TEKTURNA HCT Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Tekturna Hct, and what generic alternatives are available?
Tekturna Hct is a drug marketed by Noden Pharma and is included in one NDA. There is one patent protecting this drug and one Paragraph IV challenge.
This drug has thirty-two patent family members in twenty-five countries.
The generic ingredient in TEKTURNA HCT is aliskiren hemifumarate; hydrochlorothiazide. There are four drug master file entries for this compound. Additional details are available on the aliskiren hemifumarate; hydrochlorothiazide profile page.
DrugPatentWatch® Generic Entry Outlook for Tekturna Hct
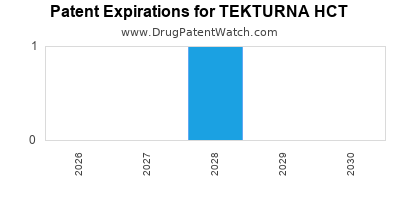
Tekturna Hct was eligible for patent challenges on March 5, 2011.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be July 13, 2028. This may change due to patent challenges or generic licensing.
There has been one patent litigation case involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for TEKTURNA HCT?
- What are the global sales for TEKTURNA HCT?
- What is Average Wholesale Price for TEKTURNA HCT?
Summary for TEKTURNA HCT
| International Patents: | 32 |
| US Patents: | 1 |
| Applicants: | 1 |
| NDAs: | 1 |
| Clinical Trials: | 18 |
| Drug Prices: | Drug price information for TEKTURNA HCT |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for TEKTURNA HCT |
| What excipients (inactive ingredients) are in TEKTURNA HCT? | TEKTURNA HCT excipients list |
| DailyMed Link: | TEKTURNA HCT at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for TEKTURNA HCT
Generic Entry Date for TEKTURNA HCT*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for TEKTURNA HCT
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Wayne State University | Phase 4 |
| University of Alabama at Birmingham | Phase 4 |
| Mayo Clinic | N/A |
Paragraph IV (Patent) Challenges for TEKTURNA HCT
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| TEKTURNA HCT | Tablets | aliskiren hemifumarate; hydrochlorothiazide | 150 mg/12.5 mg 150 mg/25 mg 300 mg/12.5 mg 300 mg/25 mg | 022107 | 1 | 2014-03-07 |
US Patents and Regulatory Information for TEKTURNA HCT
TEKTURNA HCT is protected by one US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of TEKTURNA HCT is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Noden Pharma | TEKTURNA HCT | aliskiren hemifumarate; hydrochlorothiazide | TABLET;ORAL | 022107-001 | Jan 18, 2008 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Noden Pharma | TEKTURNA HCT | aliskiren hemifumarate; hydrochlorothiazide | TABLET;ORAL | 022107-004 | Jan 18, 2008 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Noden Pharma | TEKTURNA HCT | aliskiren hemifumarate; hydrochlorothiazide | TABLET;ORAL | 022107-002 | Jan 18, 2008 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for TEKTURNA HCT
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Noden Pharma | TEKTURNA HCT | aliskiren hemifumarate; hydrochlorothiazide | TABLET;ORAL | 022107-003 | Jan 18, 2008 | ⤷ Start Trial | ⤷ Start Trial |
| Noden Pharma | TEKTURNA HCT | aliskiren hemifumarate; hydrochlorothiazide | TABLET;ORAL | 022107-001 | Jan 18, 2008 | ⤷ Start Trial | ⤷ Start Trial |
| Noden Pharma | TEKTURNA HCT | aliskiren hemifumarate; hydrochlorothiazide | TABLET;ORAL | 022107-004 | Jan 18, 2008 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for TEKTURNA HCT
When does loss-of-exclusivity occur for TEKTURNA HCT?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 1565
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 07263261
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0713338
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 54872
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 07001837
Estimated Expiration: ⤷ Start Trial
China
Patent: 1472566
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 088986
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 34968
Estimated Expiration: ⤷ Start Trial
Patent: 11439
Estimated Expiration: ⤷ Start Trial
Patent: 91878
Estimated Expiration: ⤷ Start Trial
Guatemala
Patent: 0800297
Patent: FORMULACIONES GALENICAS DE ALISQUIRENO E HIDROCLOROTIAZIDA
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 5425
Patent: תכשירים גלניקלים של אליסקירן והידרוכלורותיאזיד (Galenical formulations of aliskiren and hydrochlorothiazide)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 62556
Estimated Expiration: ⤷ Start Trial
Patent: 09541239
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 6779
Patent: GALENICAL FORMULATIONS OF ALISKIREN AND HYDROCHLOROTHIAZIDE
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 08016533
Patent: FORMULACIONES GALENICAS DE ALISQUIRENO E HIDROCLOROTIAZIDA. (GALENICAL FORMULATIONS OF ALISKIREN AND HYDROCHLOROTHIAZIDE.)
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 527
Patent: FORMES GALENIQUES ASSOCIANT ALISKIRENE ET HYDROCHLOROTHIAZIDE
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 2937
Patent: Solid oral dosage form of Aliskiren and hydrochlorothiazide which comprise a filler which is a mixture of wheat starch and lactose
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 090262
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 080373
Patent: FORMULACIONES DE ALISQUIRENO E HIDROCLOROTIAZIDA
Estimated Expiration: ⤷ Start Trial
Patent: 120990
Patent: FORMULACIONES DE ALISQUIRENO E HIDROCLOROTIAZIDA
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 91058
Patent: ГАЛЕНОВЫЙ СОСТАВ АЛИСКИРЕНА И ГИДРОХЛОРТИАЗИДА (GALENA COMPOSITION OF ALISKIREN AND HYDROCHLOROTHIAZIDE)
Estimated Expiration: ⤷ Start Trial
Patent: 09101971
Patent: ГАЛЕНОВЫЙ СОСТАВ АЛИСКИРЕНА И ГИДРОХЛОРТИАЗИДА
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 0809773
Patent: Galenical formulations of aliskiren and hydrochlorothiazide
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1442272
Estimated Expiration: ⤷ Start Trial
Patent: 090021353
Patent: GALENICAL FORMULATIONS OF ALISKIREN AND HYDROCHLOROTHIAZIDE
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 04979
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 0808358
Patent: Galenical formulations of organic compounds
Estimated Expiration: ⤷ Start Trial
Patent: 57137
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 08528
Patent: GALENICAL FORMULATIONS OF ALISKIREN AND HYDROCHLOROTHIAZIDE
Estimated Expiration: ⤷ Start Trial
United Kingdom
Patent: 12540
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering TEKTURNA HCT around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Austria | 317692 | ⤷ Start Trial | |
| Japan | 2018030894 | 循環器系疾患のためのレニン阻害剤を含む相乗的組合せ剤 (SYNERGISTIC COMBINATIONS COMPRISING RENIN INHIBITOR FOR CARDIOVASCULAR DISEASES) | ⤷ Start Trial |
| Japan | 3240322 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for TEKTURNA HCT
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1507558 | 132012902049405 | Italy | ⤷ Start Trial | PRODUCT NAME: ALISKIREN, AMLODIPINA, IDROCLOROTIAZIDE(RASITRIO); AUTHORISATION NUMBER(S) AND DATE(S): 61678/01/02/03/04/05, 20110705;EU/1/11/730/001-060, 20111122 |
| 1507558 | 12C0033 | France | ⤷ Start Trial | PRODUCT NAME: ALISKIRENE OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE E CELUI-CI, AMLODIPINE OU SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI, ET HYDROCHLOROTHIAZIDE OU SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI; NAT. REGISTRATION NO/DATE: EU/1/11/730/001 20111122; FIRST REGISTRATION: CH - 6167801 20110705 |
| 1602370 | 132009901728448 | Italy | ⤷ Start Trial | PRODUCT NAME: (ALISKIREN EMIFUMARATO/IDROCLOROTIAZIDE(RASILEZ HCT); AUTHORISATION NUMBER(S) AND DATE(S): DA EU/1/08/491/001 A EU/1/08/491/080, 20090116 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
TEKTURNA HCT: Market Dynamics and Financial Trajectory Analysis
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.