Expiring Drug Patents Cheat Sheet
We analyse the patents covering drugs in 134 countries and quickly give you the likely loss-of-exclusivity/generic entry date
Argentina: These 41 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "Argentina: These 41 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027" DrugPatentWatch.com thinkBiotech, 2026 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
When can ZOLINZA (vorinostat) generic drug versions launch?
Generic name: vorinostat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 17, 2026
Generic Entry Controlled by: Argentina Patent 55,953
Patent Title: FORMULACIONES DE ACIDO HIDROXAMICO SUBEROILANILIDA Y PROCEDIMIENTOS PARA LA PRODUCION DE LAS MISMAS
ZOLINZA is a drug marketed by Msd Sub Merck. There are three patents protecting this drug.
This drug has one hundred and thirty-one patent family members in thirty-five countries.
See drug price trends for ZOLINZA.
The generic ingredient in ZOLINZA is vorinostat. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the vorinostat profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 12, 2026
Generic Entry Controlled by: Argentina Patent 61,297
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Argentina Patent 61,510

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can JESDUVROQ (daprodustat) generic drug versions launch?
Generic name: daprodustat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: Argentina Patent 61,570
JESDUVROQ is a drug marketed by Glaxosmithkline. There are six patents protecting this drug.
This drug has forty-four patent family members in thirty-one countries.
See drug price trends for JESDUVROQ.
The generic ingredient in JESDUVROQ is daprodustat. One supplier is listed for this generic product. Additional details are available on the daprodustat profile page.
When can TEKTURNA HCT (aliskiren hemifumarate; hydrochlorothiazide) generic drug versions launch?
Generic name: aliskiren hemifumarate; hydrochlorothiazide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: Argentina Patent 61,565

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering TEKTURNA HCT
See drug price trends for TEKTURNA HCT.
The generic ingredient in TEKTURNA HCT is aliskiren hemifumarate; hydrochlorothiazide. There are four drug master file entries for this API. Additional details are available on the aliskiren hemifumarate; hydrochlorothiazide profile page.
When can EXFORGE HCT (amlodipine besylate; hydrochlorothiazide; valsartan) generic drug versions launch?
Generic name: amlodipine besylate; hydrochlorothiazide; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 27, 2026
Generic Entry Controlled by: Argentina Patent 61,627

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering EXFORGE HCT
See drug price trends for EXFORGE HCT.
The generic ingredient in EXFORGE HCT is amlodipine besylate; hydrochlorothiazide; valsartan. There are fifty drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the amlodipine besylate; hydrochlorothiazide; valsartan profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Argentina Patent 61,730

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Argentina Patent 61,730

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Argentina Patent 61,730

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can BOSULIF (bosutinib monohydrate) generic drug versions launch?
Generic name: bosutinib monohydrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 29, 2026
Generic Entry Controlled by: Argentina Patent 54,505
Patent Title: FORMAS CRISTALINAS DE 4- ((2,4-DICLORO-5-METOXIFENIL) AMINO ) -6-METOXI-7-(3-(4-METIL-1-PIPERAZINIL) PROPOXI) -3- QUINOLINCARBONITRILO Y METODOS DE PREPARACION DE LAS MISMAS
BOSULIF is a drug marketed by Pf Prism Cv. There are four patents protecting this drug and two Paragraph IV challenges. Three tentatively approved generics are ready to enter the market.
This drug has eighty-one patent family members in thirty countries. There has been litigation on patents covering BOSULIF
See drug price trends for BOSULIF.
The generic ingredient in BOSULIF is bosutinib monohydrate. There are five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the bosutinib monohydrate profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Argentina Patent 61,838
Patent Title: MODULADORES DE PROPIEDADES FARMACOCINETICAS DE TERAPEUTICOS

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Argentina Patent 65,439
Patent Title: DERIVADOS DE TIAZOLES 1,3 NO CONDENSADOS,COMPOSICIONES FARMACEUTICAS QUE LOS CONTIENEN Y USOS COMO AGENTES ANTIVIRALES, EN PARTICULAR ANTI HIV.

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Argentina Patent 67,412
Patent Title: MODULADORES DE PROPIEDADES FARMACEUTICAS DE PRODUCTOS TERAPEUTICOS

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Argentina Patent 106,924
Patent Title: MODULADORES DE PROPIEDADES FARMACOCINÉTICAS DE TERAPÉUTICOS

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Argentina Patent 61,989
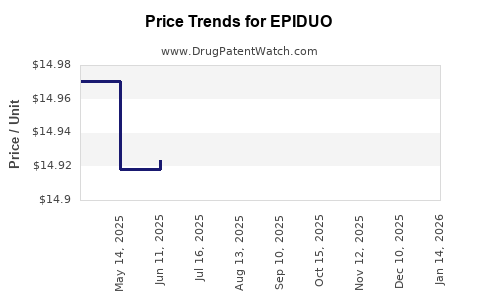
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Argentina Patent 61,989
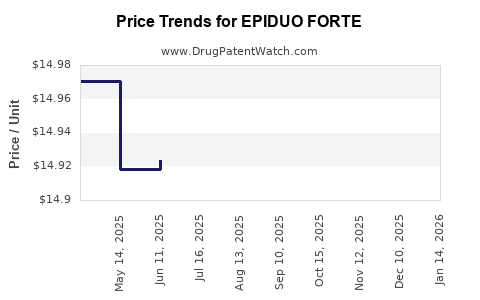
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO FORTE
See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can REZDIFFRA (resmetirom) generic drug versions launch?
Generic name: resmetirom
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 19, 2026
Generic Entry Controlled by: Argentina Patent 54,848
Patent Title: AGONISTAS DEL RECEPTOR DE LA HORMONA TIROIDEA
REZDIFFRA is a drug marketed by Madrigal. There are six patents protecting this drug.
This drug has one hundred and twenty-six patent family members in thirty-seven countries.
See drug price trends for REZDIFFRA.
The generic ingredient in REZDIFFRA is resmetirom. One supplier is listed for this generic product. Additional details are available on the resmetirom profile page.
When can OLYSIO (simeprevir sodium) generic drug versions launch?
Generic name: simeprevir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 28, 2026
Generic Entry Controlled by: Argentina Patent 55,359
Patent Title: INHIBIDORES MACROCICLICOS DEL VIRUS DE LA HEPATITIS C
OLYSIO is a drug marketed by Janssen Prods. There are eight patents protecting this drug.
This drug has sixty-four patent family members in forty-one countries.
See drug price trends for OLYSIO.
The generic ingredient in OLYSIO is simeprevir sodium. There is one drug master file entry for this API. Additional details are available on the simeprevir sodium profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: Argentina Patent 63,684

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: Argentina Patent 108,411

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 21, 2026
Generic Entry Controlled by: Argentina Patent 62,451
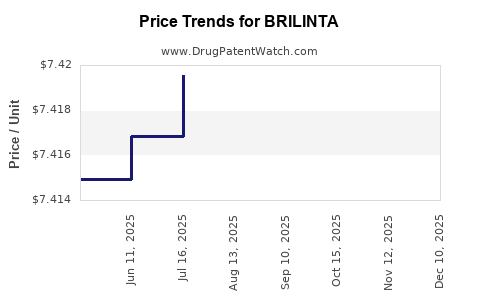
This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 29, 2026
Generic Entry Controlled by: Argentina Patent 62,501
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the macitentan profile page.
When can LIVDELZI (seladelpar lysine) generic drug versions launch?
Generic name: seladelpar lysine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 13, 2026
Generic Entry Controlled by: Argentina Patent 58,044
LIVDELZI is a drug marketed by Gilead Sciences Inc. There are six patents protecting this drug.
This drug has one hundred and thirty-eight patent family members in forty-six countries.
The generic ingredient in LIVDELZI is seladelpar lysine. One supplier is listed for this generic product. Additional details are available on the seladelpar lysine profile page.
When can REVLIMID (lenalidomide) generic drug versions launch?
Generic name: lenalidomide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 03, 2026
Generic Entry Controlled by: Argentina Patent 57,868
Patent Title: METODOS EN LOS QUE SE USA 3-(4-AMINO-1-OXO-1,3-DIHIDRO-ISOINDOL-2-IL)-PIPERIDIN-2,6-DIONA PARA EL TRATAMIENTO DE CIERTAS LEUCEMIAS
REVLIMID is a drug marketed by Bristol Myers Squibb. There are two patents protecting this drug and three Paragraph IV challenges.
This drug has three hundred and thirty-one patent family members in forty-one countries. There has been litigation on patents covering REVLIMID
See drug price trends for REVLIMID.
The generic ingredient in REVLIMID is lenalidomide. There are fourteen drug master file entries for this API. Nineteen suppliers are listed for this generic product. Additional details are available on the lenalidomide profile page.
When can ENTRESTO SPRINKLE (sacubitril; valsartan) generic drug versions launch?
Generic name: sacubitril; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 07, 2026
Generic Entry Controlled by: Argentina Patent 57,882
Patent Title: COMPUESTOS DE ACCION DOBLE DE BLOQUEADORES DEL RECEPTOR DE ANGIOTENSINA E INHIBIDORES DE ENDOPEPTIDASA NEUTRA
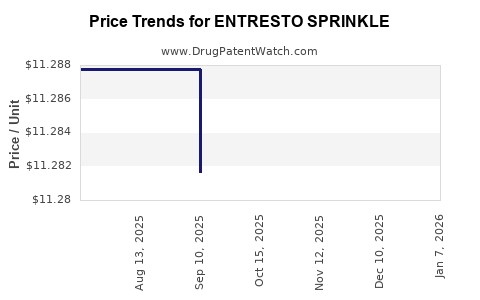
This drug has one hundred and eight patent family members in thirty-nine countries. There has been litigation on patents covering ENTRESTO SPRINKLE
See drug price trends for ENTRESTO SPRINKLE.
The generic ingredient in ENTRESTO SPRINKLE is sacubitril; valsartan. There are eleven drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the sacubitril; valsartan profile page.
When can BEVYXXA (betrixaban) generic drug versions launch?
Generic name: betrixaban
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 08, 2026
Generic Entry Controlled by: Argentina Patent 56,787
Patent Title: SALES Y POLIMORFOS FARMACEUTICOS DE UN INHIBIDOR DEL FACTOR XA
BEVYXXA is a drug marketed by Portola Pharms Inc. There are five patents protecting this drug.
This drug has sixty-seven patent family members in twenty-six countries. There has been litigation on patents covering BEVYXXA
See drug price trends for BEVYXXA.
The generic ingredient in BEVYXXA is betrixaban. Additional details are available on the betrixaban profile page.
When can AVANDAMET (metformin hydrochloride; rosiglitazone maleate) generic drug versions launch?
Generic name: metformin hydrochloride; rosiglitazone maleate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 05, 2026
Generic Entry Controlled by: Argentina Patent 57,970
Patent Title: COMPOSICIONES FARMACEUTICAS PARA EL TRATAMIENTO DE LA DIABETES MELLITUS Y CONDICIONES ASOCIADAS CON LA DIABETES MELLITUS Y PROCEDIMIENTOS PARA PREPRAR DICHAS COMPOSICIONES
AVANDAMET is a drug marketed by Sb Pharmco.
This drug has sixty-seven patent family members in twenty-six countries. There has been litigation on patents covering AVANDAMET
The generic ingredient in AVANDAMET is metformin hydrochloride; rosiglitazone maleate. There are forty-nine drug master file entries for this API. Additional details are available on the metformin hydrochloride; rosiglitazone maleate profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 05, 2026
Generic Entry Controlled by: Argentina Patent 64,149
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Argentina Patent 64,197
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Argentina Patent 77,849
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Argentina Patent 107,966
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can XERMELO (telotristat etiprate) generic drug versions launch?
Generic name: telotristat etiprate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 12, 2026
Generic Entry Controlled by: Argentina Patent 64,279
XERMELO is a drug marketed by Tersera. There are five patents protecting this drug.
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for XERMELO.
The generic ingredient in XERMELO is telotristat etiprate. One supplier is listed for this generic product. Additional details are available on the telotristat etiprate profile page.
When can EPZICOM (abacavir sulfate; lamivudine) generic drug versions launch?
Generic name: abacavir sulfate; lamivudine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 02, 2027
Generic Entry Controlled by: Argentina Patent 59,120
Patent Title: SAL DE DICARBOXILATO DEL (1S,4R) -4- (2-AMINO-6- (CICLOPROPILAMINO ) -9H- PURIN-9-IL)-2- CICLOPENTEN -1- METANOL Y PROCEDIMIENTO PARA SU PREPARACION
EPZICOM is a drug marketed by Viiv Hlthcare. Five tentatively approved generics are ready to enter the market.
This drug has seventy patent family members in twenty-nine countries. There has been litigation on patents covering EPZICOM
See drug price trends for EPZICOM.
The generic ingredient in EPZICOM is abacavir sulfate; lamivudine. There are twelve drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the abacavir sulfate; lamivudine profile page.
When can TRIZIVIR (abacavir sulfate; lamivudine; zidovudine) generic drug versions launch?
Generic name: abacavir sulfate; lamivudine; zidovudine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: January 02, 2027
Generic Entry Controlled by: Argentina Patent 59,120
Patent Title: SAL DE DICARBOXILATO DEL (1S,4R) -4- (2-AMINO-6- (CICLOPROPILAMINO ) -9H- PURIN-9-IL)-2- CICLOPENTEN -1- METANOL Y PROCEDIMIENTO PARA SU PREPARACION
TRIZIVIR is a drug marketed by Viiv Hlthcare. One tentatively approved generic is ready to enter the market.
This drug has seventy patent family members in twenty-nine countries. There has been litigation on patents covering TRIZIVIR
See drug price trends for TRIZIVIR.
The generic ingredient in TRIZIVIR is abacavir sulfate; lamivudine; zidovudine. There are twelve drug master file entries for this API. Additional details are available on the abacavir sulfate; lamivudine; zidovudine profile page.
When can OSENI (alogliptin benzoate; pioglitazone hydrochloride) generic drug versions launch?
Generic name: alogliptin benzoate; pioglitazone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 01, 2027
Generic Entry Controlled by: Argentina Patent 65,097

This drug has one hundred and one patent family members in forty-two countries. There has been litigation on patents covering OSENI
See drug price trends for OSENI.
The generic ingredient in OSENI is alogliptin benzoate; pioglitazone hydrochloride. There are ten drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate; pioglitazone hydrochloride profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Argentina Patent 65,405
PANCREAZE is a drug marketed by
This drug has one hundred and one patent family members in forty-two countries.
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can VIMOVO (esomeprazole magnesium; naproxen) generic drug versions launch?
Generic name: esomeprazole magnesium; naproxen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Argentina Patent 59,589
Patent Title: PROCESO PARA LA PREPARACION DE SAL DE MAGNESIO DE TRIHIDRATO DE S-OMEPRAZOL, SAL DE POTASIO DE S-OMEPRAZOL INTERMEDIARIA Y PROCESO PARA PREPARARLA
VIMOVO is a drug marketed by Horizon. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in forty-two countries. There has been litigation on patents covering VIMOVO
See drug price trends for VIMOVO.
The generic ingredient in VIMOVO is esomeprazole magnesium; naproxen. There are seventy-four drug master file entries for this API. Nine suppliers are listed for this generic product. Additional details are available on the esomeprazole magnesium; naproxen profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Argentina Patent 65,405
ZENPEP is a drug marketed by
This drug has one hundred and one patent family members in forty-two countries.
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Nine suppliers are listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can VARUBI (rolapitant hydrochloride) generic drug versions launch?
Generic name: rolapitant hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 22, 2027
Generic Entry Controlled by: Argentina Patent 65,802
Patent Title: FORMULACIONES DE COMPRIMIDOS QUE CONTIENEN SALES DE 8- [( 1- ( 3,5- BIS- (TRIFLUOROMETIL) FENIL) -ETOXI ) - METIL) -8- FENIL -1, 7- DIAZA- SPIRO [ 4,5] DECAN -2- ONA Y COMPRIMIDOS ELABORADOS A PARTIR DE ESTAS
VARUBI is a drug marketed by Tersera. There are eight patents protecting this drug.
This drug has one hundred and fifty-eight patent family members in thirty-five countries.
The generic ingredient in VARUBI is rolapitant hydrochloride. One supplier is listed for this generic product. Additional details are available on the rolapitant hydrochloride profile page.
When can SELZENTRY (maraviroc) generic drug versions launch?
Generic name: maraviroc
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 27, 2027
Generic Entry Controlled by: Argentina Patent 60,159
Patent Title: COMBINACIONES DE COMPUESTOS DERIVADOS DE TROPANO UTILES EN TERAPIA
This drug has one hundred and fifty-eight patent family members in thirty-five countries.
See drug price trends for SELZENTRY.
The generic ingredient in SELZENTRY is maraviroc. There are two drug master file entries for this API. Nine suppliers are listed for this generic product. Additional details are available on the maraviroc profile page.
When can EDARBI (azilsartan kamedoxomil) generic drug versions launch?
Generic name: azilsartan kamedoxomil
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 28, 2027
Generic Entry Controlled by: Argentina Patent 65,850
Patent Title: COMPOSICION FARMACEUTICA SOLIDA QUE COMPRENDE UN DERIVADO DE BENZIMIDAZOL-7-CARBOXILATO Y UN AGENTE DE CONTROL DE PH
This drug has twenty-one patent family members in seventeen countries. There has been litigation on patents covering EDARBI
See drug price trends for EDARBI.
The generic ingredient in EDARBI is azilsartan kamedoxomil. There are six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the azilsartan kamedoxomil profile page.
When can INLYTA (axitinib) generic drug versions launch?
Generic name: axitinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 05, 2027
Generic Entry Controlled by: Argentina Patent 65,931
INLYTA is a drug marketed by Pf Prism Cv. There are four patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has ninety-seven patent family members in thirty countries. There has been litigation on patents covering INLYTA
See drug price trends for INLYTA.
The generic ingredient in INLYTA is axitinib. There are four drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the axitinib profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 03, 2027
Generic Entry Controlled by: Argentina Patent 59,656
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 03, 2027
Generic Entry Controlled by: Argentina Patent 107,711
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can CONTRAVE (bupropion hydrochloride; naltrexone hydrochloride) generic drug versions launch?
Generic name: bupropion hydrochloride; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 05, 2027
Generic Entry Controlled by: Argentina Patent 61,233
Patent Title: FORMULACION DE LIBERACION PROLONGADA DE NALTREXONA
CONTRAVE is a drug marketed by Nalpropion. There are eighteen patents protecting this drug and one Paragraph IV challenge.
This drug has two hundred and twenty-seven patent family members in forty-five countries. There has been litigation on patents covering CONTRAVE
See drug price trends for CONTRAVE.
The generic ingredient in CONTRAVE is bupropion hydrochloride; naltrexone hydrochloride. There are thirty-eight drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the bupropion hydrochloride; naltrexone hydrochloride profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 15, 2027
Generic Entry Controlled by: Argentina Patent 61,481
Patent Title: COMPUESTOS CON ACTIVIDAD COMBINADA SOBRE SERT, 5-HT3 Y 5-HT1A

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2027
Generic Entry Controlled by: Argentina Patent 67,440
Patent Title: ÁCIDO BILIAR SINTETICO, UNA COMPOSICION QUE LO COMPRENDE, UN METODO PARA SU PREPARACION, COMPUESTOS INTERMEDIARIOS DE SíNTESIS Y SU EMPLEO EN LA ELIMINACION DE LOS DEPoSITOS DE GRASA CORPORAL
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2027
Generic Entry Controlled by: Argentina Patent 112,325
Patent Title: MÉTODO DE PREPARACIÓN DEL ÁCIDO BILIAR SINTÉTICO
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can OJEMDA (tovorafenib) generic drug versions launch?
Generic name: tovorafenib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 29, 2027
Generic Entry Controlled by: Argentina Patent 67,354
OJEMDA is a drug marketed by Day One Biopharms. There are two patents protecting this drug.
This drug has fifty patent family members in twenty-seven countries.
The generic ingredient in OJEMDA is tovorafenib. One supplier is listed for this generic product. Additional details are available on the tovorafenib profile page.
Argentina Branded and Generic Drug Markets Assessment: Regulatory Opportunities and Challenges
More… ↓
DrugPatentWatch cited by CNN, NEJM, Nature Journals, and more …

Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
