OSENI Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Oseni, and what generic alternatives are available?
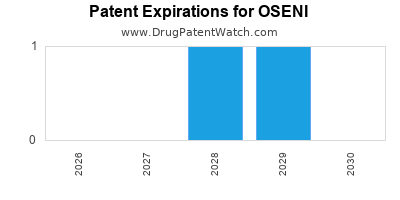
Oseni is a drug marketed by Takeda Pharms Usa and is included in one NDA. There are two patents protecting this drug.
This drug has one hundred and one patent family members in forty-two countries.
The generic ingredient in OSENI is alogliptin benzoate; pioglitazone hydrochloride. There are ten drug master file entries for this compound. Two suppliers are listed for this compound. Additional details are available on the alogliptin benzoate; pioglitazone hydrochloride profile page.
DrugPatentWatch® Generic Entry Outlook for Oseni
Oseni was eligible for patent challenges on January 25, 2017.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be June 4, 2029. This may change due to patent challenges or generic licensing.
There have been five patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for OSENI?
- What are the global sales for OSENI?
- What is Average Wholesale Price for OSENI?
Summary for OSENI
| International Patents: | 101 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 2 |
| Patent Applications: | 83 |
| Drug Prices: | Drug price information for OSENI |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for OSENI |
| What excipients (inactive ingredients) are in OSENI? | OSENI excipients list |
| DailyMed Link: | OSENI at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for OSENI
Generic Entry Date for OSENI*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Pharmacology for OSENI
US Patents and Regulatory Information for OSENI
OSENI is protected by two US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of OSENI is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Takeda Pharms Usa | OSENI | alogliptin benzoate; pioglitazone hydrochloride | TABLET;ORAL | 022426-004 | Jan 25, 2013 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Takeda Pharms Usa | OSENI | alogliptin benzoate; pioglitazone hydrochloride | TABLET;ORAL | 022426-001 | Jan 25, 2013 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Takeda Pharms Usa | OSENI | alogliptin benzoate; pioglitazone hydrochloride | TABLET;ORAL | 022426-005 | Jan 25, 2013 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for OSENI
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Takeda Pharms Usa | OSENI | alogliptin benzoate; pioglitazone hydrochloride | TABLET;ORAL | 022426-003 | Jan 25, 2013 | ⤷ Start Trial | ⤷ Start Trial |
| Takeda Pharms Usa | OSENI | alogliptin benzoate; pioglitazone hydrochloride | TABLET;ORAL | 022426-005 | Jan 25, 2013 | ⤷ Start Trial | ⤷ Start Trial |
| Takeda Pharms Usa | OSENI | alogliptin benzoate; pioglitazone hydrochloride | TABLET;ORAL | 022426-004 | Jan 25, 2013 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for OSENI
When does loss-of-exclusivity occur for OSENI?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 5097
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 08211981
Estimated Expiration: ⤷ Start Trial
Austria
Patent: 88227
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0807453
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 77201
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 08000279
Estimated Expiration: ⤷ Start Trial
China
Patent: 1646420
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 992
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0110094
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 11264
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 07905
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 009000195
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 099608
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 5180
Estimated Expiration: ⤷ Start Trial
Patent: 0970726
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 07905
Estimated Expiration: ⤷ Start Trial
Georgia, Republic of
Patent: 0125410
Estimated Expiration: ⤷ Start Trial
Germany
Patent: 2008003522
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 38188
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 0108
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 84968
Estimated Expiration: ⤷ Start Trial
Patent: 10517937
Estimated Expiration: ⤷ Start Trial
Jordan
Patent: 50
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 7596
Patent: SOLID PREPARATION COMPRISING ALOGLIPTIN AND PIOGLITAZONE
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 09008100
Patent: PREPARACION SOLIDA QUE COMPRENDE ALOGLIPTINA Y PIOGLITAZONA. (SOLID PREPARATION COMPRISING ALOGLIPTIN AND PIOGLITAZONE.)
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 239
Patent: ČVRSTI PREPARAT KOJI SADRŽI ALOGLIPTIN I PIOGLITAZON (SOLID PREPARATION COMPRISING ALOGLIPTIN AND PIOGLITAZONE)
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 169
Patent: إعداد متين يحتوي على الالوكليبتين و البيوجليتازون.
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 9008
Patent: SOLID PREPARATION COMPRISING ALOGLIPTIN AND PIOGLITAZONE
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 081663
Patent: PREPARACION SOLIDA QUE COMPRENDE ALOGLIPTIN Y PIOGLITAZONA
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 07905
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 07905
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 592
Patent: ČVRSTI PREPARAT KOJI SADRŽI ALOGLIPTIN I PIOGLITAZON (SOLID PREPARATION COMPRISING ALOGLIPTIN AND PIOGLITAZONE)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 07905
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 0905621
Patent: Solid preparation comprising alogliptin and pioglitazone
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1486091
Estimated Expiration: ⤷ Start Trial
Patent: 090109115
Patent: SOLID PREPARATION COMPRISING ALOGLIPTIN AND PIOGLITAZONE
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 54397
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 0836775
Patent: Solid preparation
Estimated Expiration: ⤷ Start Trial
Patent: 1350143
Patent: Solid preparation
Estimated Expiration: ⤷ Start Trial
Patent: 53041
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 09000317
Patent: SOLID PREPARATION COMPRISING ALOGLIPTIN AND PIOGLITAZONE
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 828
Patent: ТВЕРДАЯ РЕЦЕПТУРА, КОТОРАЯ СОДЕРЖИТ АЛОГЛИПТИН И ПИОГЛИТАЗОН;ТВЕРДА РЕЦЕПТУРА, ЩО МІСТИТЬ АЛОГЛІПТИН І ПІОГЛІТАЗОН (SOLID PREPARATION COMPRISING ALOGLIPTIN AND PIOGLITAZONE)
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering OSENI around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Austria | E488227 | ⤷ Start Trial | |
| Luxembourg | 92614 | ⤷ Start Trial | |
| Japan | 2017008092 | グルコース代謝を調節する方法、およびそれに関連する試薬 (METHODS FOR REGULATING GLUCOSE METABOLISM AND REAGENTS RELATED THERETO) | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for OSENI
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1084705 | C01084705/03 | Switzerland | ⤷ Start Trial | PRODUCT NAME: SAXAGLIPTIN; REGISTRATION NO/DATE: SWISSMEDIC 59390 05.02.2010 |
| 1586571 | CR 2014 00011 | Denmark | ⤷ Start Trial | PRODUCT NAME: ALOGLIPTIN; REG. NO/DATE: EU/1/13/844/001-027 20130923 |
| 1174135 | SPC/GB10/011 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: THE COMBINATION OF PIOGLITAZONE OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, ESPECIALLY THE HYDROCHLORIDE SALT, AND GLIMEPIRIDE.; REGISTERED: UK EU/1/06/366/001 20070108; UK EU/1/06/366/002 20070108; UK EU/1/06/366/003 20070108; UK EU/1/06/366/004 20070108; UK EU/1/06/366/005 20070108; UK EU/1/06/366/006 20070108; UK EU/1/06/366/019 20070108; UK EU/1/06/366/020 20070108; UK EU/1/06/366/021 20070108; UK EU/1/06/366/022 20070108; UK EU/1/06/366/013 20070108; UK EU/1/06/366/014 20070108; UK EU/1/06/366/015 20070108; UK EU/1/06/366/016 20070108; UK EU/1/06/366/017 20070108; UK EU/1/06/366/018 20070108; UK EU/1/06/366/007 20070108; UK EU/1/06/366/008 20070108; UK EU |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
OSENI (alogliptin + pioglitazone) Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.