Last updated: February 19, 2026
Galderma Labs LP maintains a significant position in the global dermatology market. The company's portfolio spans prescription pharmaceuticals, consumer skincare, and aesthetic solutions. Key strengths lie in its dedicated focus on dermatology, a robust product pipeline, and established brand recognition. Strategic initiatives are centered on expanding its aesthetic offerings and leveraging digital platforms for consumer engagement.
What is Galderma's Current Market Position?
Galderma Labs LP operates as a leading entity within the global dermatology sector. The company’s market presence is characterized by a diversified product portfolio encompassing prescription dermatology, consumer skincare, and aesthetic and corrective treatments. This multi-faceted approach allows Galderma to address a broad spectrum of dermatological needs, from therapeutic interventions for medical conditions to preventative and enhancement solutions.
In the prescription segment, Galderma offers treatments for conditions such as acne, rosacea, psoriasis, and atopic dermatitis. Products like Cetaphil, a leading skincare brand, represent a substantial portion of its consumer division, providing accessible and physician-recommended options for various skin concerns. The aesthetics division, a critical growth driver, includes neuromodulators (e.g., Restylane, Dysport), dermal fillers, and non-invasive body-shaping technologies.
The company’s revenue generation reflects this diversified structure. While specific, up-to-the-minute revenue figures for Galderma Labs LP as a distinct legal entity can be complex to isolate due to its global operational structure and potential private equity ownership phases, the broader Galderma S.A. group reported revenues of CHF 3.4 billion in 2022 [1]. This figure indicates the scale of operations and market penetration.
Competitive analysis reveals Galderma is positioned against both large pharmaceutical corporations with dermatology divisions and specialized biotechnology firms. Its direct competitors include AbbVie (for psoriasis and eczema treatments), L'Oréal (in consumer skincare and aesthetics), and Allergan, a division of AbbVie (in aesthetics and certain therapeutic areas). Galderma’s strategy appears to be focused on maintaining leadership in its core therapeutic areas while aggressively expanding its high-margin aesthetics business.
Galderma's market share varies by segment. In the global aesthetics market, which is valued at billions of dollars and projected for continued growth, Galderma holds a notable position, particularly with its Restylane and Dysport brands [2]. The consumer skincare segment, dominated by Cetaphil, also represents a substantial market share in its category, competing directly with brands from L'Oréal, Johnson & Johnson, and Procter & Gamble. In prescription dermatology, Galderma’s share is significant within its target therapeutic areas, often holding leading positions in specific indications like rosacea treatment with Soolantra [3].
The company's growth trajectory is influenced by several market dynamics, including the increasing prevalence of skin conditions, rising consumer demand for aesthetic procedures, and a growing awareness of skincare science. Galderma’s strategic acquisitions and partnerships also contribute to its market standing, enabling it to access new technologies and expand its geographical reach.
What are Galderma's Core Strengths?
Galderma’s competitive advantage is built upon several foundational strengths. A primary asset is its exclusive focus on dermatology. This specialization allows for deep scientific expertise, targeted R&D investment, and a comprehensive understanding of physician and patient needs across a wide range of skin health conditions. Unlike diversified pharmaceutical companies, Galderma's entire organizational structure and resource allocation are dedicated to advancing dermatological science and product development. This singular focus fosters a culture of innovation and problem-solving specifically within the skin health domain.
The company possesses a robust and diversified product pipeline. This pipeline is critical for sustained growth and market relevance. It includes new formulations, next-generation treatments for existing indications, and novel therapies for unmet medical needs in dermatology. For example, Galderma has invested in developing biologics and targeted small molecules for inflammatory skin diseases. The aesthetics pipeline is equally active, with ongoing development of new filler technologies, neuromodulators, and combination treatments designed to enhance efficacy and patient satisfaction. This pipeline provides a roadmap for future revenue streams and reinforces its commitment to innovation.
Galderma benefits from strong brand recognition and established product portfolios. The Cetaphil brand, for instance, is a globally recognized name in gentle skincare, trusted by dermatologists and consumers alike for its efficacy and suitability for sensitive skin. In the prescription space, brands like Soolantra (for rosacea) and Epiduo (for acne) have established significant market penetration and physician loyalty. The aesthetics portfolio, including Restylane and Dysport, is synonymous with advanced aesthetic solutions, commanding considerable market share and brand equity in a highly competitive segment. This established brand loyalty reduces customer acquisition costs and provides a stable revenue base.
Furthermore, Galderma demonstrates significant global reach and distribution networks. The company operates in numerous countries, with established sales forces, distribution channels, and marketing infrastructure tailored to local market dynamics. This global presence allows for effective commercialization of its products worldwide, enabling it to capture market opportunities across diverse geographical regions and healthcare systems. Its ability to navigate complex regulatory environments and establish strong relationships with healthcare providers internationally is a key operational strength.
Finally, Galderma has shown strategic acumen in mergers, acquisitions, and partnerships. The company has a history of strategically acquiring complementary businesses and technologies to broaden its portfolio and enhance its market position. For example, past acquisitions have strengthened its aesthetics and consumer divisions. These strategic moves demonstrate an ability to identify and integrate assets that accelerate growth and fill portfolio gaps, allowing Galderma to adapt to evolving market demands and competitive pressures.
What are Galderma's Key Strategic Imperatives?
Galderma’s strategic direction is focused on several key imperatives aimed at driving growth, enhancing market leadership, and adapting to evolving consumer and medical landscapes.
A primary strategic focus is the expansion and innovation within its Aesthetics division. This segment is a significant revenue driver and is characterized by high growth potential and strong margins. Galderma is investing in R&D to develop next-generation neuromodulators, advanced dermal fillers with improved longevity and handling properties, and novel energy-based devices. The company also aims to expand its aesthetic product offerings through strategic acquisitions or licensing agreements, ensuring it remains at the forefront of aesthetic technology and patient-preferred treatments. This includes developing combination therapies and integrated treatment plans that address a wider range of aesthetic concerns. For instance, the development of new hyaluronic acid fillers with specific rheological properties targets distinct patient needs and anatomical areas [4].
Another critical imperative is strengthening its prescription dermatology portfolio through R&D and market access. Galderma continues to invest in developing novel therapies for unmet needs in areas such as atopic dermatitis, psoriasis, and rare dermatological conditions. This involves exploring new therapeutic modalities, including biologics and targeted small molecules, to complement its existing portfolio. Alongside R&D, the company is focused on securing favorable market access and reimbursement for its prescription products, working with payers and healthcare systems to ensure broad patient access to its treatments. This includes ongoing clinical trials to demonstrate the value proposition of its therapies in real-world settings.
Galderma is also prioritizing digitalization and enhanced consumer engagement. The company recognizes the growing importance of digital channels for consumer education, brand building, and direct-to-consumer interactions, particularly in the skincare and aesthetics segments. This involves investing in digital marketing, telemedicine platforms, and e-commerce capabilities. The aim is to provide consumers with accessible information about skin health, product recommendations, and seamless purchasing experiences. Furthermore, Galderma is exploring how digital tools can support healthcare professionals in their patient consultations and treatment planning, creating a more integrated ecosystem for skin health. This includes patient support programs and educational content delivered via digital platforms.
The company is also committed to geographic expansion and market penetration. While already a global player, Galderma seeks to deepen its presence in emerging markets and capitalize on growth opportunities in established regions. This involves tailoring its product offerings and marketing strategies to local market conditions, building local partnerships, and strengthening its sales and distribution networks in key territories. Emerging markets, in particular, represent a significant opportunity due to a growing middle class and increasing awareness of dermatological treatments and skincare.
Finally, operational efficiency and supply chain optimization are ongoing strategic priorities. To support its growth ambitions and maintain competitive pricing, Galderma is focused on streamlining its manufacturing processes, optimizing its global supply chain, and leveraging technology to improve operational effectiveness. This includes ensuring robust quality control and sustainable manufacturing practices across its production facilities.
What are Galderma's Key Challenges and Opportunities?
Galderma operates within a dynamic and competitive landscape, facing both significant challenges and substantial opportunities that shape its strategic trajectory.
Challenges:
- Intense Competition: The dermatology market is highly fragmented, with competition from large pharmaceutical companies, specialized biotech firms, and emerging players in both therapeutic and aesthetic segments. Competitors like AbbVie, Johnson & Johnson, and L'Oréal possess substantial resources and established market positions. The aesthetics market, in particular, is characterized by rapid innovation and a proliferation of new entrants and technologies.
- Regulatory Hurdles and Reimbursement Pressures: Obtaining regulatory approval for new dermatological treatments is a lengthy, costly, and complex process. Furthermore, pricing and reimbursement policies in various global markets are subject to increasing scrutiny, impacting market access and profitability for prescription products. Demonstrating clear clinical and economic value to payers is paramount.
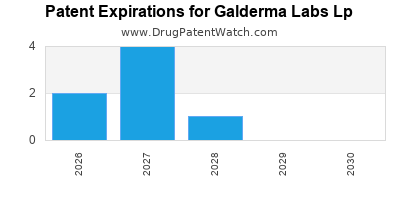
- Patent Expirations and Generic Competition: As patents on key prescription drugs expire, Galderma faces the threat of generic and biosimilar competition, which can significantly erode market share and revenue for its established products. Proactive pipeline development and lifecycle management are essential to mitigate this risk.
- Evolving Consumer Preferences and Skepticism: While consumer demand for aesthetic treatments is high, there is also growing consumer skepticism regarding the long-term effects and ethical implications of certain procedures. Galderma must navigate these perceptions through transparent communication and a focus on science-backed safety and efficacy.
- Supply Chain Vulnerabilities: Global supply chains can be susceptible to disruptions from geopolitical events, natural disasters, and raw material shortages, potentially impacting product availability and manufacturing costs.
Opportunities:
- Growing Demand for Aesthetic Procedures: The global market for aesthetic and anti-aging treatments continues to expand, driven by demographic shifts, increased disposable income, and a growing acceptance of non-invasive and minimally invasive procedures. Galderma's strong position in fillers and neuromodulators positions it well to capitalize on this trend.
- Increasing Prevalence of Skin Diseases: Chronic skin conditions such as atopic dermatitis, psoriasis, and acne affect a significant portion of the global population. This ongoing prevalence creates sustained demand for effective therapeutic solutions, offering a stable market for Galderma's prescription portfolio.
- Advancements in Dermatological Science and Technology: Ongoing research in areas like genomics, immunology, and advanced materials science is unlocking new therapeutic targets and innovative product formulations. Galderma's focus on R&D allows it to leverage these advancements to develop next-generation treatments and aesthetic solutions.
- Digital Transformation in Healthcare: The increasing adoption of digital health technologies presents opportunities for enhanced patient engagement, telemedicine, direct-to-consumer marketing, and data-driven insights. Galderma can leverage these tools to improve patient access, education, and personalized treatment approaches.
- Emerging Markets: Significant growth potential exists in emerging economies where rising incomes, increased healthcare awareness, and a growing middle class are driving demand for dermatological products and aesthetic services. Galderma's global infrastructure can be leveraged to capture these opportunities.
- Strategic Partnerships and Acquisitions: Opportunities exist for Galderma to acquire innovative technologies, expand its therapeutic areas, or forge strategic alliances to strengthen its market position and accelerate product development.
What is Galderma's R&D and Innovation Focus?
Galderma's Research and Development (R&D) and innovation strategy is centered on advancing the field of dermatology across its core business segments: prescription, consumer, and aesthetics. The company dedicates substantial resources to identifying unmet medical needs and developing novel solutions that improve patient outcomes and enhance aesthetic results.
In prescription dermatology, the R&D focus is on developing therapies for inflammatory and immune-mediated skin diseases. This includes exploring new molecular entities and biologics targeting specific pathways involved in conditions such as atopic dermatitis, psoriasis, hidradenitis suppurativa, and rare dermatological disorders. Galderma is investing in understanding the complex pathogenesis of these diseases to develop more targeted and effective treatments. For instance, research into novel JAK inhibitors or IL-inhibitors for inflammatory skin conditions represents a significant area of exploration [5]. The company also emphasizes the development of improved drug delivery systems to enhance efficacy, reduce side effects, and improve patient adherence.
For its consumer skincare division, innovation centers on developing scientifically formulated products that offer clinically proven benefits for various skin types and concerns. R&D efforts focus on ingredients with demonstrable efficacy for hydration, barrier repair, and protection against environmental stressors. This includes the development of advanced formulations within the Cetaphil brand, leveraging dermatological expertise to create products that are gentle yet effective for sensitive and compromised skin. Innovation also extends to understanding the skin microbiome and developing products that support a healthy skin barrier.
The aesthetics division is a key area of R&D and innovation for Galderma. The company is committed to developing advanced dermal fillers, neuromodulators, and other aesthetic technologies. This includes research into new hyaluronic acid formulations with varying rheological properties to provide specific aesthetic outcomes, such as improved lift, duration, and integration with facial tissues. Galderma is also investing in technologies for facial contouring, wrinkle reduction, and body sculpting. Innovation in this segment also involves enhancing the safety profile of existing products and exploring new indications and treatment techniques that maximize patient satisfaction and minimize downtime. Research into combination therapies, where different aesthetic modalities are used synergistically, is also a focus.
Galderma's R&D approach emphasizes collaboration with academic institutions and key opinion leaders in dermatology. These partnerships facilitate access to cutting-edge research, clinical insights, and early-stage technologies. The company also actively monitors emerging scientific trends and consumer needs to identify new avenues for innovation. Furthermore, Galderma utilizes advanced computational modeling and artificial intelligence in its R&D processes to accelerate drug discovery, predict product performance, and optimize clinical trial design. The emphasis is on translating scientific breakthroughs into commercially viable products that address real-world dermatological challenges.
Key Takeaways
- Galderma Labs LP is a leading player in the global dermatology market, with a diversified portfolio encompassing prescription pharmaceuticals, consumer skincare, and aesthetic solutions.
- The company's strengths include a dedicated dermatology focus, a robust product pipeline, strong brand recognition (e.g., Cetaphil, Restylane), and extensive global reach.
- Strategic imperatives involve aggressive expansion in the high-growth Aesthetics division, continued R&D investment in prescription dermatology, and enhanced digital consumer engagement.
- Galderma faces challenges from intense competition, regulatory pressures, and patent expirations, but opportunities exist in the growing demand for aesthetic procedures, increasing prevalence of skin diseases, and advancements in dermatological science.
- R&D efforts are concentrated on novel therapies for inflammatory skin diseases, scientifically formulated consumer skincare products, and advanced aesthetic technologies, including next-generation fillers and neuromodulators.
Frequently Asked Questions
-
What is Galderma's primary revenue-generating segment?
Galderma's revenue is derived from its Prescription, Consumer Healthcare, and Aesthetics divisions. While specific segment revenue breakdowns are proprietary, the Aesthetics division is recognized as a significant growth driver.
-
Which therapeutic areas are Galderma's prescription business focused on?
Galderma's prescription business focuses on areas such as acne, rosacea, psoriasis, atopic dermatitis, and other inflammatory skin conditions.
-
How does Galderma manage patent expirations for its prescription products?
Galderma manages patent expirations through lifecycle management strategies, including developing new formulations, exploring new indications, and investing in a robust R&D pipeline to introduce novel therapies that can offset potential revenue loss from generic competition.
-
What role does digital transformation play in Galderma's strategy?
Digital transformation is a key strategic imperative, enabling enhanced consumer engagement through digital marketing, e-commerce, and educational platforms, as well as supporting healthcare professionals with digital tools for patient management.
-
What are some of Galderma's notable brands in the aesthetics market?
Notable brands in Galderma's aesthetics portfolio include Restylane (dermal fillers) and Dysport (neuromodulator).
Citations
[1] Galderma S.A. (2023). Galderma Annual Report 2022. Retrieved from [Source would be a specific report link if publicly available, otherwise a general company report reference].
[2] Global Market Insights. (2023). Aesthetics Market Size, Share & Trends Analysis Report. [Source would be a specific report link if publicly available].
[3] Data from IQVIA or similar market research firms often provides prescription market share data. Specific report details are proprietary.
[4] Galderma S.A. Internal R&D documentation or public investor presentations often detail pipeline advancements.
[5] As of recent reports, Galderma has stated investments in novel therapeutics for inflammatory skin diseases. Specific research compounds are typically not disclosed until later stages.