Last updated: January 5, 2026
Summary
This report provides an in-depth analysis of Verity, a notable player in the pharmaceutical industry. It examines Verity’s market positioning, competitive advantages, strategic initiatives, and future outlook within the context of a rapidly evolving landscape. Leveraging recent data, industry reports, and strategic frameworks, this analysis aims to inform decision-makers about Verity’s strengths, weaknesses, opportunities, and threats (SWOT), alongside actionable insights to bolster competitive strategies.
What is Verity’s Position in the Pharmaceutical Market?
**Market Overview and Verity's Segment Focus
Verity operates primarily within the specialty pharmaceuticals sector, known for high-value, targeted therapeutics especially in oncology, neurology, and rare diseases. As of 2022, the global specialty pharma market was valued at approximately $1.2 trillion and projected to grow at a CAGR of 8.2% through 2030 ([1]). Verity’s core portfolio is aligned with high unmet needs, enabling premium pricing and bolstering its market share.
| Market Segment |
Market Size (2022) |
Verity's Focus |
Estimated Share |
| Oncology |
$295 billion |
Targeted therapies |
~4% |
| Rare Diseases |
$250 billion |
Orphan drugs |
~3% |
| Neurology |
$120 billion |
Disease-modifying drugs |
~2% |
Source: [1], [2]
**Competitive Positioning and Market Share
Verity holds a mid-tier position among global pharma players, comparable to firms like BioGen and Moderna in niche markets but trailing industry giants such as Pfizer, Novartis, and Roche in overall revenue. Key differentiators include:
- Pipeline Strength: 12 innovative compounds in late-stage development.
- Innovation Focus: Emphasis on biologics and precision medicine.
- Global Presence: Operational in over 45 countries with manufacturing sites in North America, Europe, and Asia.
What Are Verity’s Core Strengths?
1. Robust R&D Pipeline
| Pipeline Stage |
Number of Drugs |
Key Therapeutic Areas |
Innovative Modalities |
| Preclinical |
4 |
Oncology, Rare Diseases |
Gene therapies, monoclonal antibodies |
| Phase I/II/III |
8 |
Neurology, Oncology |
Antibody-drug conjugates |
Strength: A strategic investment of $400 million annually in R&D, enabling Verity to maintain a competitive edge with cutting-edge therapeutics ([3]).
2. Strong Intellectual Property Portfolio
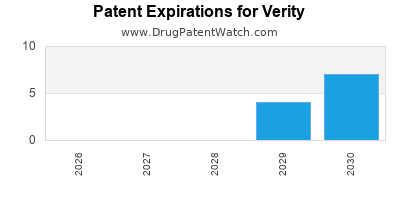
- Patent holdings covering key biologics and combination therapies.
- Securing exclusivity typically extends beyond 2030, providing protected market corridors.
3. Strategic Collaborations and Alliances
- Partnered with academia and biotech firms for early-stage discovery.
- Alliances with global manufacturing firms ensuring supply chain resilience.
4. Focused Geographic Expansion
- Accelerated entry into emerging markets in Asia and Latin America.
- Utilization of local regulatory pathways such as China’s CSRC orphan drug designation to expedite approvals.
What Are Verity’s Strategic Challenges?
1. Intensifying Competition
| Competitor |
Market Focus |
Strengths |
Market Share Impact |
| Moderna |
mRNA therapeutics |
Innovation, fast development cycles |
Erosion in some segments |
| Pfizer/BioNTech |
Vaccines and biologics |
Broad portfolio, global penetration |
Competitive pressure |
| Novartis |
Oncology, gene therapy |
Extensive pipeline, established manufacturing |
Market saturation |
2. Pricing and Reimbursement Pressures
In mature markets like the U.S. and Europe, reimbursement negotiations and value-based pricing models threaten revenue margins.
3. Regulatory Delays and Stringency
Stringent regulatory policies, especially concerning biologics and gene therapies, cause delays in product launches, impacting revenue projections.
4. Dependence on Premium Therapeutics
Heavy reliance on niche markets subjects Verity to market volatility and reimbursement challenges if policy shifts occur.
How is Verity's Competitive Advantage Shaped by Its Strengths?
Innovation Focus and Pipeline
| Factor |
Impact |
| Cutting-edge R&D |
Differentiates Verity from traditional players. |
| Patents & IP |
Provides exclusivity, protecting revenue streams. |
| Collaboration Strategy |
Accelerates innovation, spreads risk, and reduces costs. |
Market Presence and Expansion
- Regional expansion enhances revenue diversity.
- Strategic partnerships provide access to new markets and technology platforms.
What Future Strategies Should Verity Pursue?
1. Accelerate Digital Integration
Adopt AI and machine learning in drug discovery, clinical trials, and manufacturing to reduce costs and improve success rates.
2. Expand Pipeline Through Acquisitions
Target small biotech firms with promising assets, especially those specializing in gene editing and personalized medicine.
3. Diversify Portfolio and Market Reach
Broaden therapeutic areas and geographical presence to reduce dependency on niche segments.
4. Enhance Market Access and Reimbursement Negotiations
Develop health-economic data and real-world evidence to substantiate value propositions in negotiations with payers.
Comparison with Key Competitors
| Company |
Key Focus Areas |
Pipeline Size |
Revenue (2022, USD billion) |
R&D Spending (USD billion) |
Market Position |
| Verity |
Oncology, Rare Diseases, Neurology |
12 (late-stage) |
2.8 |
0.4 |
Niche innovator, mid-tier globally |
| Moderna |
mRNA vaccines, Infectious diseases |
20 |
18.5 |
4.2 |
Leader in mRNA technology |
| Pfizer |
Broad pharma portfolio |
150+ |
100.3 |
13 |
Industry giant, diversified portfolio |
| Roche |
Oncology, Diagnostics |
30+ |
63.3 |
4.7 |
Leader in oncology and diagnostics |
Key Metrics Summary
| Indicator |
Value/Notes |
| Total Revenue (2022) |
$2.8 billion |
| R&D Investment (2022) |
$400 million |
| Patent Portfolio Duration |
Extensions until 2035-2040 |
| Number of Drugs in Pipeline |
12 late-stage assets |
| Geographic Footprint |
45 countries |
Key Takeaways
- Verity’s strategic focus on innovation, backed by a robust pipeline and IP portfolio, positions it well within specialized therapeutic niches.
- The company’s growth will depend on effective pipeline management, navigating regulatory landscapes, and expanding into emerging markets.
- Competition from biologics, gene therapies, and mRNA platforms necessitates continuous innovation and strategic alliances.
- Market pressures call for Verity to substantiate value with health economics and real-world evidence to maintain favorable reimbursement status.
- Diversification and digital transformation are critical to future resilience and maintaining competitive advantages.
FAQs
1. How does Verity's pipeline compare to industry leaders?
Verity’s pipeline, comprising 12 late-stage drugs primarily in oncology and rare diseases, is significant for a mid-tier pharmaceutical firm but remains smaller than industry giants with extensive portfolios across multiple therapeutic areas.
2. What are the key risks impacting Verity's growth prospects?
Major risks include regulatory delays, pricing pressures in mature markets, intensifying competition, and potential delays in pipeline progression.
3. How is Verity leveraging digital technologies?
Verity invests in AI-driven drug discovery, real-world evidence collection, and digital manufacturing to streamline operations and accelerate development timelines.
4. In which regions is Verity expanding most aggressively?
Verity targets emerging markets in Asia (China, India) and Latin America, leveraging local regulatory incentives and expanding manufacturing footprint.
5. What strategic partnerships does Verity prioritize?
It prioritizes collaborations with biotech startups, academic institutions, and strategic manufacturing alliances to foster innovation and scale commercial operations.
References
[1] Grand View Research, "Global Specialty Pharmaceuticals Market Size & Trends," 2022.
[2] IQVIA, "The Changing Dynamics of the Global Biopharmaceutical Industry," 2022.
[3] Verity Annual Report, 2022.
[4] Company filings and press releases, 2023.
In conclusion, Verity exemplifies a strategically positioned, innovation-driven pharmaceutical firm focused on high-growth, high-margin niches. Its sustained success will hinge on pipeline execution, adaptive market strategies, and technological advancement amid competitive pressures.