Expiring Drug Patents Cheat Sheet
We analyse the patents covering drugs in 134 countries and quickly give you the likely loss-of-exclusivity/generic entry date
Uruguay: These 8 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "Uruguay: These 8 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027" DrugPatentWatch.com thinkBiotech, 2026 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 12, 2026
Generic Entry Controlled by: Uruguay Patent 30,406
Patent Title: POLIMORFOS DE N-HIDROXI-3-[4-[[[2-(2-METIL-1H-INDOL-3-IL]AMINO]METIL]FENIL]-2E-2-PROPENAMIDA
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can OLYSIO (simeprevir sodium) generic drug versions launch?
Generic name: simeprevir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 28, 2026
Generic Entry Controlled by: Uruguay Patent 29,703
Patent Title: "INHIBIDORES MACROCÍCLICOS DEL VIRUS DE LA HEPATITIS C"
OLYSIO is a drug marketed by Janssen Prods. There are eight patents protecting this drug.
This drug has sixty-four patent family members in forty-one countries.
See drug price trends for OLYSIO.
The generic ingredient in OLYSIO is simeprevir sodium. There is one drug master file entry for this API. Additional details are available on the simeprevir sodium profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 21, 2026
Generic Entry Controlled by: Uruguay Patent 30,551
Patent Title: COMPOSICIONES FARMACÉUTICAS CONTENIENDO {1S-[1A(ALFA),2 A(ALFA), 3B(BETA)(1S*,2R*),5 B(BETA)]}-3-7-{[2-3,4-DIFLUOROFENIL)CICLOPROPIL]AMINO}-5-(PROPILITIO-3H-1,2,3-TRIAZOLO [4,5-D] PIRIMIDIN-3-Y 1-5-(2-HIDROXIETOXI)CICLOPENTANO-1,2-DIOL
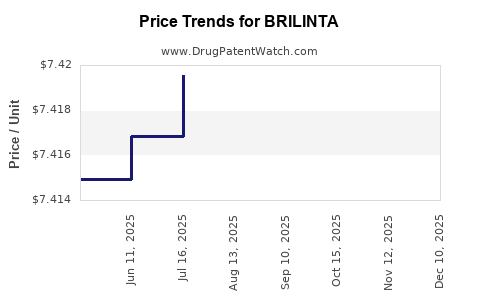
This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can LIVDELZI (seladelpar lysine) generic drug versions launch?
Generic name: seladelpar lysine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 14, 2026
Generic Entry Controlled by: Uruguay Patent 29,791
Patent Title: SALES NOVEDOSAS DE LISINA DE DERIVADOS DE ACIDO 4-( (FENOXIALQUIL)TIO)-FENOXIACETICO
LIVDELZI is a drug marketed by Gilead Sciences Inc. There are six patents protecting this drug.
This drug has one hundred and thirty-eight patent family members in forty-six countries.
The generic ingredient in LIVDELZI is seladelpar lysine. One supplier is listed for this generic product. Additional details are available on the seladelpar lysine profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 05, 2026
Generic Entry Controlled by: Uruguay Patent 30,762
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can LASTACAFT (alcaftadine) generic drug versions launch?
Generic name: alcaftadine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 02, 2027
Generic Entry Controlled by: Uruguay Patent 30,254
Patent Title: TRATAMIENTOS PARA LA ALERGIA OCULAR
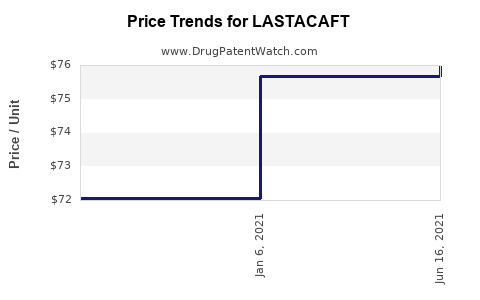
This drug has forty-six patent family members in thirty countries. There has been litigation on patents covering LASTACAFT
See drug price trends for LASTACAFT.
The generic ingredient in LASTACAFT is alcaftadine. There are six drug master file entries for this API. Four suppliers are listed for this generic product. Additional details are available on the alcaftadine profile page.
When can TRADJENTA (linagliptin) generic drug versions launch?
Generic name: linagliptin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 03, 2027
Generic Entry Controlled by: Uruguay Patent 30,319
Patent Title: FORMULACIONES DE INHIBIDORES DE DPP IV
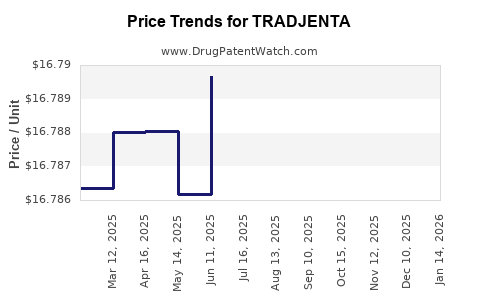
This drug has three hundred and eighty-one patent family members in forty-one countries. There has been litigation on patents covering TRADJENTA
See drug price trends for TRADJENTA.
The generic ingredient in TRADJENTA is linagliptin. There are nineteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the linagliptin profile page.
When can SIGNIFOR LAR (pasireotide pamoate) generic drug versions launch?
Generic name: pasireotide pamoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 24, 2027
Generic Entry Controlled by: Uruguay Patent 31,103
Patent Title: COMPOSICIONES FARMACEUTICAS EN BASE A MICROPARTICULAS COMPRENDIENDO UN ANALOGO DE SOMATOSTATINA, PROCESO DE PREPARACION Y APLICACIONES
SIGNIFOR LAR is a drug marketed by Recordati Rare. There are three patents protecting this drug.
This drug has three hundred and eighty-one patent family members in forty-one countries.
See drug price trends for SIGNIFOR LAR.
The generic ingredient in SIGNIFOR LAR is pasireotide pamoate. One supplier is listed for this generic product. Additional details are available on the pasireotide pamoate profile page.
Uruguay Drug Market Assessment: Regulatory Landscape and Opportunities
More… ↓
DrugPatentWatch cited by CNN, NEJM, Nature Journals, and more …

Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
