lysine. There are three drug master file entries for this API. Additional details are available on the acetylcysteine lysine profile page.
When can ALTABAX (retapamulin) generic drug versions launch?
Generic name: retapamulin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 14, 2027
Generic Entry Controlled by: United States Patent Patent 7,875,630
ALTABAX is a drug marketed by Almirall. There is one patent protecting this drug.
This drug has thirteen patent family members in six countries.
See drug price trends for ALTABAX.
The generic ingredient in ALTABAX is retapamulin. Additional details are available on the retapamulin profile page.
When can ADVIL ALLERGY SINUS (chlorpheniramine maleate; ibuprofen; pseudoephedrine hydrochloride) generic drug versions launch?
Generic name: chlorpheniramine maleate; ibuprofen; pseudoephedrine hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 28, 2027
Generic Entry Controlled by: United States Patent Patent 7,863,287
ADVIL ALLERGY SINUS is a drug marketed by Haleon Us Holdings. There is one patent protecting this drug.
This drug has thirty-four patent family members in eighteen countries.
The generic ingredient in ADVIL ALLERGY SINUS is chlorpheniramine maleate; ibuprofen; pseudoephedrine hydrochloride. There are twenty-nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the chlorpheniramine maleate; ibuprofen; pseudoephedrine hydrochloride profile page.
When can DUAVEE (bazedoxifene acetate; estrogens, conjugated) generic drug versions launch?
Generic name: bazedoxifene acetate; estrogens, conjugated
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 10, 2027
Generic Entry Controlled by: United States Patent Patent 7,683,051
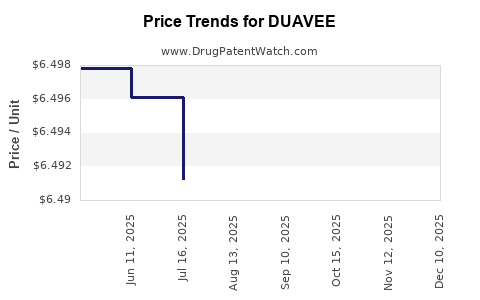
This drug has twenty-two patent family members in twenty countries.
See drug price trends for DUAVEE.
The generic ingredient in DUAVEE is bazedoxifene acetate; estrogens, conjugated. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the bazedoxifene acetate; estrogens, conjugated profile page.
When can LIVMARLI (maralixibat chloride) generic drug versions launch?
Generic name: maralixibat chloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 13, 2027
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
LIVMARLI is a drug marketed by Mirum. There are nine patents protecting this drug.
This drug has one hundred and thirty-four patent family members in twenty-four countries. There has been litigation on patents covering LIVMARLI
See drug price trends for LIVMARLI.
The generic ingredient in LIVMARLI is maralixibat chloride. One supplier is listed for this generic product. Additional details are available on the maralixibat chloride profile page.
When can AVEED (testosterone undecanoate) generic drug versions launch?
Generic name: testosterone undecanoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 14, 2027
Generic Entry Controlled by: United States Patent Patent 7,718,640
AVEED is a drug marketed by Endo Operations. There are two patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has fifty-two patent family members in thirty-eight countries. There has been litigation on patents covering AVEED
See drug price trends for AVEED.
The generic ingredient in AVEED is testosterone undecanoate. There are sixty-nine drug master file entries for this API. Four suppliers are listed for this generic product. Additional details are available on the testosterone undecanoate profile page.
When can DYANAVEL XR 10 (amphetamine; amphetamine aspartate/dextroamphetamine sulfate) generic drug versions launch?
Generic name: amphetamine; amphetamine aspartate/dextroamphetamine sulfate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 15, 2027
Generic Entry Controlled by: United States Patent Patent 8,747,902
DYANAVEL XR 10 is a drug marketed by Tris Pharma Inc. There are five patents protecting this drug.
This drug has twenty-one patent family members in fourteen countries.
The generic ingredient in DYANAVEL XR 10 is amphetamine; amphetamine aspartate/dextroamphetamine sulfate. There are fifty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the amphetamine; amphetamine aspartate/dextroamphetamine sulfate profile page.
When can VICTRELIS (boceprevir) generic drug versions launch?
Generic name: boceprevir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 17, 2027
Generic Entry Controlled by: United States Patent Patent 8,119,602
VICTRELIS is a drug marketed by Merck Sharp Dohme. There are two patents protecting this drug.
This drug has twenty-seven patent family members in seventeen countries.
The generic ingredient in VICTRELIS is boceprevir. Additional details are available on the boceprevir profile page.
When can VARUBI (rolapitant hydrochloride) generic drug versions launch?
Generic name: rolapitant hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 04, 2027
Generic Entry Controlled by: United States Patent Patent 8,178,550
VARUBI is a drug marketed by Tersera. There are eight patents protecting this drug.
This drug has one hundred and fifty-eight patent family members in thirty-five countries.
The generic ingredient in VARUBI is rolapitant hydrochloride. One supplier is listed for this generic product. Additional details are available on the rolapitant hydrochloride profile page.
When can SAVAYSA (edoxaban tosylate) generic drug versions launch?
Generic name: edoxaban tosylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 18, 2027
Generic Entry Controlled by: United States Patent Patent 7,365,205
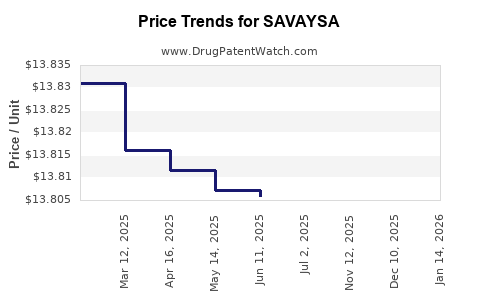
This drug has one hundred and one patent family members in thirty-four countries. There has been litigation on patents covering SAVAYSA
See drug price trends for SAVAYSA.
The generic ingredient in SAVAYSA is edoxaban tosylate. There are four drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the edoxaban tosylate profile page.
When can CLINOLIPID 20% (olive oil; soybean oil) generic drug versions launch?
Generic name: olive oil; soybean oil
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 24, 2027
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
CLINOLIPID 20% is a drug marketed by Baxter Hlthcare Corp.
This drug has one hundred and one patent family members in thirty-four countries.
The generic ingredient in CLINOLIPID 20% is olive oil; soybean oil. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the olive oil; soybean oil profile page.
When can CHLORAPREP ONE-STEP (chlorhexidine gluconate; isopropyl alcohol) generic drug versions launch?
Generic name: chlorhexidine gluconate; isopropyl alcohol
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 25, 2027
Generic Entry Controlled by: United States Patent Patent 7,422,388
CHLORAPREP ONE-STEP is a drug marketed by Becton Dickinson Co.
This drug has one hundred and one patent family members in thirty-four countries.
The generic ingredient in CHLORAPREP ONE-STEP is chlorhexidine gluconate; isopropyl alcohol. There are fifty-eight drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the chlorhexidine gluconate; isopropyl alcohol profile page.
When can GENVOYA (cobicistat; elvitegravir; emtricitabine; tenofovir alafenamide fumarate) generic drug versions launch?
Generic name: cobicistat; elvitegravir; emtricitabine; tenofovir alafenamide fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 26, 2027
Generic Entry Controlled by: United States Patent Patent 8,981,103
GENVOYA is a drug marketed by Gilead Sciences Inc. There are nine patents protecting this drug.
This drug has four hundred and sixty-three patent family members in fifty-three countries. There has been litigation on patents covering GENVOYA
See drug price trends for GENVOYA.
The generic ingredient in GENVOYA is cobicistat; elvitegravir; emtricitabine; tenofovir alafenamide fumarate. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the cobicistat; elvitegravir; emtricitabine; tenofovir alafenamide fumarate profile page.
When can STRIBILD (cobicistat; elvitegravir; emtricitabine; tenofovir disoproxil fumarate) generic drug versions launch?
Generic name: cobicistat; elvitegravir; emtricitabine; tenofovir disoproxil fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 26, 2027
Generic Entry Controlled by: United States Patent Patent 8,981,103
STRIBILD is a drug marketed by Gilead Sciences Inc. There are seven patents protecting this drug and one Paragraph IV challenge.
This drug has four hundred and five patent family members in forty-eight countries. There has been litigation on patents covering STRIBILD
See drug price trends for STRIBILD.
The generic ingredient in STRIBILD is cobicistat; elvitegravir; emtricitabine; tenofovir disoproxil fumarate. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the cobicistat; elvitegravir; emtricitabine; tenofovir disoproxil fumarate profile page.
When can VITEKTA (elvitegravir) generic drug versions launch?
Generic name: elvitegravir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 26, 2027
Generic Entry Controlled by: United States Patent Patent 8,981,103
VITEKTA is a drug marketed by Gilead Sciences Inc. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has ninety-two patent family members in thirty-six countries. There has been litigation on patents covering VITEKTA
The generic ingredient in VITEKTA is elvitegravir. There are six drug master file entries for this API. Additional details are available on the elvitegravir profile page.
When can AMYVID (florbetapir f-18) generic drug versions launch?
Generic name: florbetapir f-18
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 30, 2027
Generic Entry Controlled by: United States Patent Patent 7,687,052
AMYVID is a drug marketed by Avid Radiopharms Inc. There are two patents protecting this drug.
This drug has fifty-one patent family members in thirty-three countries. There has been litigation on patents covering AMYVID
The generic ingredient in AMYVID is florbetapir f-18. One supplier is listed for this generic product. Additional details are available on the florbetapir f-18 profile page.
When can VELTASSA (patiromer sorbitex calcium) generic drug versions launch?
Generic name: patiromer sorbitex calcium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 29, 2027
Generic Entry Controlled by: United States Patent Patent 8,282,913
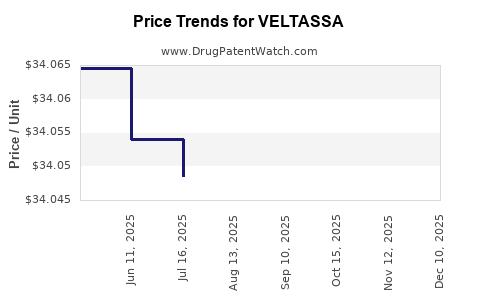
This drug has two hundred and seven patent family members in twenty-seven countries. There has been litigation on patents covering VELTASSA
See drug price trends for VELTASSA.
The generic ingredient in VELTASSA is patiromer sorbitex calcium. One supplier is listed for this generic product. Additional details are available on the patiromer sorbitex calcium profile page.
When can CONTRAVE (bupropion hydrochloride; naltrexone hydrochloride) generic drug versions launch?
Generic name: bupropion hydrochloride; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 04, 2027
Generic Entry Controlled by: United States Patent Patent 9,107,837
CONTRAVE is a drug marketed by Nalpropion. There are eighteen patents protecting this drug and one Paragraph IV challenge.
This drug has two hundred and twenty-seven patent family members in forty-five countries. There has been litigation on patents covering CONTRAVE
See drug price trends for CONTRAVE.
The generic ingredient in CONTRAVE is bupropion hydrochloride; naltrexone hydrochloride. There are thirty-eight drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the bupropion hydrochloride; naltrexone hydrochloride profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2027
Generic Entry Controlled by: United States Patent Patent 8,158,156
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 21, 2027
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can PREZCOBIX (cobicistat; darunavir) generic drug versions launch?
Generic name: cobicistat; darunavir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 26, 2027
Generic Entry Controlled by: United States Patent Patent 7,700,645
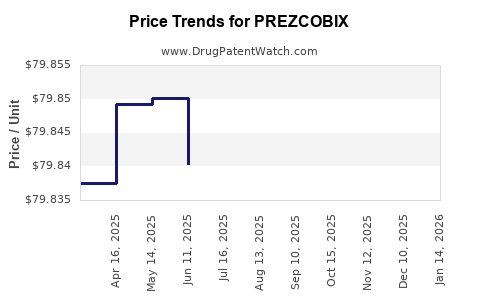
This drug has three hundred and fifty-four patent family members in forty-one countries. There has been litigation on patents covering PREZCOBIX
See drug price trends for PREZCOBIX.
The generic ingredient in PREZCOBIX is cobicistat; darunavir. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the cobicistat; darunavir profile page.
When can PARAGARD T 380A (copper) generic drug versions launch?
Generic name: copper
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2027
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
PARAGARD T 380A is a drug marketed by Coopersurgical.
This drug has three hundred and fifty-four patent family members in forty-one countries.
The generic ingredient in PARAGARD T 380A is copper. There are fifteen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the copper profile page.
When can DOJOLVI (triheptanoin) generic drug versions launch?
Generic name: triheptanoin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 30, 2027
Generic Entry Controlled by: United States FDA Regulatory Exclusivity
DOJOLVI is a drug marketed by Ultragenyx Pharm Inc. There are two patents protecting this drug.
This drug has thirty-six patent family members in seventeen countries.
See drug price trends for DOJOLVI.
The generic ingredient in DOJOLVI is triheptanoin. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the triheptanoin profile page.
When can BYFAVO (remimazolam besylate) generic drug versions launch?
Generic name: remimazolam besylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 10, 2027
Generic Entry Controlled by: United States Patent Patent 10,961,250
BYFAVO is a drug marketed by Acacia. There are eleven patents protecting this drug.
This drug has fifty-three patent family members in twenty-four countries. There has been litigation on patents covering BYFAVO
See drug price trends for BYFAVO.
The generic ingredient in BYFAVO is remimazolam besylate. One supplier is listed for this generic product. Additional details are available on the remimazolam besylate profile page.
When can SPINRAZA (nusinersen sodium) generic drug versions launch?
Generic name: nusinersen sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 11, 2027
Generic Entry Controlled by: United States Patent Patent 7,838,657
SPINRAZA is a drug marketed by Biogen Idec. There are seven patents protecting this drug.
This drug has one hundred and six patent family members in twenty-eight countries. There has been litigation on patents covering SPINRAZA
See drug price trends for SPINRAZA.
The generic ingredient in SPINRAZA is nusinersen sodium. One supplier is listed for this generic product. Additional details are available on the nusinersen sodium profile page.
United States Branded and Generic Drug Markets Assessment and Regulatory Opportunities and Challenges
More… ↓
DrugPatentWatch cited by CNN, NEJM, Nature Journals, and more …

Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
