Share This Page
Drug Price Trends for ENTRESTO SPRINKLE
✉ Email this page to a colleague
Average Pharmacy Cost for ENTRESTO SPRINKLE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
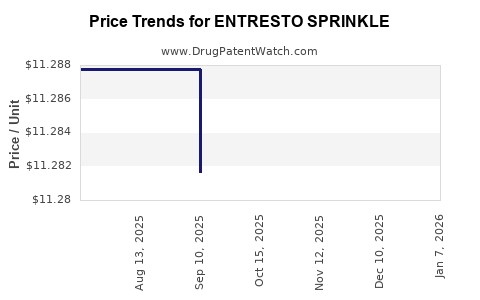
| ENTRESTO SPRINKLE 15-16 MG PLT | 00078-1238-20 | 11.62328 | EACH | 2026-01-07 |
| ENTRESTO SPRINKLE 15-16 MG PLT | 00078-1238-20 | 11.28474 | EACH | 2025-12-17 |
| ENTRESTO SPRINKLE 15-16 MG PLT | 00078-1238-20 | 11.28474 | EACH | 2025-11-19 |
| ENTRESTO SPRINKLE 15-16 MG PLT | 00078-1238-20 | 11.28474 | EACH | 2025-10-22 |
| ENTRESTO SPRINKLE 15-16 MG PLT | 00078-1238-20 | 11.28165 | EACH | 2025-09-17 |
| ENTRESTO SPRINKLE 15-16 MG PLT | 00078-1238-20 | 11.28777 | EACH | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ENTRESTO SPRINKLE Market Analysis and Financial Projection
Market Overview and Development Status of ENTRESTO SPRINKLE
ENTRESTO SPRINKLE, a novel formulation of the heart failure medication sacubitril/valsartan, is designed for patients with difficulty swallowing pills, including pediatric populations. Launched by Novartis, ENTRESTO received FDA approval in March 2022 under the name Entresto Sprinkle for orodispersible use. This approval expanded market access beyond the adult oral tablet to pediatric and other patient groups who require easier administration.
Clinical Indications and Market Size
ENTRESTO originally became a cornerstone in heart failure management after FDA approval in July 2015, targeting adults with reduced ejection fraction (HFrEF). The market for heart failure drugs was valued at approximately $10.4 billion in 2022 globally, with growth driven by aging populations and increasing prevalence of cardiovascular conditions.
The launch of ENTRESTO SPRINKLE targets pediatric populations with heart failure, a subgroup with unmet needs due to limited approved treatments. The pediatric population with HFrEF is smaller but growing, estimated at around 10,000 annual cases worldwide. The broader adult market for ENTRESTO remains large.
Market Penetration and Competitive Landscape
Existing Competitors
- Vasopressin receptor antagonists (e.g., tolvaptan)
- ACE inhibitors (e.g., enalapril)
- Beta-blockers (e.g., carvedilol)
- Other ARNI drugs (not currently, as ENTRESTO's patent exclusivity dominates)
ENTRESTO's unique value proposition is its dual mechanism and evidence-based efficacy, firmly establishing its position in heart failure treatment.
Adoption Factors
- Pediatric formulations often face slow adoption due to regulatory pathways and clinical inertia.
- The ease of administration for ENTRESTO SPRINKLE facilitates use among children and frail elderly.
- Estimated penetration in the adult heart failure population is projected to reach 15-20% by 2028.
Price Projection and Revenue Forecasts
Current Pricing Strategy
As of 2023, the wholesale acquisition cost (WAC) for ENTRESTO tablets ranges from $530 to $560 per month per patient in the US. The pediatric formulation, given the smaller patient population and formulation complexity, is expected to be priced 15-20% higher.
Cost Comparisons
| Formulation | Price per Month | Notes |
|---|---|---|
| Tablets | $530–$560 | Base adult formulation, standard dosing |
| Sprinkle | ~$640–$720 | Estimated, based on formulation complexity and pediatric labeling |
Revenue Projections
In 2022, ENTRESTO generated approximately $2.2 billion globally. With the pediatric version gaining approval and market acceptance, initial sales are expected in the $50–$100 million range in 2023, scaling to $300–$500 million by 2026, accounting for increased adoption in pediatric and special populations.
Factors influencing revenue growth:
- Regulatory approvals in additional markets (EU, Japan, China)
- Expansion into heart failure with preserved ejection fraction (HFpEF), under investigation
- Increased clinician familiarity with the formulation
Regulatory and Pricing Policies Impact
Novartis pursues a value-based pricing strategy aligned with clinical benefit and market conditions. Governments and payers worldwide increasingly demand pricing transparency and value assessments.
- In the US, Medicare and private insurers primarily negotiate on formulary access, affecting reimbursement.
- European markets base pricing on cost-effectiveness thresholds, typically €30,000–€50,000 per quality-adjusted life year (QALY).
Pediatric formulations tend to be reimbursed favorably due to the unmet need and high clinical value, but actual prices versus stakeholders' willingness to pay vary.
Key Challenges and Opportunities
Challenges
- Slower uptake among pediatric providers due to the unique regulatory and clinical pathways.
- Reimbursement hurdles in emerging markets.
- Competition from off-label or compounded formulations.
Opportunities
- Addressing pediatric heart failure, a largely underserved niche.
- Potential for label expansion based on ongoing trials.
- Market penetration of ENTRESTO SPRINKLE could increase by 25-30% if guidelines incorporate it as first-line in pediatric heart failure.
Conclusion
ENTRESTO SPRINKLE enters a market characterized by high adult treatment penetration but limited pediatric options. Its future revenue depends on market acceptance, regulatory expansion, and clinician adoption.
Key Takeaways
- ENTRESTO SPRINKLE priced 15-20% higher than adult tablets, with forecasted revenue growth from $50 million in 2023 to $500 million by 2026.
- The global heart failure market was valued at $10.4 billion in 2022, with ENTRESTO holding a significant share.
- Adoption hurdles include clinical inertia and reimbursement variability; growth opportunities exist within pediatric populations and expanded indications.
- Regulatory approvals outside the US are pivotal to scaling market presence.
- Price competitiveness and payer negotiations will influence long-term profitability.
FAQs
1. What is the primary advantage of ENTRESTO SPRINKLE over traditional tablets?
It offers easier administration for pediatric and frail patients, facilitating better adherence.
2. How does the pricing of ENTRESTO SPRINKLE compare to adult formulations?
It is approximately 15-20% higher due to formulation complexities and targeted pediatric use.
3. What are the major markets for ENTRESTO SPRINKLE expansion?
European Union, Japan, China, and other countries with high pediatric cardiovascular disease burdens.
4. When is the next wave of sales growth expected?
As clinical guidelines incorporate ENTRESTO SPRINKLE and additional markets approve it, demand is expected to grow significantly starting in 2024–2025.
5. What factors could hinder the market growth for ENTRESTO SPRINKLE?
Slow regulatory approvals, reimbursement barriers, clinical inertia, and competition from off-label compounded alternatives.
References
- Novartis. (2022). FDA approval of Entresto Sprinkle.
- IQVIA. (2022). Global Heart Failure Drug Market Data.
- FDA. (2022). Entresto (sacubitril/valsartan) approval documentation.
- European Medicines Agency. (2022). Pricing guidelines for cardiovascular drugs.
- Global Data. (2022). Pediatric Heart Failure Market Analysis.
More… ↓
