33 Drugs Facing NCE-1 / Abbreviated New Drug Application acceptance dates in 2026 - 2027
Loss of Exclusivity / End of Market Exclusivity Period dates
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Friedman, Yali, "33 Drugs Facing NCE-1 / Abbreviated New Drug Application acceptance dates in 2026 - 2027" DrugPatentWatch.com thinkBiotech, 2026 www.drugpatentwatch.com/p/nce-1/.
Media collateral
These NCE-1 dates indicate the first opportunity for generic drug companies to file Abbreviated New Drug Applications (ANDAs) for generic entry into branded drug markets. Generic launch is dependent on many factors, including FDA approval and patents. This information is provided as a rough estimate of generic application, and does not indicate when generics will launch. For more information see the complete DrugPatentWatch database.
When can drug patent challenges be filed against VTAMA?
Generic name: tapinarof
NCE-1 Date: May 2026
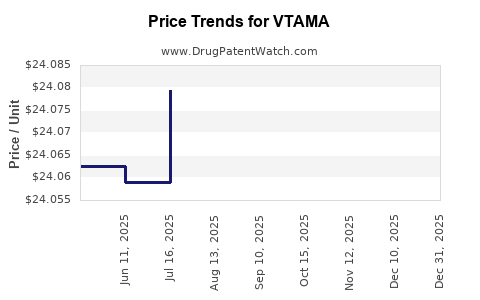
This drug has eighty-one patent family members in thirty-nine countries. There has been litigation on patents covering VTAMA
See drug price trends for VTAMA.
The generic ingredient in VTAMA is tapinarof. Additional details are available on the tapinarof profile page.
When can drug patent challenges be filed against ZTALMY?
Generic name: ganaxolone
NCE-1 Date: June 2026
ZTALMY is a drug marketed by Immedica Pharma. There are eleven patents protecting this drug.
This drug has forty-nine patent family members in sixteen countries. There has been litigation on patents covering ZTALMY
See drug price trends for ZTALMY.
The generic ingredient in ZTALMY is ganaxolone. Additional details are available on the ganaxolone profile page.
When can drug patent challenges be filed against AMVUTTRA?
Generic name: vutrisiran sodium
NCE-1 Date: June 2026
AMVUTTRA is a drug marketed by Alnylam Pharms Inc. There are thirteen patents protecting this drug.
This drug has two hundred and fifty-nine patent family members in forty-nine countries. There has been litigation on patents covering AMVUTTRA
See drug price trends for AMVUTTRA.
The generic ingredient in AMVUTTRA is vutrisiran sodium. Additional details are available on the vutrisiran sodium profile page.
When can drug patent challenges be filed against BLUDIGO?
Generic name: indigotindisulfonate sodium
NCE-1 Date: July 2026
BLUDIGO is a drug marketed by Provepharm Sas. There are three patents protecting this drug.
This drug has two patent family members in two countries.
See drug price trends for BLUDIGO.
The generic ingredient in BLUDIGO is indigotindisulfonate sodium. There are two drug master file entries for this API. Additional details are available on the indigotindisulfonate sodium profile page.
When can drug patent challenges be filed against SOTYKTU?
Generic name: deucravacitinib
NCE-1 Date: September 2026
SOTYKTU is a drug marketed by Bristol. There are four patents protecting this drug.
This drug has one hundred and four patent family members in forty countries.
See drug price trends for SOTYKTU.
The generic ingredient in SOTYKTU is deucravacitinib. Additional details are available on the deucravacitinib profile page.
When can drug patent challenges be filed against TERLIVAZ?
Generic name: terlipressin acetate
NCE-1 Date: September 2026
TERLIVAZ is a drug marketed by Mallinckrodt Ireland. There is one patent protecting this drug.
This drug has thirty-six patent family members in seventeen countries.
See drug price trends for TERLIVAZ.
The generic ingredient in TERLIVAZ is terlipressin acetate. There are two drug master file entries for this API. Additional details are available on the terlipressin acetate profile page.
When can drug patent challenges be filed against ELUCIREM?
Generic name: gadopiclenol
NCE-1 Date: September 2026
ELUCIREM is a drug marketed by Guerbet. There are four patents protecting this drug.
This drug has one hundred and seven patent family members in twenty-eight countries.
See drug price trends for ELUCIREM.
The generic ingredient in ELUCIREM is gadopiclenol. Additional details are available on the gadopiclenol profile page.
When can drug patent challenges be filed against OMLONTI?
Generic name: omidenepag isopropyl
NCE-1 Date: September 2026
OMLONTI is a drug marketed by Ocuvex Therap. There are thirteen patents protecting this drug.
This drug has one hundred and thirty-seven patent family members in thirty-two countries.
The generic ingredient in OMLONTI is omidenepag isopropyl. Additional details are available on the omidenepag isopropyl profile page.
When can drug patent challenges be filed against RELYVRIO?
Generic name: sodium phenylbutyrate; taurursodiol
NCE-1 Date: September 2026
RELYVRIO is a drug marketed by Amylyx. There are five patents protecting this drug.
This drug has sixty-five patent family members in twenty-five countries.
See drug price trends for RELYVRIO.
The generic ingredient in RELYVRIO is sodium phenylbutyrate; taurursodiol. There are one thousand four hundred and seventy-two drug master file entries for this API. Additional details are available on the sodium phenylbutyrate; taurursodiol profile page.
When can drug patent challenges be filed against LYTGOBI?
Generic name: futibatinib
NCE-1 Date: September 2026
LYTGOBI is a drug marketed by Taiho Oncology. There are three patents protecting this drug.
This drug has seventy-nine patent family members in twenty-five countries.
See drug price trends for LYTGOBI.
The generic ingredient in LYTGOBI is futibatinib. Additional details are available on the futibatinib profile page.
When can drug patent challenges be filed against REZLIDHIA?
Generic name: olutasidenib
NCE-1 Date: December 2026
REZLIDHIA is a drug marketed by Rigel Pharms. There are fourteen patents protecting this drug.
This drug has one hundred and ten patent family members in thirty-eight countries. There has been litigation on patents covering REZLIDHIA
See drug price trends for REZLIDHIA.
The generic ingredient in REZLIDHIA is olutasidenib. Additional details are available on the olutasidenib profile page.
When can drug patent challenges be filed against KRAZATI?
Generic name: adagrasib
NCE-1 Date: December 2026
KRAZATI is a drug marketed by Bristol. There are four patents protecting this drug.
This drug has eighty-nine patent family members in thirty-four countries.
See drug price trends for KRAZATI.
The generic ingredient in KRAZATI is adagrasib. Additional details are available on the adagrasib profile page.
When can drug patent challenges be filed against SUNLENCA?
Generic name: lenacapavir sodium
NCE-1 Date: December 2026
SUNLENCA is a drug marketed by Gilead Sciences Inc. There are five patents protecting this drug.
This drug has two hundred and fifteen patent family members in forty-seven countries.
See drug price trends for SUNLENCA.
The generic ingredient in SUNLENCA is lenacapavir sodium. Additional details are available on the lenacapavir sodium profile page.
When can drug patent challenges be filed against YEZTUGO?
Generic name: lenacapavir sodium
NCE-1 Date: December 2026
YEZTUGO is a drug marketed by Gilead Sciences Inc. There are four patents protecting this drug.
This drug has two hundred and five patent family members in forty-seven countries.
The generic ingredient in YEZTUGO is lenacapavir sodium. Additional details are available on the lenacapavir sodium profile page.
When can drug patent challenges be filed against XENOVIEW?
Generic name: xenon xe-129 hyperpolarized
NCE-1 Date: December 2026
XENOVIEW is a drug marketed by Polarean. There are two patents protecting this drug.
This drug has six patent family members in six countries.
The generic ingredient in XENOVIEW is xenon xe-129 hyperpolarized. There are twelve drug master file entries for this API. Additional details are available on the xenon xe-129 hyperpolarized profile page.
When can drug patent challenges be filed against BRENZAVVY?
Generic name: bexagliflozin
NCE-1 Date: January 2027
BRENZAVVY is a drug marketed by Theracosbio. There are six patents protecting this drug.
This drug has seventy-three patent family members in thirty-four countries. There has been litigation on patents covering BRENZAVVY
The generic ingredient in BRENZAVVY is bexagliflozin. Additional details are available on the bexagliflozin profile page.
When can drug patent challenges be filed against JAYPIRCA?
Generic name: pirtobrutinib
NCE-1 Date: January 2027
JAYPIRCA is a drug marketed by Loxo Oncol. There are seven patents protecting this drug.
This drug has one hundred and six patent family members in forty countries.
See drug price trends for JAYPIRCA.
The generic ingredient in JAYPIRCA is pirtobrutinib. Additional details are available on the pirtobrutinib profile page.
When can drug patent challenges be filed against ORSERDU?
Generic name: elacestrant hydrochloride
NCE-1 Date: January 2027
ORSERDU is a drug marketed by Stemline Therap. There are seven patents protecting this drug.
This drug has one hundred and sixty-eight patent family members in thirty countries. There has been litigation on patents covering ORSERDU
See drug price trends for ORSERDU.
The generic ingredient in ORSERDU is elacestrant hydrochloride. Additional details are available on the elacestrant hydrochloride profile page.
When can drug patent challenges be filed against JESDUVROQ?
Generic name: daprodustat
NCE-1 Date: February 2027
JESDUVROQ is a drug marketed by Glaxosmithkline. There are six patents protecting this drug.
This drug has forty-four patent family members in thirty-one countries.
See drug price trends for JESDUVROQ.
The generic ingredient in JESDUVROQ is daprodustat. Additional details are available on the daprodustat profile page.
When can drug patent challenges be filed against FILSPARI?
Generic name: sparsentan
NCE-1 Date: February 2027
FILSPARI is a drug marketed by Travere. There is one patent protecting this drug.
This drug has twenty-nine patent family members in fourteen countries.
See drug price trends for FILSPARI.
The generic ingredient in FILSPARI is sparsentan. Additional details are available on the sparsentan profile page.
When can drug patent challenges be filed against SKYCLARYS?
Generic name: omaveloxolone
NCE-1 Date: February 2027
SKYCLARYS is a drug marketed by Biogen Us. There are seven patents protecting this drug.
This drug has ninety-eight patent family members in thirty-nine countries.
See drug price trends for SKYCLARYS.
The generic ingredient in SKYCLARYS is omaveloxolone. Additional details are available on the omaveloxolone profile page.
When can drug patent challenges be filed against ZAVZPRET?
Generic name: zavegepant hydrochloride
NCE-1 Date: March 2027
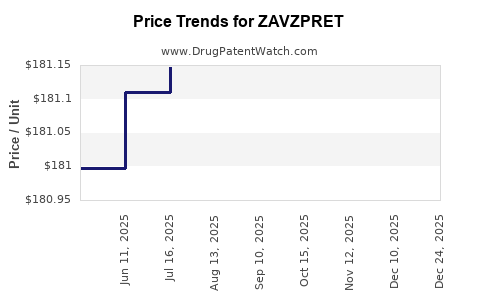
This drug has thirty-six patent family members in twenty-nine countries.
See drug price trends for ZAVZPRET.
The generic ingredient in ZAVZPRET is zavegepant hydrochloride. Additional details are available on the zavegepant hydrochloride profile page.
When can drug patent challenges be filed against DAYBUE?
Generic name: trofinetide
NCE-1 Date: March 2027
DAYBUE is a drug marketed by Acadia Pharms Inc. There are four patents protecting this drug.
This drug has fifty-two patent family members in twenty-six countries.
See drug price trends for DAYBUE.
The generic ingredient in DAYBUE is trofinetide. Additional details are available on the trofinetide profile page.
When can drug patent challenges be filed against DAYBUE STIX?
Generic name: trofinetide
NCE-1 Date: March 2027
DAYBUE STIX is a drug marketed by Acadia Pharms Inc. There are four patents protecting this drug.
This drug has fifty-two patent family members in twenty-six countries.
The generic ingredient in DAYBUE STIX is trofinetide. Additional details are available on the trofinetide profile page.
When can drug patent challenges be filed against REZZAYO?
Generic name: rezafungin acetate
NCE-1 Date: March 2027
REZZAYO is a drug marketed by Mundipharma. There are seven patents protecting this drug.
This drug has one hundred and four patent family members in twenty-seven countries.
See drug price trends for REZZAYO.
The generic ingredient in REZZAYO is rezafungin acetate. Additional details are available on the rezafungin acetate profile page.
When can drug patent challenges be filed against JOENJA?
Generic name: leniolisib phosphate
NCE-1 Date: March 2027
JOENJA is a drug marketed by Pharming. There is one patent protecting this drug.
This drug has forty-six patent family members in forty countries.
See drug price trends for JOENJA.
The generic ingredient in JOENJA is leniolisib phosphate. Additional details are available on the leniolisib phosphate profile page.
When can drug patent challenges be filed against QALSODY?
Generic name: tofersen
NCE-1 Date: April 2027
QALSODY is a drug marketed by Biogen Ma. There are three patents protecting this drug.
This drug has sixty-nine patent family members in twenty-nine countries.
See drug price trends for QALSODY.
The generic ingredient in QALSODY is tofersen. Additional details are available on the tofersen profile page.
When can drug patent challenges be filed against VEOZAH?
Generic name: fezolinetant
NCE-1 Date: May 2027
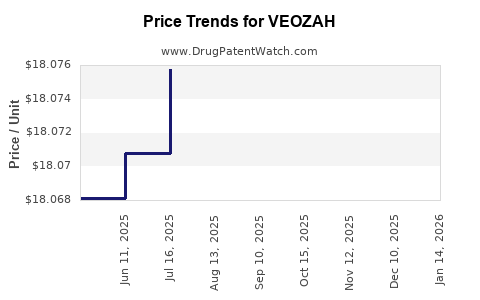
This drug has sixty-nine patent family members in thirty countries.
See drug price trends for VEOZAH.
The generic ingredient in VEOZAH is fezolinetant. Additional details are available on the fezolinetant profile page.
When can drug patent challenges be filed against MIEBO?
Generic name: perfluorohexyloctane
NCE-1 Date: May 2027
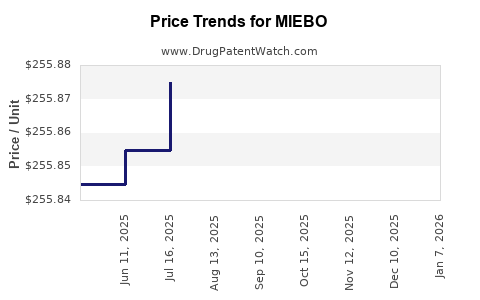
This drug has one hundred and nine patent family members in sixteen countries. There has been litigation on patents covering MIEBO
See drug price trends for MIEBO.
The generic ingredient in MIEBO is perfluorohexyloctane. Additional details are available on the perfluorohexyloctane profile page.
When can drug patent challenges be filed against XACDURO (COPACKAGED)?
Generic name: durlobactam sodium; durlobactam sodium; sulbactam sodium
NCE-1 Date: May 2027
XACDURO (COPACKAGED) is a drug marketed by Entasis Therap. There are four patents protecting this drug.
This drug has eighty patent family members in forty-three countries. There has been litigation on patents covering XACDURO (COPACKAGED)
The generic ingredient in XACDURO (COPACKAGED) is durlobactam sodium; durlobactam sodium; sulbactam sodium. Additional details are available on the durlobactam sodium; durlobactam sodium; sulbactam sodium profile page.
When can drug patent challenges be filed against POSLUMA?
Generic name: flotufolastat f-18 gallium
NCE-1 Date: May 2027
POSLUMA is a drug marketed by Blue Earth. There are six patents protecting this drug.
This drug has ninety patent family members in twenty-one countries.
See drug price trends for POSLUMA.
The generic ingredient in POSLUMA is flotufolastat f-18 gallium. Additional details are available on the flotufolastat f-18 gallium profile page.
When can drug patent challenges be filed against PAXLOVID (COPACKAGED)?
Generic name: nirmatrelvir; ritonavir
NCE-1 Date: May 2027
PAXLOVID (COPACKAGED) is a drug marketed by Pfizer. There are two patents protecting this drug.
This drug has seventy-five patent family members in thirty-eight countries. There has been litigation on patents covering PAXLOVID (COPACKAGED)
The generic ingredient in PAXLOVID (COPACKAGED) is nirmatrelvir; ritonavir. Additional details are available on the nirmatrelvir; ritonavir profile page.
When can drug patent challenges be filed against INPEFA?
Generic name: sotagliflozin
NCE-1 Date: May 2027
This drug has eighty-one patent family members in thirty-three countries.
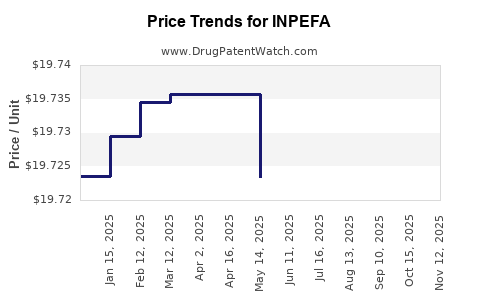
See drug price trends for INPEFA.
The generic ingredient in INPEFA is sotagliflozin. Additional details are available on the sotagliflozin profile page.

