ENTRESTO SPRINKLE Drug Patent Profile
✉ Email this page to a colleague
When do Entresto Sprinkle patents expire, and when can generic versions of Entresto Sprinkle launch?
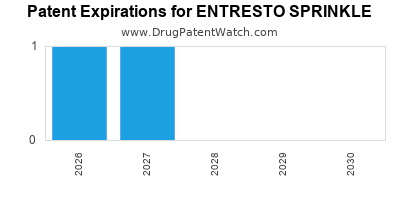
Entresto Sprinkle is a drug marketed by Novartis and is included in one NDA. There are three patents protecting this drug.
This drug has one hundred and eight patent family members in thirty-nine countries.
The generic ingredient in ENTRESTO SPRINKLE is sacubitril; valsartan. There are eleven drug master file entries for this compound. Twenty-four suppliers are listed for this compound. Additional details are available on the sacubitril; valsartan profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Entresto Sprinkle
A generic version of ENTRESTO SPRINKLE was approved as sacubitril; valsartan by ALEMBIC on May 28th, 2024.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ENTRESTO SPRINKLE?
- What are the global sales for ENTRESTO SPRINKLE?
- What is Average Wholesale Price for ENTRESTO SPRINKLE?
Summary for ENTRESTO SPRINKLE
| International Patents: | 108 |
| US Patents: | 3 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
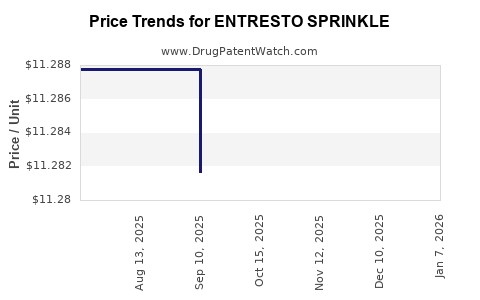
| Drug Prices: | Drug price information for ENTRESTO SPRINKLE |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for ENTRESTO SPRINKLE |
| DailyMed Link: | ENTRESTO SPRINKLE at DailyMed |
Pharmacology for ENTRESTO SPRINKLE
| Drug Class | Angiotensin 2 Receptor Blocker Neprilysin Inhibitor |
| Mechanism of Action | Angiotensin 2 Receptor Antagonists Neprilysin Inhibitors |
US Patents and Regulatory Information for ENTRESTO SPRINKLE
ENTRESTO SPRINKLE is protected by three US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Novartis | ENTRESTO SPRINKLE | sacubitril; valsartan | CAPSULE, PELLETS;ORAL | 218591-001 | Apr 12, 2024 | RX | Yes | No | 10,722,471 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Novartis | ENTRESTO SPRINKLE | sacubitril; valsartan | CAPSULE, PELLETS;ORAL | 218591-002 | Apr 12, 2024 | RX | Yes | Yes | 10,722,471 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Novartis | ENTRESTO SPRINKLE | sacubitril; valsartan | CAPSULE, PELLETS;ORAL | 218591-001 | Apr 12, 2024 | RX | Yes | No | 9,388,134 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Novartis | ENTRESTO SPRINKLE | sacubitril; valsartan | CAPSULE, PELLETS;ORAL | 218591-001 | Apr 12, 2024 | RX | Yes | No | 8,877,938 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Novartis | ENTRESTO SPRINKLE | sacubitril; valsartan | CAPSULE, PELLETS;ORAL | 218591-002 | Apr 12, 2024 | RX | Yes | Yes | 8,877,938 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ENTRESTO SPRINKLE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Novartis | ENTRESTO SPRINKLE | sacubitril; valsartan | CAPSULE, PELLETS;ORAL | 218591-001 | Apr 12, 2024 | 8,101,659 | ⤷ Start Trial |
| Novartis | ENTRESTO SPRINKLE | sacubitril; valsartan | CAPSULE, PELLETS;ORAL | 218591-002 | Apr 12, 2024 | 8,101,659 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for ENTRESTO SPRINKLE
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Novartis Europharm Limited | Neparvis | sacubitril, valsartan | EMEA/H/C/004343Paediatric heart failureNeparvis is indicated in children and adolescents aged one year or older for treatment of symptomatic chronic heart failure with left ventricular systolic dysfunction (see section 5.1).Adult heart failureNeparvis is indicated in adult patients for treatment of symptomatic chronic heart failure with reduced ejection fraction (see section 5.1). | Authorised | no | no | no | 2016-05-26 | |
| Novartis Europharm Limited | Entresto | sacubitril, valsartan | EMEA/H/C/004062Paediatric heart failureEntresto is indicated in children and adolescents aged one year or older for treatment of symptomatic chronic heart failure with left ventricular systolic dysfunction.Adult heart failureEntresto is indicated in adult patients for treatment of symptomatic chronic heart failure with reduced ejection fraction. | Authorised | no | no | no | 2015-11-19 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for ENTRESTO SPRINKLE
When does loss-of-exclusivity occur for ENTRESTO SPRINKLE?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 7882
Patent: COMPUESTOS DE ACCION DOBLE DE BLOQUEADORES DEL RECEPTOR DE ANGIOTENSINA E INHIBIDORES DE ENDOPEPTIDASA NEUTRA
Estimated Expiration: ⤷ Start Trial
Patent: 6614
Patent: COMPUESTOS ORGÁNICOS
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0605921
Patent: compostos orgánicos
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 90511
Patent: COMBINAISON PHARMACEUTIQUES D'UN ANTAGONISTE DE RECEPTEUR D'ANGIOTENSINE ET D'UN INHIBITEUR DE NEP (PHARMACEUTICAL COMBINATIONS OF AN ANGIOTENSIN RECEPTOR ANTAGONIST AND AN NEP INHIBITOR)
Estimated Expiration: ⤷ Start Trial
China
Patent: 2702119
Patent: Pharmaceutical combinations of an angiotensin receptor antagonist and an NEP inhibitor
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0140274
Estimated Expiration: ⤷ Start Trial
Patent: 0201605
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 21003
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 40828
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 600025
Estimated Expiration: ⤷ Start Trial
Patent: 100003
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 4027
Patent: תרכובת טריסודיום [3-((1s, 3r)-1-ביפניל-4-ילמתיל-3-אתוקסיקרבוניל-1-בוטילקרבמויל)פרופיונאט-(s)-'3-מתיל-'2-(פנטהנויל{" 2-(טטרזול-5-ילאט)ביפניל-'4-ילמתיל}אמינו)בוטיראט] המיפנטההידראט, תכשירים רוקחיים שלה, שיטה להכנתה ושימוש בה להכנת תרופות (Compound trisodium [3-((1s,3r)-1-biphenyl-4-ylmethyl-3-ethoxycarbonyl-1-butylcarbamoyl)propionate-(s)-3’-methyl-2’-(pentanoyl{2’’-(tetrazol-5-ylate)biphenyl-4’-ylmethyl}amino)butyrate] hemipentahydrate, its pharmaceutical compositions, method for its preparation and use thereof in the preparation of medicaments)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 08542447
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 948158
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 5462
Patent: PHARMACEUTICAL COMBINATIONS OF AN ANGIOTENSIN RECEPTOR ANTAGONIST AND AN NEP INHIBITOR
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 07008075
Patent: COMBINACIONES FARMACEUTICAS DE UN ANTAGONISTA DEL RECEPTOR DE ANGIOTENSINA Y UN INHIBIDOR DE ENDOPEPTIDASA NEUTRAL (NEP). (PHARMACEUTICAL COMBINATIONS OF AN ANGIOTENSIN RECEPTOR ANTAGONIST AND AN NEP INHIBITOR.)
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 128
Patent: COMBINAISONS PHARMACEUTIQUES D'UN ANTAGONISTE DE RECEPTUER D'ANGIOTENSINE ET D'UN INHIBITEUR DE NEP
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 4006
Patent: Pharmaceutical combinations of an angiotensin receptor antagonist and an NEP inhibitor
Estimated Expiration: ⤷ Start Trial
Patent: 0626
Patent: Pharmaceutical combinations of an angiotensin receptor antagonist and an NEP inhibitor
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 151656
Patent: Farmasøytiske kombinasjoner av en angiotensinreseptorantagonist og en nepinhibitor
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 48158
Estimated Expiration: ⤷ Start Trial
Patent: 40828
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 48158
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 03668
Patent: ФАРМАЦЕВТИЧЕСКИЕ КОМБИНАЦИИ АНТАГОНИСТА РЕЦЕПТОРА АНГИОТЕНЗИНА И ИНГИБИТОРА NEP (PHARMACEUTICAL COMBINATIONS OF ANGIOTENSIN RECEPTOR ANTAGONIST AND NEP INHIBITOR)
Estimated Expiration: ⤷ Start Trial
Patent: 07123671
Patent: ФАРМАЦЕВТИЧЕСКИЕ КОМБИНАЦИИ АНТАГОНИСТА РЕЦЕПТОРА АНГИОИЕНЗИНА И ИНГИБИТОРА NEP
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 40828
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1432821
Estimated Expiration: ⤷ Start Trial
Patent: 1549318
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1402111
Patent: Dual-acting compound, pharmaceutical composition containing it, process for its preparation and use thereof
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 07264
Patent: ORGANIC COMPOUNDS
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ENTRESTO SPRINKLE around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Brazil | 122018015003 | kit compreendendo composições farmacêuticas de inibidor da nep e de valsartan ou seus sais | ⤷ Start Trial |
| Norway | 340315 | ⤷ Start Trial | |
| Taiwan | I554504 | ⤷ Start Trial | |
| Japan | 2011207898 | PHARMACEUTICAL COMPOSITION CONTAINING VALSARTAN AND NEP INHIBITOR | ⤷ Start Trial |
| Denmark | 2340828 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ENTRESTO SPRINKLE
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1948158 | 122016000038 | Germany | ⤷ Start Trial | PRODUCT NAME: SACUBITRIL/VALSARTAN, ALS SACUBITRIL VALSARTAN NATRIUM SALZKOMPLEX, D.H. TRINATRIUM-(3-((1 S,3R)-1-BIPHENYL-4-YLMETHYL-3-ETHOXYCARBONYL-1-BUTYLCARBAMOYL)PROPIONAT-(S)-3'-METHYL-2'-(PENTANOYL-(2"-(TETRAZOL-5-YLAT)BIPHENYL-4'-YLMETHYL)AMINO)BUTYRAT)-HEMIPENTAHYDRAT.; REGISTRATION NO/DATE: EU/1/15/1058 20151119 |
| 2340828 | LUC00195 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: SACUBITRIL ET VALSARTAN, SOUS FORME DE COMPLEXE DE SEL DE SODIUM SACUBITRIL VALSARTAN, C'EST-A-DIRE (((S)-N-VALERYL-N-((2'-(1H-TETRAZOLE-5-YL)-BIPHENYL-4-YL)-METHYL)-VALINE) ((2R,4S)-5-BIPHENYL-4-YL-4-(3-CARBOXY-PROPIONYLAMINO)-2-METHYL-ESTER ETHYLIQUE D'ACIDE PENTANOIQUE))NA3 X H2O, DANS LEQUEL X EST 0 A 3; AUTHORISATION NUMBER AND DATE: EU/1/15/1058 20151123 |
| 2340828 | 122021000003 | Germany | ⤷ Start Trial | PRODUCT NAME: SACUBITRIL/VALSARTAN, ALS SACUBITRIL VALSARTAN NATRIUM SALZKOMPLEX, D.H. (((S)-N-VALERYL-N-((2'-(1H-TETRAZOL-5-YL)-BIPHENYL-4-YL)-METHYL)-VALIN) ((2R,4S)-5-BIPHENYL-4-YL-4-(3-CARBOXY-PROPIONYLAMINO)-2-METHYL-PENTANSAEUREETHYLESTER))NA 3 * X H 2 0, WORIN X 0 BIS 3 IST; REGISTRATION NO/DATE: EU/1/15/1058 20151119 |
| 1948158 | C01948158/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: SACUBITRIL UND VALSARTAN; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 65673 17.09.2015 |
| 2340828 | 2021C/502 | Belgium | ⤷ Start Trial | PRODUCT NAME: SACUBITRIL/VALSARTAN, ALS HET SACUBITRIL VALSARTAN NATRIUMZOUTCOMPLEX, I.E. (((S)-N-VALERYL-N-((2'-(1H-TETRAZOL-5-YL)-BIFENYL-4-YL)-METHYL)-VALINE) ((2R,4S)-5-BIFENYL-4-YL-4-(3-CARBOXY-PROPIONYLAMINO)-2-METHYL-PENTAANZUURETHYLESTER))NA3 . X H2O, WAARBIJ X 0 TOT 3 IS; AUTHORISATION NUMBER AND DATE: EU/1/15/1058 (C(2015) 8288) 20151123 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for ENTRESTO SPRINKLE
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.