Share This Page
Drug Price Trends for BRILINTA
✉ Email this page to a colleague
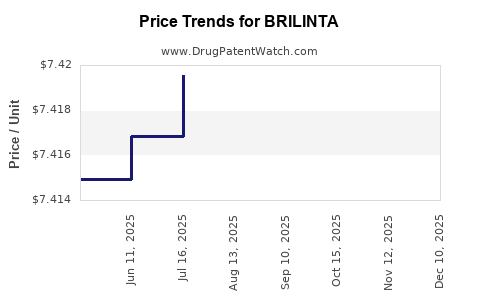
Average Pharmacy Cost for BRILINTA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BRILINTA 90 MG TABLET | 00186-0777-60 | 7.42305 | EACH | 2026-03-18 |
| BRILINTA 60 MG TABLET | 00186-0776-60 | 7.42787 | EACH | 2026-03-18 |
| BRILINTA 90 MG TABLET | 00186-0777-39 | 7.42305 | EACH | 2026-03-18 |
| BRILINTA 60 MG TABLET | 00186-0776-60 | 7.42595 | EACH | 2026-02-18 |
| BRILINTA 90 MG TABLET | 00186-0777-39 | 7.42161 | EACH | 2026-02-18 |
| BRILINTA 90 MG TABLET | 00186-0777-60 | 7.42161 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Brilinta Market Analysis and Price Projections
Brilinta (ticagrelor), developed by AstraZeneca, is a P2Y12 platelet inhibitor approved for reducing the rate of thrombotic cardiovascular events in patients with acute coronary syndrome (ACS) or prior myocardial infarction (MI). It competes primarily with clopidogrel, prasugrel, and newer antiplatelet agents.
Market Size and Growth Drivers
As of 2023, the global antiplatelet drugs market is valued at approximately $11 billion, with Brilinta holding a 20% share. Its revenue in 2022 reached $2.2 billion, reflecting sustained demand driven by proven efficacy and favorable safety profile compared to alternatives.
Key growth drivers:
- Aging populations increase the incidence of ACS and MI.
- Clinical guidelines favor ticagrelor for certain patient populations.
- Increased adoption in secondary prevention of ischemic events.
- Ongoing expansion in emerging markets.
Competitive Landscape
Brilinta faces competition from:
- Clopidogrel (Plavix): cheaper, generic version available since 2012.
- Prasugrel (Effient): similar efficacy with different safety profiles.
- Adenosine diphosphate (ADP) inhibitors: newer agents like cangrelor.
- Anticoagulants: apixaban, rivaroxaban in specific scenarios.
While Brilinta's patent expired in the U.S. in 2025, AstraZeneca has several patents in process to extend market exclusivity through formulation or method-of-use protections.
Regulatory and Patent Outlook
- U.S. Patent Expiry: 2025 (patent protection lapses, enabling generics).
- Worldwide patent challenges are ongoing, with legal disputes in key markets.
- Patent extensions via formulation patents could delay generic entry until 2027.
Price Trajectory and Future Projections
Current Pricing Landscape
- Brilinta's U.S. wholesale acquisition cost (WAC) in 2023 is approximately $420 per month.
- In Europe, prices vary between €250-€350 per month depending on country.
Price Trends (2023-2030)
| Year | Estimated Average Price (U.S.) | Explanation |
|---|---|---|
| 2023 | $420 per month | Stabilized pre-generic entry, with minimal discounts. |
| 2024 | $400 - $440 per month | Slight decrease as generic competition begins in late 2024. |
| 2025 | $200 - $250 per month (post-generic) | Price drop predicted as generics enter the market. |
| 2026-2030 | Stable at $150 - $200 per month | Generic market stabilization, possible price erosion. |
Market Dynamics Impacting Prices
- Generic Entry: The expiration of key patents in 2025 allows generics, leading to price erosion.
- Rebates and Contracts: Payers increasingly use rebates to manage costs, which can effectively lower net prices.
- Regulatory Interventions: Price control policies in Europe and other regions may influence future pricing.
Revenue Projections
| Year | Estimated U.S. Revenue | Assumptions |
|---|---|---|
| 2023 | $2.2 billion | Stable, with current market share and pricing. |
| 2024 | $1.8 - $2.0 billion | Slight decline anticipated, before generic entry. |
| 2025 | $1.0 - $1.2 billion | Sharp decline expected with generic market penetration. |
| 2026+ | $500 million - $800 million | Steady decline, stabilized with generic competition. |
Note: International revenues will mirror price declines and market penetration rates.
Key Factors Influencing Future Market and Pricing
- Therapeutic Positioning: Where Brilinta remains preferred over generics depends on clinician familiarity, brand loyalty, and real-world outcomes.
- Pricing Strategies: AstraZeneca may pursue pricing agreements, rebate strategies, or formulation patents to extend exclusivity.
- Pipeline Developments: An ongoing pipeline of biosimilars or new formulations could influence pricing further.
- Market Penetration in Emerging Economies: Lower-price biosimilars or generics could accelerate access but reduce margins.
Summary
Brilinta’s market faces significant change once patent protections lapse in 2025. Anticipated price drops in the U.S. are substantial, following generic entry, with revised revenue streams reflective of this impact. Strategic moves around patent extensions, biosimilar development, and regional pricing could influence future margins.
Key Takeaways
- Brilinta holds a 20% share of the global antiplatelet market, valued at approximately $11 billion.
- U.S. patent expiry expected in 2025; generic competition will significantly lower prices.
- Current price in the U.S. is around $420/month, projected to decline to $200-$250 post-generic entry.
- Revenue declines are expected from 2025 onward, with U.S. revenue falling below original peaks.
- Regulatory and market strategies will determine how AstraZeneca sustains profitability post-patent.
FAQs
1. When will generic versions of Brilinta become available?
Typically, after patent expiry—expected in 2025—generics are launched within 6-12 months, assuming no litigations delay entry.
2. How does Brilinta compare to clopidogrel in efficacy?
Brilinta has demonstrated superior efficacy in reducing thrombotic events in ACS patients but is associated with a higher bleeding risk and cost.
3. What are the implications of patent expiration for AstraZeneca?
Patent expiration leads to price erosion, loss of exclusivity, and a shift toward biosimilar and generic market segments.
4. How might pricing strategies extend Brilinta’s exclusivity?
Patents on formulations or methods-of-use, alongside legal protections, can delay generic entry and support higher prices temporarily.
5. What role will emerging health technologies play in this market?
Innovations in antiplatelet therapy, personalized medicine, and combination formulations could influence market share and pricing in the future.
Sources:
- IQVIA. "Global Cardiovascular Drugs Market Data," 2023.
- AstraZeneca. "Brilinta (ticagrelor) Medicines," 2023.
- FDA. "Patent and Exclusivity Data," 2023.
- EvaluatePharma. "World Preview 2022, Outlook to 2027."
- US Patent office records.
More… ↓
