Expiring Drug Patents Cheat Sheet
We analyse the patents covering drugs in 134 countries and quickly give you the likely loss-of-exclusivity/generic entry date
Brazil: These 44 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "Brazil: These 44 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027" DrugPatentWatch.com thinkBiotech, 2026 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
Market Analysis and Financial Projection
More… ↓
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 12, 2026
Generic Entry Controlled by: Brazil Patent PI0712993
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 16, 2026
Generic Entry Controlled by: Brazil Patent 122,020,011,899
Patent Title: processos para a preparação de 1-[2-(2,4-dimetil-fenilsulfanil)-fenil]-piperazina e para a fabricação do sal de adição de ácido bromídrico correspondente

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 16, 2026
Generic Entry Controlled by: Brazil Patent 122,020,011,920
Patent Title: composto, composição farmacêutica, método para tratar uma doença, uso de um composto, e processos para a preparação de um composto, e para a fabricação de um composto

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Brazil Patent PI0714039
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Brazil Patent PI0712936

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can JESDUVROQ (daprodustat) generic drug versions launch?
Generic name: daprodustat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: Brazil Patent PI0713446
JESDUVROQ is a drug marketed by Glaxosmithkline. There are six patents protecting this drug.
This drug has forty-four patent family members in thirty-one countries.
See drug price trends for JESDUVROQ.
The generic ingredient in JESDUVROQ is daprodustat. One supplier is listed for this generic product. Additional details are available on the daprodustat profile page.
When can TEKTURNA HCT (aliskiren hemifumarate; hydrochlorothiazide) generic drug versions launch?
Generic name: aliskiren hemifumarate; hydrochlorothiazide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: Brazil Patent PI0713338

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering TEKTURNA HCT
See drug price trends for TEKTURNA HCT.
The generic ingredient in TEKTURNA HCT is aliskiren hemifumarate; hydrochlorothiazide. There are four drug master file entries for this API. Additional details are available on the aliskiren hemifumarate; hydrochlorothiazide profile page.
When can EXFORGE HCT (amlodipine besylate; hydrochlorothiazide; valsartan) generic drug versions launch?
Generic name: amlodipine besylate; hydrochlorothiazide; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 27, 2026
Generic Entry Controlled by: Brazil Patent PI0713785

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering EXFORGE HCT
See drug price trends for EXFORGE HCT.
The generic ingredient in EXFORGE HCT is amlodipine besylate; hydrochlorothiazide; valsartan. There are fifty drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the amlodipine besylate; hydrochlorothiazide; valsartan profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Brazil Patent 122,017,015,106

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Brazil Patent 122,017,021,516

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Brazil Patent PI0713544

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Brazil Patent 122,017,015,106

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Brazil Patent 122,017,021,516

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Brazil Patent PI0713544

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Brazil Patent 122,017,015,106

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Brazil Patent 122,017,021,516

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Brazil Patent PI0713544

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Brazil Patent PI0714055
Patent Title: moduladores de propriedades farmacocinéticas de terapêuticos

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Brazil Patent PI0807581
Patent Title: MODULADORES DE PROPRIEDADES FARMACOCINÉTICAS DE PRODUTOS TERAPÊUTICOS

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can BYFAVO (remimazolam besylate) generic drug versions launch?
Generic name: remimazolam besylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 10, 2026
Generic Entry Controlled by: Brazil Patent PI0714886
Patent Title: sais de benzodiazepina de aÇço rÁpida e suas formas polimàrficas
BYFAVO is a drug marketed by Acacia. There are eleven patents protecting this drug.
This drug has fifty-three patent family members in twenty-four countries. There has been litigation on patents covering BYFAVO
See drug price trends for BYFAVO.
The generic ingredient in BYFAVO is remimazolam besylate. One supplier is listed for this generic product. Additional details are available on the remimazolam besylate profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Brazil Patent PI0713182
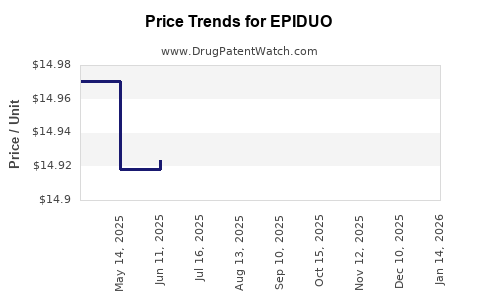
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Brazil Patent PI0713182
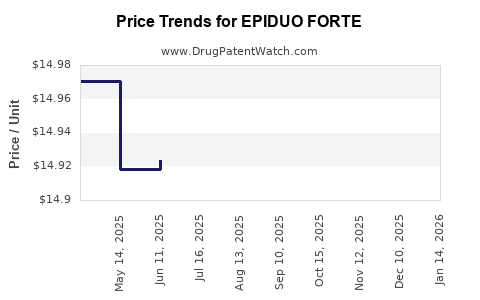
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO FORTE
See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can OLYSIO (simeprevir sodium) generic drug versions launch?
Generic name: simeprevir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 28, 2026
Generic Entry Controlled by: Brazil Patent PI0614654
Patent Title: inibidores macrocìclicos de vìrus de hepatite c
OLYSIO is a drug marketed by Janssen Prods. There are eight patents protecting this drug.
This drug has sixty-four patent family members in forty-one countries.
See drug price trends for OLYSIO.
The generic ingredient in OLYSIO is simeprevir sodium. There is one drug master file entry for this API. Additional details are available on the simeprevir sodium profile page.
When can SALONPAS (menthol; methyl salicylate) generic drug versions launch?
Generic name: menthol; methyl salicylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 04, 2026
Generic Entry Controlled by: Brazil Patent PI0712396
SALONPAS is a drug marketed by Hisamitsu Pharm Co. There are two patents protecting this drug.
This drug has twenty patent family members in sixteen countries.
The generic ingredient in SALONPAS is menthol; methyl salicylate. There are eighteen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the menthol; methyl salicylate profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: Brazil Patent PI0716483

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 15, 2026
Generic Entry Controlled by: Brazil Patent PI0614411
Patent Title: núcleos de micropéletes de pancreatina adequados para revestimento entérico
CREON is a drug marketed by
This drug has ninety-three patent family members in thirty-one countries.
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 29, 2026
Generic Entry Controlled by: Brazil Patent PI0715698
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the macitentan profile page.
When can LIVDELZI (seladelpar lysine) generic drug versions launch?
Generic name: seladelpar lysine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 13, 2026
Generic Entry Controlled by: Brazil Patent PI0615835
LIVDELZI is a drug marketed by Gilead Sciences Inc. There are six patents protecting this drug.
This drug has one hundred and thirty-eight patent family members in forty-six countries.
The generic ingredient in LIVDELZI is seladelpar lysine. One supplier is listed for this generic product. Additional details are available on the seladelpar lysine profile page.
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 22, 2026
Generic Entry Controlled by: Brazil Patent PI0616324
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can TYMLOS (abaloparatide) generic drug versions launch?
Generic name: abaloparatide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 03, 2026
Generic Entry Controlled by: Brazil Patent PI0719821
Patent Title: composiÇço estÁvel na armazenagem apropriada para administraÇço a pacientes e mÉtodo de tratamento de osteoporose nestes

This drug has fifty-six patent family members in twenty-nine countries. There has been litigation on patents covering TYMLOS
See drug price trends for TYMLOS.
The generic ingredient in TYMLOS is abaloparatide. One supplier is listed for this generic product. Additional details are available on the abaloparatide profile page.
When can TYMLOS (abaloparatide) generic drug versions launch?
Generic name: abaloparatide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 03, 2026
Generic Entry Controlled by: Brazil Patent PI0722428
Patent Title: USO DE COMPOSIÇÃO ESTÁVEL NO ARMAZENAMENTO

This drug has fifty-six patent family members in twenty-nine countries. There has been litigation on patents covering TYMLOS
See drug price trends for TYMLOS.
The generic ingredient in TYMLOS is abaloparatide. One supplier is listed for this generic product. Additional details are available on the abaloparatide profile page.
When can INREBIC (fedratinib hydrochloride) generic drug versions launch?
Generic name: fedratinib hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: Brazil Patent 122,021,011,787
INREBIC is a drug marketed by Bristol-myers. There are five patents protecting this drug.
This drug has one hundred and nineteen patent family members in forty-two countries. There has been litigation on patents covering INREBIC
See drug price trends for INREBIC.
The generic ingredient in INREBIC is fedratinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the fedratinib hydrochloride profile page.
When can INREBIC (fedratinib hydrochloride) generic drug versions launch?
Generic name: fedratinib hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: Brazil Patent 122,021,011,788
INREBIC is a drug marketed by Bristol-myers. There are five patents protecting this drug.
This drug has one hundred and nineteen patent family members in forty-two countries. There has been litigation on patents covering INREBIC
See drug price trends for INREBIC.
The generic ingredient in INREBIC is fedratinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the fedratinib hydrochloride profile page.
When can BEVYXXA (betrixaban) generic drug versions launch?
Generic name: betrixaban
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 07, 2026
Generic Entry Controlled by: Brazil Patent PI0618362
Patent Title: sais farmacêuticos inovadores e polimorfos de um inibidor de fator xa
BEVYXXA is a drug marketed by Portola Pharms Inc. There are five patents protecting this drug.
This drug has sixty-seven patent family members in twenty-six countries. There has been litigation on patents covering BEVYXXA
See drug price trends for BEVYXXA.
The generic ingredient in BEVYXXA is betrixaban. Additional details are available on the betrixaban profile page.
When can ENTRESTO SPRINKLE (sacubitril; valsartan) generic drug versions launch?
Generic name: sacubitril; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 08, 2026
Generic Entry Controlled by: Brazil Patent PI0605921
Patent Title: compostos orgánicos
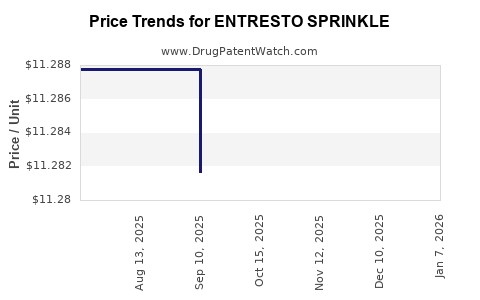
This drug has one hundred and eight patent family members in thirty-nine countries. There has been litigation on patents covering ENTRESTO SPRINKLE
See drug price trends for ENTRESTO SPRINKLE.
The generic ingredient in ENTRESTO SPRINKLE is sacubitril; valsartan. There are eleven drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the sacubitril; valsartan profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 05, 2026
Generic Entry Controlled by: Brazil Patent PI0719693
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Brazil Patent PI0722383
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Brazil Patent PI0815979
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can XERMELO (telotristat etiprate) generic drug versions launch?
Generic name: telotristat etiprate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 12, 2026
Generic Entry Controlled by: Brazil Patent PI0720270
XERMELO is a drug marketed by Tersera. There are five patents protecting this drug.
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for XERMELO.
The generic ingredient in XERMELO is telotristat etiprate. One supplier is listed for this generic product. Additional details are available on the telotristat etiprate profile page.
When can PICATO (ingenol mebutate) generic drug versions launch?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 18, 2026
Generic Entry Controlled by: Brazil Patent PI0619919
Patent Title: composições terapêuticas

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Brazil Patent PI0807718
PANCREAZE is a drug marketed by
This drug has thirty-five patent family members in twenty-one countries.
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Brazil Patent PI0807718
ZENPEP is a drug marketed by
This drug has thirty-five patent family members in twenty-one countries.
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can DYANAVEL XR (amphetamine) generic drug versions launch?
Generic name: amphetamine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 15, 2027
Generic Entry Controlled by: Brazil Patent PI0709606
Patent Title: suspensão líquida administrável oralmente com características de liberação modificada
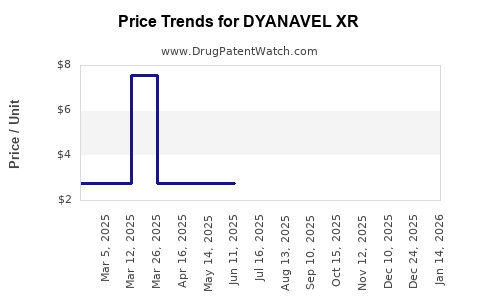
This drug has twenty-one patent family members in fourteen countries. There has been litigation on patents covering DYANAVEL XR
See drug price trends for DYANAVEL XR.
The generic ingredient in DYANAVEL XR is amphetamine. There are fifty-five drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the amphetamine profile page.
When can TUZISTRA XR (chlorpheniramine polistirex; codeine polistirex) generic drug versions launch?
Generic name: chlorpheniramine polistirex; codeine polistirex
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 15, 2027
Generic Entry Controlled by: Brazil Patent PI0709606
Patent Title: suspensão líquida administrável oralmente com características de liberação modificada
TUZISTRA XR is a drug marketed by Tris Pharma Inc. There are two patents protecting this drug.
This drug has twenty-one patent family members in fourteen countries. There has been litigation on patents covering TUZISTRA XR
See drug price trends for TUZISTRA XR.
The generic ingredient in TUZISTRA XR is chlorpheniramine polistirex; codeine polistirex. There are twenty-nine drug master file entries for this API. Additional details are available on the chlorpheniramine polistirex; codeine polistirex profile page.
When can XENLETA (lefamulin acetate) generic drug versions launch?
Generic name: lefamulin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 20, 2027
Generic Entry Controlled by: Brazil Patent PI0809023
XENLETA is a drug marketed by Hong Kong. There are four patents protecting this drug.
This drug has one hundred and twenty patent family members in thirty-six countries. There has been litigation on patents covering XENLETA
See drug price trends for XENLETA.
The generic ingredient in XENLETA is lefamulin acetate. Additional details are available on the lefamulin acetate profile page.
When can INLYTA (axitinib) generic drug versions launch?
Generic name: axitinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 05, 2027
Generic Entry Controlled by: Brazil Patent PI0809471
INLYTA is a drug marketed by Pf Prism Cv. There are four patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has ninety-seven patent family members in thirty countries. There has been litigation on patents covering INLYTA
See drug price trends for INLYTA.
The generic ingredient in INLYTA is axitinib. There are four drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the axitinib profile page.
When can SYMDEKO (COPACKAGED) (ivacaftor; ivacaftor, tezacaftor) generic drug versions launch?
Generic name: ivacaftor; ivacaftor, tezacaftor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 09, 2027
Generic Entry Controlled by: Brazil Patent PI0710965
Patent Title: moduladores de transportadores de cassete de ligação de atp
SYMDEKO (COPACKAGED) is a drug marketed by Vertex Pharms Inc. There are twenty-six patents protecting this drug.
This drug has four hundred and twenty-eight patent family members in forty-one countries. There has been litigation on patents covering SYMDEKO (COPACKAGED)
The generic ingredient in SYMDEKO (COPACKAGED) is ivacaftor; ivacaftor, tezacaftor. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivacaftor; ivacaftor, tezacaftor profile page.
When can QVAR REDIHALER (beclomethasone dipropionate) generic drug versions launch?
Generic name: beclomethasone dipropionate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 11, 2027
Generic Entry Controlled by: Brazil Patent PI0809477
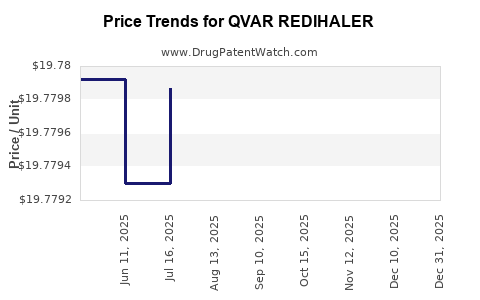
This drug has two hundred and seventy-eight patent family members in twenty-seven countries. There has been litigation on patents covering QVAR REDIHALER
See drug price trends for QVAR REDIHALER.
The generic ingredient in QVAR REDIHALER is beclomethasone dipropionate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the beclomethasone dipropionate profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 03, 2027
Generic Entry Controlled by: Brazil Patent PI0721651
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can TRELSTAR (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 06, 2027
Generic Entry Controlled by: Brazil Patent PI0812250
Patent Title: COMPOSIÇÃO FARMACÊUTICA DE LIBERAÇÃO LENTA FEITA DE MICROPARTÍCULAS
TRELSTAR is a drug marketed by Verity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
See drug price trends for TRELSTAR.
The generic ingredient in TRELSTAR is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can TRIPTODUR KIT (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 06, 2027
Generic Entry Controlled by: Brazil Patent PI0812250
Patent Title: COMPOSIÇÃO FARMACÊUTICA DE LIBERAÇÃO LENTA FEITA DE MICROPARTÍCULAS
TRIPTODUR KIT is a drug marketed by Azurity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
The generic ingredient in TRIPTODUR KIT is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 15, 2027
Generic Entry Controlled by: Brazil Patent PI0713425
Patent Title: composto, composição farmacêutica, método para tratar uma doença, uso de um composto, e processos para a preparação de um composto, e para a fabricação de um composto

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2027
Generic Entry Controlled by: Brazil Patent PI0813140
Patent Title: COMPOSIÇÃO, MÉTODO E PREPARAÇÃO DE ÁCIDO BILIAR SINTÉTICO
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can OJEMDA (tovorafenib) generic drug versions launch?
Generic name: tovorafenib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 29, 2027
Generic Entry Controlled by: Brazil Patent PI0813499
OJEMDA is a drug marketed by Day One Biopharms. There are two patents protecting this drug.
This drug has fifty patent family members in twenty-seven countries.
The generic ingredient in OJEMDA is tovorafenib. One supplier is listed for this generic product. Additional details are available on the tovorafenib profile page.
When can TWIRLA (ethinyl estradiol; levonorgestrel) generic drug versions launch?
Generic name: ethinyl estradiol; levonorgestrel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 10, 2027
Generic Entry Controlled by: Brazil Patent PI0814697
Patent Title: dispositivo de liberação dérmica com uma vedação no local
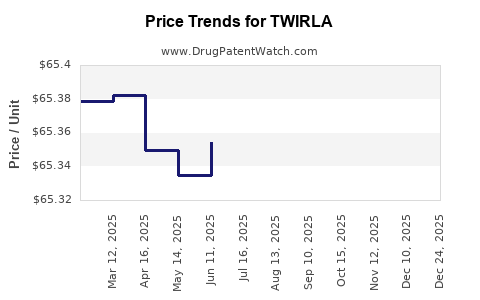
This drug has nineteen patent family members in twelve countries.
See drug price trends for TWIRLA.
The generic ingredient in TWIRLA is ethinyl estradiol; levonorgestrel. There are twenty-six drug master file entries for this API. Twenty-three suppliers are listed for this generic product. Additional details are available on the ethinyl estradiol; levonorgestrel profile page.
Brazil Drug Market: Patent Landscape and Regulatory Opportunities
More… ↓
DrugPatentWatch cited by CNN, NEJM, Nature Journals, and more …

Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
