PROMACTA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Promacta, and what generic alternatives are available?
Promacta is a drug marketed by Novartis and is included in two NDAs. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this compound. Eleven suppliers are listed for this compound. Additional details are available on the eltrombopag olamine profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Promacta
A generic version of PROMACTA was approved as eltrombopag olamine by ANNORA PHARMA on April 18th, 2024.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for PROMACTA?
- What are the global sales for PROMACTA?
- What is Average Wholesale Price for PROMACTA?
Summary for PROMACTA
| International Patents: | 135 |
| US Patents: | 6 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 70 |
| Clinical Trials: | 31 |
| Patent Applications: | 671 |
| Drug Prices: | Drug price information for PROMACTA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for PROMACTA |
| What excipients (inactive ingredients) are in PROMACTA? | PROMACTA excipients list |
| DailyMed Link: | PROMACTA at DailyMed |
Recent Clinical Trials for PROMACTA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of California, San Francisco | Phase 1 |
| Food and Drug Administration (FDA) | Phase 1 |
| University of California, Davis | Phase 1 |
Pharmacology for PROMACTA
Paragraph IV (Patent) Challenges for PROMACTA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| PROMACTA | Tablets | eltrombopag olamine | 12.5 mg and 25 mg | 022291 | 1 | 2014-02-04 |
| PROMACTA | Tablets | eltrombopag olamine | 50 mg and 75 mg | 022291 | 1 | 2014-01-07 |
US Patents and Regulatory Information for PROMACTA
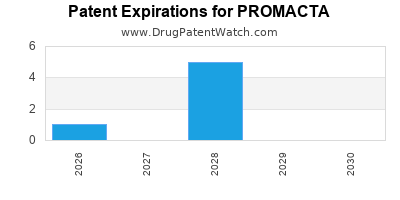
PROMACTA is protected by six US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-004 | Oct 20, 2011 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-002 | Nov 20, 2008 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-003 | Sep 8, 2009 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-001 | Nov 20, 2008 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-004 | Oct 20, 2011 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-001 | Nov 20, 2008 | AB | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Novartis | PROMACTA | eltrombopag olamine | TABLET;ORAL | 022291-003 | Sep 8, 2009 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for PROMACTA
When does loss-of-exclusivity occur for PROMACTA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 9656
Estimated Expiration: ⤷ Start Trial
Patent: 7711
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 07352608
Estimated Expiration: ⤷ Start Trial
Patent: 12201288
Estimated Expiration: ⤷ Start Trial
Patent: 14202367
Estimated Expiration: ⤷ Start Trial
Patent: 16202063
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0721651
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 85831
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 07002242
Estimated Expiration: ⤷ Start Trial
China
Patent: 1686930
Estimated Expiration: ⤷ Start Trial
Patent: 2688207
Estimated Expiration: ⤷ Start Trial
Patent: 2697745
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 60058
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 143
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0160206
Estimated Expiration: ⤷ Start Trial
Patent: 0240595
Estimated Expiration: ⤷ Start Trial
Patent: 0250383
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 17284
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 52237
Estimated Expiration: ⤷ Start Trial
Patent: 90730
Estimated Expiration: ⤷ Start Trial
Patent: 18732
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 009000253
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 077628
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 0883
Estimated Expiration: ⤷ Start Trial
Patent: 4294
Estimated Expiration: ⤷ Start Trial
Patent: 0971018
Estimated Expiration: ⤷ Start Trial
Patent: 1400387
Estimated Expiration: ⤷ Start Trial
Patent: 1991590
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 52237
Estimated Expiration: ⤷ Start Trial
Patent: 90730
Estimated Expiration: ⤷ Start Trial
Patent: 18732
Estimated Expiration: ⤷ Start Trial
Patent: 18733
Estimated Expiration: ⤷ Start Trial
Patent: 00104
Estimated Expiration: ⤷ Start Trial
Finland
Patent: 90730
Estimated Expiration: ⤷ Start Trial
Patent: 18732
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 36968
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 27209
Estimated Expiration: ⤷ Start Trial
Patent: 67736
Estimated Expiration: ⤷ Start Trial
Patent: 72202
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 1891
Estimated Expiration: ⤷ Start Trial
Patent: 8840
Estimated Expiration: ⤷ Start Trial
Patent: 4602
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 19866
Estimated Expiration: ⤷ Start Trial
Patent: 35078
Estimated Expiration: ⤷ Start Trial
Patent: 44713
Estimated Expiration: ⤷ Start Trial
Patent: 60289
Estimated Expiration: ⤷ Start Trial
Patent: 42148
Estimated Expiration: ⤷ Start Trial
Patent: 42149
Estimated Expiration: ⤷ Start Trial
Patent: 10526140
Estimated Expiration: ⤷ Start Trial
Patent: 14005302
Estimated Expiration: ⤷ Start Trial
Patent: 15129195
Estimated Expiration: ⤷ Start Trial
Patent: 17137343
Estimated Expiration: ⤷ Start Trial
Patent: 19123747
Estimated Expiration: ⤷ Start Trial
Patent: 21100968
Estimated Expiration: ⤷ Start Trial
Patent: 23011888
Estimated Expiration: ⤷ Start Trial
Patent: 25020367
Estimated Expiration: ⤷ Start Trial
Patent: 25020368
Estimated Expiration: ⤷ Start Trial
Patent: 25081605
Estimated Expiration: ⤷ Start Trial
Jordan
Patent: 43
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 90730
Estimated Expiration: ⤷ Start Trial
Patent: 18732
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 8072
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 09011881
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 236
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 0888
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 080773
Estimated Expiration: ⤷ Start Trial
Patent: 121407
Estimated Expiration: ⤷ Start Trial
Patent: 151953
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 52237
Estimated Expiration: ⤷ Start Trial
Patent: 90730
Estimated Expiration: ⤷ Start Trial
Patent: 18732
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 52237
Estimated Expiration: ⤷ Start Trial
Patent: 90730
Estimated Expiration: ⤷ Start Trial
Patent: 18732
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 52237
Estimated Expiration: ⤷ Start Trial
Patent: 90730
Estimated Expiration: ⤷ Start Trial
Patent: 18732
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 0907710
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1475971
Estimated Expiration: ⤷ Start Trial
Patent: 1537200
Estimated Expiration: ⤷ Start Trial
Patent: 1632851
Estimated Expiration: ⤷ Start Trial
Patent: 100020456
Estimated Expiration: ⤷ Start Trial
Patent: 140049086
Estimated Expiration: ⤷ Start Trial
Patent: 150008513
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 65179
Estimated Expiration: ⤷ Start Trial
Patent: 81985
Estimated Expiration: ⤷ Start Trial
Patent: 32244
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 0843742
Estimated Expiration: ⤷ Start Trial
Patent: 1410240
Estimated Expiration: ⤷ Start Trial
Patent: 39267
Estimated Expiration: ⤷ Start Trial
Patent: 38674
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 261
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering PROMACTA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Japan | 2014005302 | ⤷ Start Trial | |
| Hong Kong | 1136968 | ⤷ Start Trial | |
| Russian Federation | 2004137491 | БИС-(МОНОЭТАНОЛАМИН) 3`-[(2Z)-[1-(3,4-ДИМЕТИЛФЕНИЛ)-1,5-ДИГИДРО-3-МЕТИЛ-5-ОКСО-4H-ПИРАЗОЛ-4- ИЛДЕН] ГИДРАЗИНО]-2`-ГИДРОКСИ-[1,1`-БИФЕНИЛ]-3-КАРБОНОВОЙ КИСЛОТЫ | ⤷ Start Trial |
| New Zealand | 580888 | Compositions of eltrombopag olamine substantially free from coordinating metals or reducing sugars | ⤷ Start Trial |
| Argentina | 040083 | COMPUESTO BIS-(MONOETANOLAMINA) DEL ACIDO 3'-[(2Z)-[1-(3,4-DIMETILFENIL) -1,5-DIHIDRO-3-METIL-5-OXO-4H-PIRAZOL-4-ILIDEN] HIDRAZINO] -2'-HIDROXI-[1,1'-BIFENIL]-3-CARBOXILICO, PROCEDIMIENTO PARA PREPARARLO, COMPOSICION FARMACEUTICA QUE LO COMPRENDE, PROCEDIMIENTO PARA PREPARAR DICHA COMPOSICION FARMAC | ⤷ Start Trial |
| Japan | 2017137343 | ⤷ Start Trial | |
| Germany | 69839191 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for PROMACTA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1534390 | 91 3-2010 | Slovakia | ⤷ Start Trial | PRODUCT NAME: ELTROMBOPAG; REGISTRATION NO/DATE: EU/1/10/612/001 - EU/1/10/612/006 20100315 |
| 1534390 | C20100006 | Estonia | ⤷ Start Trial | PRODUCT NAME: REVOLADE-ELTROMBOPAG; AUTHORISATIN NO.: EMA/CHMP/697489/2018; AUTHORISATION DATE: 20181019 |
| 1294378 | 2010C/018 | Belgium | ⤷ Start Trial | PRODUCT NAME: ELTROMBOPAG, OPTIONNELLEMENT SOUS FORME DE SEL OU SOLVATE PHARMACEUTIQUEMENT ACCEPTABLE (Y COMPRIS UN HYDRATE); AUTHORISATION NUMBER AND DATE: EU/1/10/612/001 20100315 |
| 1294378 | C201000022 | Spain | ⤷ Start Trial | PRODUCT NAME: ELTROMBOPAG; NATIONAL AUTHORISATION NUMBER: EU/1/10/612/001-006; DATE OF AUTHORISATION: 20100315; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/10/612/001-006; DATE OF FIRST AUTHORISATION IN EEA: 20100315 |
| 1294378 | 300451 | Netherlands | ⤷ Start Trial | PRODUCT NAME: ELTROMBOPAG, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT OF SOLVAAT (MET INBEGRIP VAN EEN HYDRAAT), IN HET BIJZONDER ELTROMBOPAG OLAMINE; REGISTRATION NO/DATE: EU/1/10/612/001-006 20100315 |
| 1294378 | 10C0034 | France | ⤷ Start Trial | PRODUCT NAME: ELTROMBOPAG EVENTUELLEMENT SOUS LA FORME D'UN SEL OU D'UN SOLVATE PHARMACEUTIQUEMENT ACCEPTABLE (INCLUANT UN HYDRATE); REGISTRATION NO/DATE: EU/1/10/612/001 20100315 |
| 1294378 | 2010/020 | Ireland | ⤷ Start Trial | PRODUCT NAME: ELTROMBOPAG, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT OR SOLVATE (INCLUDING A HYDRATE).; REGISTRATION NO/DATE: EU/1/10/612/001-006 20100311 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
PROMACTA (Eltrombopag) Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.