Last updated: December 31, 2025
Summary
Actelion, a biotech pioneer specializing in rare and orphan diseases, has established itself as a key player within the pulmonary arterial hypertension (PAH) space and other niche therapeutic areas. Acquired by Johnson & Johnson in 2017 for approximately $30 billion, Actelion’s strategic positioning reflects a focus on innovative therapies, accelerated growth through pipelines, and expanding global reach. This analysis evaluates Actelion’s current market position, core strengths, competitive landscape, and strategic pathways amid evolving industry dynamics.
What is Actelion’s Market Position in the Global Pharma Ecosystem?
Market Segmentation and Revenue Footprints
| Area of Focus |
Key Products |
Market Share (Est.) |
Estimated Revenue (2022) |
Notes |
| Pulmonary Hypertension (PAH) |
Tracleer (bosentan), Uptravi (selexipag), Ventavis (iloprost) |
~25-30% in global PAH market |
~$2.0 billion (2022) |
Leader in orphan PAH therapies; maintains a dominant market share |
| Rare Diseases & Orphan Drugs |
Multiple pipeline candidates and partnerships |
Growing segment; niche-focused |
N/A |
Strategic expansion into other rare diseases like systemic sclerosis |
Global Presence & Market Penetration
- Geography: Operates robustly across North America, Europe, and Asia-Pacific.
- Global Reach: Post-acquisition, enhanced distribution networks and strategic partnerships facilitate better penetration, especially in emerging markets.
- Competitive Position:
- Leading in PAH versus competitors like Gilead Sciences (Letairis), Bayer (Adempas).
- Emerging in Rare Diseases: Focused investments to diversify revenue streams away from dependence on PAH treatments.
What Are Actelion’s Core Strengths in a Competitive Landscape?
Innovative Product Portfolio & R&D Pipeline
- Market-Leading Drugs:
- Tracleer: First approved oral endothelin receptor antagonist, 2001.
- Uptravi: Oral selective IP receptor agonist, approved in 2015.
- Ventavis: Inhaled prostacyclin, approved in 2004.
- Pipeline Diversification:
- Focused on rare and orphan diseases, including systemic sclerosis-associated pulmonary arterial hypertension (SSc-PAH) and other fibrotic disorders.
- Key candidates: Selexipag analogs, gene therapies, and biologics targeting disease pathways.
Strategic R&D & Innovation
- High R&D Investment: Allocated approximately 20% of revenues to pipeline development (2022 figures).
- Collaborations & Partnerships:
- Strategic alliances with academic institutions and biotech firms to enhance innovation.
- Licensing agreements to expand pipeline efficacy and reach.
Regulatory & Market Access Strengths
- A track record of successful regulatory filings globally—FDA, EMA, PMDA.
- Early payer engagement strategies, including risk-sharing schemes, bolster market access.
Operational & Organizational Capabilities
- Integrated Global Supply Chain: Robust manufacturing and distribution, crucial for orphan drug niche markets.
- Expertise in Rare Disease: Deep understanding of patient needs and regulatory pathways.
What Are the Strategic Challenges and Risks Faced by Actelion?
Intensified Competition in PAH and Orphan Markets
| Competitor |
Focus Area |
Key Products |
Competitive Threat Level |
Notes |
| Gilead Sciences |
PAH, Fibrosis |
Letairis, Vosevi |
High |
Expanding pipeline with új formulations & combos |
| Bayer |
PAH, Oncology |
Adempas, Belvar |
Moderate |
Diversification beyond PAH |
| United Therapeutics |
PAH, Rare Diseases |
Remodulin, Tyvaso |
Moderate |
Strong market presence, pipeline expansion |
Pricing & Reimbursement Pressures
- Increasing scrutiny of drug prices, especially in the US and Europe, puts margin pressure.
- Reimbursement hurdles for orphan drugs across different jurisdictions.
Pipeline Risks
- High R&D costs with uncertain success rates.
- Potential delays or failures in clinical development affecting revenue forecasts.
Regulatory & Market Dynamics
- Evolving guidelines for orphan and rare disease therapies.
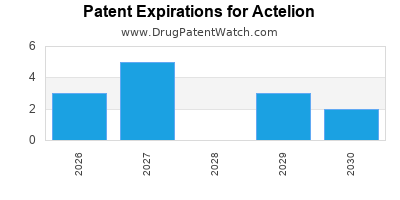
- Patent cliffs looming for key products post-2030.
What Strategic Opportunities Can Actelion Exploit?
Expansion into Adjacent Therapeutic Areas
- Fibrotic Diseases & Pulmonology:
- Leverage existing PAH expertise to develop treatments for systemic sclerosis and idiopathic pulmonary fibrosis.
- Gene & Cell Therapies:
- Invest in cutting-edge therapies to address unmet needs.
Digital Transformation & Data Analytics
- Utilize AI/ML for faster drug discovery.
- Harness real-world evidence for efficacy and safety optimization.
Global Market Penetration Strategies
| Strategy |
Action Items |
Expected Impact |
| Local Partnerships |
Collaborate with regional biotech & pharma |
Increased access and faster regulatory approvals |
| Market-Specific Pricing |
Adaptive pricing models |
Improved reimbursement status |
Pipeline Optimization & Diversification
- Focus on high unmet needs within rare diseases.
- Use accelerated approval pathways for promising candidates.
Comparison with Leading Competitors
| Aspect |
Actelion |
Gilead Sciences |
Bayer |
United Therapeutics |
| Core Focus |
PAH & Rare Diseases |
Viral & Pulmonary |
Cardiovascular, Oncology |
Pulmonary & Rare Diseases |
| Revenue (2022) |
~$2.0 billion |
~$30 billion (Gilead total) |
~$20 billion (Bayer pharma) |
~$2.0 billion (UT) |
| R&D Investment |
20% of revenues |
16% of revenues |
10% of revenues |
20% of revenues |
| Pipeline Strength |
Focused, niche, growing |
Diversified, broad |
Diversified, strong |
Focused on pulmonary and rare |
Conclusion: Is Actelion Positioned for Future Growth?
Actelion's unique positioning as a leader in orphan pulmonary hypertension therapies, combined with strategic acquisitions and pipeline expansion, underscore its resilience and growth potential. Its core strengths—innovative product portfolio, regulatory expertise, and global footprint—provide a competitive moat. However, challenges such as intensified competition, pricing pressures, and pipeline risks require vigilant strategic management.
Looking ahead, Actelion’s success hinges on its ability to innovate within its niche, capitalize on adjacent rare disease markets, and adapt to geopolitical and regulatory shifts impacting pharma markets globally.
Key Takeaways
- Actelion maintains a dominant position in the PAH market, with a diversified portfolio of therapies and a robust pipeline.
- Strategic expansion into other rare and fibrotic diseases offers growth opportunities amid competitive pressures.
- Key strengths include high R&D investment, global distribution, and regulatory expertise.
- Intensified competition and pricing pressures present ongoing risks.
- Collaboration, pipeline innovation, and geographic expansion are vital for future sustainability.
FAQs
1. How does Actelion differentiate itself from competitors?
Actelion's differentiation stems from its early leadership in PAH therapies, a targeted focus on rare diseases, a solid R&D pipeline, and global manufacturing capabilities.
2. What are the main growth areas for Actelion over the next five years?
The primary growth avenues include expanding beyond PAH into other rare and fibrotic diseases, leveraging gene and biologic therapies, and deepening global market access, especially in emerging economies.
3. How does regulatory policy impact Actelion’s strategic prospects?
Stringent regulatory pathways for orphan drugs can delay product launches but also provide market exclusivity incentives. Navigating these effectively is pivotal for pipeline success.
4. What are potential risks associated with Actelion’s pipeline?
Risks include clinical trial failures, regulatory setbacks, and high development costs, which could impact projected revenues.
5. How critical is digital innovation for Actelion’s future?
Digital transformation and data analytics are increasingly vital for accelerating drug discovery, optimizing trials, and patient monitoring, making them strategic priorities for sustained growth.
References
[1] Johnson & Johnson Annual Report 2022.
[2] Actelion Financials & Pipeline Data (2022).
[3] EvaluatePharma World Preview 2023.
[4] ClinicalTrials.gov.
[5] Industry Reports on Rare Disease Market Dynamics, 2022/2023.
Disclaimer: All data and analysis are based on publicly available information as of 2023 and may evolve with new market developments.