Expiring Drug Patents Cheat Sheet
We analyse the patents covering drugs in 134 countries and quickly give you the likely loss-of-exclusivity/generic entry date
Hong Kong: These 38 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "Hong Kong: These 38 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027" DrugPatentWatch.com thinkBiotech, 2026 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 15, 2026
Generic Entry Controlled by: Hong Kong Patent 1,134,775
Patent Title: 抗濫用藥物製劑 (ABUSE RESISTANT DRUG FORMULATION)
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 15, 2026
Generic Entry Controlled by: Hong Kong Patent 1,193,567
Patent Title: 抗濫用藥物配方 (ABUSE RESISTANT DRUG FORMULATION)
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 16, 2026
Generic Entry Controlled by: Hong Kong Patent 1,172,014
Patent Title: 作為用於治療認知損傷的、具有結合的對血清素再吸收、 活性的化合物的 -二甲基苯基硫烷基 -苯基 哌嗪 (1- [2- (2, 4-DIMETHYLPHENYLSULFANYL) -PHENYL] PIPERAZINE AS A COMPOUND WITH COMBINED SEROTONIN REUPTAKE, 5-HT3 AND 5-HT1A ACTIVITY FOR THE TREATMENT OF COGNITIVE IMPAIRMENT 5-HT3 5-HT1A 1-[2-(24-)-])

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Hong Kong Patent 1,173,980
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Hong Kong Patent 1,130,245

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can JESDUVROQ (daprodustat) generic drug versions launch?
Generic name: daprodustat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: Hong Kong Patent 1,130,671
JESDUVROQ is a drug marketed by Glaxosmithkline. There are six patents protecting this drug.
This drug has forty-four patent family members in thirty-one countries.
See drug price trends for JESDUVROQ.
The generic ingredient in JESDUVROQ is daprodustat. One supplier is listed for this generic product. Additional details are available on the daprodustat profile page.
When can EXFORGE HCT (amlodipine besylate; hydrochlorothiazide; valsartan) generic drug versions launch?
Generic name: amlodipine besylate; hydrochlorothiazide; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 27, 2026
Generic Entry Controlled by: Hong Kong Patent 1,133,818

This drug has forty-four patent family members in thirty-one countries. There has been litigation on patents covering EXFORGE HCT
See drug price trends for EXFORGE HCT.
The generic ingredient in EXFORGE HCT is amlodipine besylate; hydrochlorothiazide; valsartan. There are fifty drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the amlodipine besylate; hydrochlorothiazide; valsartan profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Hong Kong Patent 1,127,359

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Hong Kong Patent 1,127,359

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: Hong Kong Patent 1,127,359

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Hong Kong Patent 1,126,485
Patent Title: 藥動性質治療調節器 (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Hong Kong Patent 1,137,451
Patent Title: 藥動性質治療的調節器 (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Hong Kong Patent 1,143,144
Patent Title: MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: Hong Kong Patent 1,232,217
Patent Title: 治療劑的藥代動力學特性調節劑 (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can BYFAVO (remimazolam besylate) generic drug versions launch?
Generic name: remimazolam besylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 10, 2026
Generic Entry Controlled by: Hong Kong Patent 1,137,739
Patent Title: 短效苯並二氮雜噁鹽及其多晶型 (SHORT-ACTING BENZODIAZEPINE SALTS AND THEIR POLYMORPHIC FORMS)
BYFAVO is a drug marketed by Acacia. There are eleven patents protecting this drug.
This drug has fifty-three patent family members in twenty-four countries. There has been litigation on patents covering BYFAVO
See drug price trends for BYFAVO.
The generic ingredient in BYFAVO is remimazolam besylate. One supplier is listed for this generic product. Additional details are available on the remimazolam besylate profile page.
When can BYFAVO (remimazolam besylate) generic drug versions launch?
Generic name: remimazolam besylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 10, 2026
Generic Entry Controlled by: Hong Kong Patent 1,189,383
Patent Title: 短效苯並二氮雜䓬鹽及其多晶型 (SHORT-ACTING BENZODIAZEPINE SALTS AND THEIR POLYMORPHIC FORMS)
BYFAVO is a drug marketed by Acacia. There are eleven patents protecting this drug.
This drug has fifty-three patent family members in twenty-four countries. There has been litigation on patents covering BYFAVO
See drug price trends for BYFAVO.
The generic ingredient in BYFAVO is remimazolam besylate. One supplier is listed for this generic product. Additional details are available on the remimazolam besylate profile page.
When can BYFAVO (remimazolam besylate) generic drug versions launch?
Generic name: remimazolam besylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 10, 2026
Generic Entry Controlled by: Hong Kong Patent 1,202,113
Patent Title: 短效苯並二氮雜䓬鹽及其多晶型 (SHORT-ACTING BENZODIAZEPINE SALTS AND THEIR POLYMORPHIC FORMS)
BYFAVO is a drug marketed by Acacia. There are eleven patents protecting this drug.
This drug has fifty-three patent family members in twenty-four countries. There has been litigation on patents covering BYFAVO
See drug price trends for BYFAVO.
The generic ingredient in BYFAVO is remimazolam besylate. One supplier is listed for this generic product. Additional details are available on the remimazolam besylate profile page.
When can BYFAVO (remimazolam besylate) generic drug versions launch?
Generic name: remimazolam besylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 10, 2026
Generic Entry Controlled by: Hong Kong Patent 1,202,114
Patent Title: 短效苯並二氮雜䓬鹽及其多晶型 (SHORT-ACTING BENZODIAZEPINE SALTS AND THEIR POLYMORPHIC FORMS)
BYFAVO is a drug marketed by Acacia. There are eleven patents protecting this drug.
This drug has fifty-three patent family members in twenty-four countries. There has been litigation on patents covering BYFAVO
See drug price trends for BYFAVO.
The generic ingredient in BYFAVO is remimazolam besylate. One supplier is listed for this generic product. Additional details are available on the remimazolam besylate profile page.
When can INVOKAMET XR (canagliflozin; metformin hydrochloride) generic drug versions launch?
Generic name: canagliflozin; metformin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 31, 2026
Generic Entry Controlled by: Hong Kong Patent 1,086,277
Patent Title: NOVEL COMPOUNDS HAVING INHIBITORY ACTIVITY AGAINST SODIUM-DEPENDANT TRANSPORTER
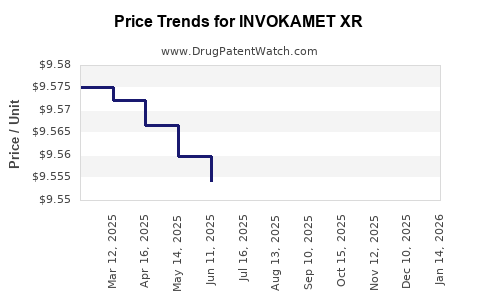
This drug has two hundred and twenty patent family members in forty-five countries. There has been litigation on patents covering INVOKAMET XR
See drug price trends for INVOKAMET XR.
The generic ingredient in INVOKAMET XR is canagliflozin; metformin hydrochloride. There are twenty-one drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the canagliflozin; metformin hydrochloride profile page.
When can INVOKANA (canagliflozin) generic drug versions launch?
Generic name: canagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 31, 2026
Generic Entry Controlled by: Hong Kong Patent 1,086,277
Patent Title: NOVEL COMPOUNDS HAVING INHIBITORY ACTIVITY AGAINST SODIUM-DEPENDANT TRANSPORTER
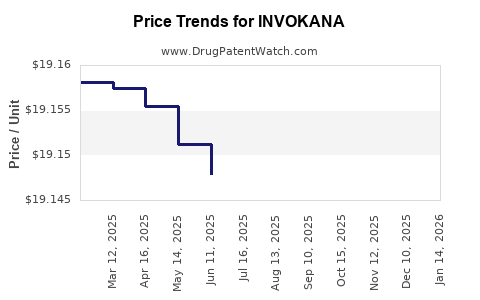
This drug has two hundred and twenty patent family members in forty-five countries. There has been litigation on patents covering INVOKANA
See drug price trends for INVOKANA.
The generic ingredient in INVOKANA is canagliflozin. There are twenty-one drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the canagliflozin profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: Hong Kong Patent 1,126,486

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: Hong Kong Patent 1,201,535

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 15, 2026
Generic Entry Controlled by: Hong Kong Patent 1,119,559
Patent Title: PANCREATIN MICROPELLET CORES SUITABLE FOR ENTERIC COATING
CREON is a drug marketed by
This drug has ninety-three patent family members in thirty-one countries.
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 29, 2026
Generic Entry Controlled by: Hong Kong Patent 1,133,597
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the macitentan profile page.
When can LIVDELZI (seladelpar lysine) generic drug versions launch?
Generic name: seladelpar lysine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 13, 2026
Generic Entry Controlled by: Hong Kong Patent 1,122,699
Patent Title: 苯氧基烷基 硫代 -苯氧基乙酸衍生物的新型賴氨酸鹽 (NOVEL LYSINE SALTS OF 4-((PHENOXYALKYL) THIO)-PHENOXYACETIC ACID DERIVATIVES 4-(())-)
LIVDELZI is a drug marketed by Gilead Sciences Inc. There are six patents protecting this drug.
This drug has one hundred and thirty-eight patent family members in forty-six countries.
The generic ingredient in LIVDELZI is seladelpar lysine. One supplier is listed for this generic product. Additional details are available on the seladelpar lysine profile page.
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 22, 2026
Generic Entry Controlled by: Hong Kong Patent 1,117,762
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can TYMLOS (abaloparatide) generic drug versions launch?
Generic name: abaloparatide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 03, 2026
Generic Entry Controlled by: Hong Kong Patent 1,214,181
Patent Title: 包含 的穩定組合物及其用途 (A STABLE COMPOSITION COMPRISING PTHRP AND USES THEREOF PTHRP)

This drug has fifty-six patent family members in twenty-nine countries. There has been litigation on patents covering TYMLOS
See drug price trends for TYMLOS.
The generic ingredient in TYMLOS is abaloparatide. One supplier is listed for this generic product. Additional details are available on the abaloparatide profile page.
When can INREBIC (fedratinib hydrochloride) generic drug versions launch?
Generic name: fedratinib hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: Hong Kong Patent 1,121,148
INREBIC is a drug marketed by Bristol-myers. There are five patents protecting this drug.
This drug has one hundred and nineteen patent family members in forty-two countries. There has been litigation on patents covering INREBIC
See drug price trends for INREBIC.
The generic ingredient in INREBIC is fedratinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the fedratinib hydrochloride profile page.
When can BEVYXXA (betrixaban) generic drug versions launch?
Generic name: betrixaban
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 07, 2026
Generic Entry Controlled by: Hong Kong Patent 1,115,131
Patent Title: PHARMACEUTICAL SALTS AND POLYMORPHS OF N-(5-CHLORO-2-PYRIDINYL)-2-[[4- [(DIMETHYLAMINO)IMINOMETHYL]BENZOYL]AMINO]-5-METHOXY-BENZAMIDE , A FACTOR XA INHIBITOR
BEVYXXA is a drug marketed by Portola Pharms Inc. There are five patents protecting this drug.
This drug has sixty-seven patent family members in twenty-six countries. There has been litigation on patents covering BEVYXXA
See drug price trends for BEVYXXA.
The generic ingredient in BEVYXXA is betrixaban. Additional details are available on the betrixaban profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 05, 2026
Generic Entry Controlled by: Hong Kong Patent 1,214,513
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: Hong Kong Patent 1,134,290
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can LETAIRIS (ambrisentan) generic drug versions launch?
Generic name: ambrisentan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 12, 2026
Generic Entry Controlled by: Hong Kong Patent 1,218,393
Patent Title: 用於治療肺高壓的複合物 (COMPOSITION FOR TREATING A PULMONARY HYPERTENSION)
LETAIRIS is a drug marketed by Gilead. There are three patents protecting this drug and one Paragraph IV challenge.
This drug has fifty-one patent family members in twenty-six countries. There has been litigation on patents covering LETAIRIS
See drug price trends for LETAIRIS.
The generic ingredient in LETAIRIS is ambrisentan. There are nine drug master file entries for this API. Thirteen suppliers are listed for this generic product. Additional details are available on the ambrisentan profile page.
When can VYKAT XR (diazoxide choline) generic drug versions launch?
Generic name: diazoxide choline
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 20, 2026
Generic Entry Controlled by: Hong Kong Patent 1,117,078
Patent Title: SALTS OF POTASSIUM ATP CHANNEL OPENERS AND USES THEREOF
VYKAT XR is a drug marketed by Soleno Therap. There are six patents protecting this drug.
This drug has seventy-eight patent family members in twenty-two countries.
The generic ingredient in VYKAT XR is diazoxide choline. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the diazoxide choline profile page.
When can OSENI (alogliptin benzoate; pioglitazone hydrochloride) generic drug versions launch?
Generic name: alogliptin benzoate; pioglitazone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 01, 2027
Generic Entry Controlled by: Hong Kong Patent 1,138,188

This drug has one hundred and one patent family members in forty-two countries. There has been litigation on patents covering OSENI
See drug price trends for OSENI.
The generic ingredient in OSENI is alogliptin benzoate; pioglitazone hydrochloride. There are ten drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate; pioglitazone hydrochloride profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Hong Kong Patent 1,142,818
PANCREAZE is a drug marketed by
This drug has one hundred and one patent family members in forty-two countries.
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Hong Kong Patent 1,142,818
ZENPEP is a drug marketed by
This drug has one hundred and one patent family members in forty-two countries.
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can VANFLYTA (quizartinib dihydrochloride) generic drug versions launch?
Generic name: quizartinib dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 16, 2027
Generic Entry Controlled by: Hong Kong Patent 1,131,141
VANFLYTA is a drug marketed by Daiichi Sankyo Inc. There are eleven patents protecting this drug.
This drug has one hundred and twenty-six patent family members in thirty-two countries.
See drug price trends for VANFLYTA.
The generic ingredient in VANFLYTA is quizartinib dihydrochloride. One supplier is listed for this generic product. Additional details are available on the quizartinib dihydrochloride profile page.
When can XENLETA (lefamulin acetate) generic drug versions launch?
Generic name: lefamulin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 20, 2027
Generic Entry Controlled by: Hong Kong Patent 1,140,185
XENLETA is a drug marketed by Hong Kong. There are four patents protecting this drug.
This drug has one hundred and twenty patent family members in thirty-six countries. There has been litigation on patents covering XENLETA
See drug price trends for XENLETA.
The generic ingredient in XENLETA is lefamulin acetate. Additional details are available on the lefamulin acetate profile page.
When can INLYTA (axitinib) generic drug versions launch?
Generic name: axitinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 05, 2027
Generic Entry Controlled by: Hong Kong Patent 1,193,405
Patent Title: 適用於治療哺乳動物異常細胞生長的 甲基-氨甲酰基 苯基硫基 吡啶- -基 乙烯基 吲唑的晶型 (CRYSTALLINE FORMS OF 6-[2-(METHYLCARBAMOYL)PHENYLSULFANYL]-3-E-[2-(PYRIDIN- 2-YL)ETHENYL]INDAZOLE SUITABLE FOR THE TREATMENT OF ABNORMAL CELL GROWTH IN MAMMALS 6-[2-(-)]-3-E-[2-(- 2-)])
INLYTA is a drug marketed by Pf Prism Cv. There are four patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has ninety-seven patent family members in thirty countries. There has been litigation on patents covering INLYTA
See drug price trends for INLYTA.
The generic ingredient in INLYTA is axitinib. There are four drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the axitinib profile page.
When can QVAR REDIHALER (beclomethasone dipropionate) generic drug versions launch?
Generic name: beclomethasone dipropionate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 11, 2027
Generic Entry Controlled by: Hong Kong Patent 1,141,349
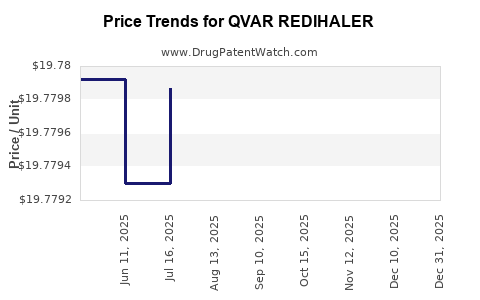
This drug has two hundred and seventy-eight patent family members in twenty-seven countries. There has been litigation on patents covering QVAR REDIHALER
See drug price trends for QVAR REDIHALER.
The generic ingredient in QVAR REDIHALER is beclomethasone dipropionate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the beclomethasone dipropionate profile page.
When can TRELSTAR (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 06, 2027
Generic Entry Controlled by: Hong Kong Patent 1,141,737
Patent Title: SLOW RELEASE PHARMACEUTICAL COMPOSITION MADE OF MICROPARTICLES
TRELSTAR is a drug marketed by Verity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
See drug price trends for TRELSTAR.
The generic ingredient in TRELSTAR is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can TRIPTODUR KIT (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 06, 2027
Generic Entry Controlled by: Hong Kong Patent 1,141,737
Patent Title: SLOW RELEASE PHARMACEUTICAL COMPOSITION MADE OF MICROPARTICLES
TRIPTODUR KIT is a drug marketed by Azurity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
The generic ingredient in TRIPTODUR KIT is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can JAKAFI (ruxolitinib phosphate) generic drug versions launch?
Generic name: ruxolitinib phosphate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 13, 2027
Generic Entry Controlled by: Hong Kong Patent 1,198,652
Patent Title: 激酶抑制劑 -吡咯 嘧啶- -基 -吡唑- -基 -環戊基丙腈的結晶鹽 (CRYSTALLINE SALTS OF THE JANUS KINASE INHIBITOR (R)-3-(4-(7H-PYRROLO[2,3- D]PYRIMIDIN-4-YL)-1H-PYRAZOL-1-YL)-3-CYCLOPENTYLPROPANENITRILE JANUS (R)-3-(4-(7H-[23-D]-4-)-1H--1-)-3-)
JAKAFI is a drug marketed by Incyte Corp. There are eight patents protecting this drug and one Paragraph IV challenge.
This drug has two hundred and thirty-six patent family members in forty-six countries. There has been litigation on patents covering JAKAFI
See drug price trends for JAKAFI.
The generic ingredient in JAKAFI is ruxolitinib phosphate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ruxolitinib phosphate profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 15, 2027
Generic Entry Controlled by: Hong Kong Patent 1,134,483
Patent Title: 1- [2- (2, 4-DIMETHYLPHENYLSULFANYL)-PHENYL] PIPERAZINE AS A COMPOUND WITH COMBINED SEROTONIN REUPTAKE, 5-HT3 AND 5-HT1A ACTIVITY FOR THE TREATMENT OF COGNITIVE IMPAIRMENT

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2027
Generic Entry Controlled by: Hong Kong Patent 1,165,807
Patent Title: 合成的胆汁酸製備方法 (SYNTHETIC BILE ACID PREPARATION)
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can TWIRLA (ethinyl estradiol; levonorgestrel) generic drug versions launch?
Generic name: ethinyl estradiol; levonorgestrel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 10, 2027
Generic Entry Controlled by: Hong Kong Patent 1,142,798
Patent Title: 具有原位密封的皮膚遞送裝置 (DERMAL DELIVERY DEVICE WITH IN SITU SEAL)
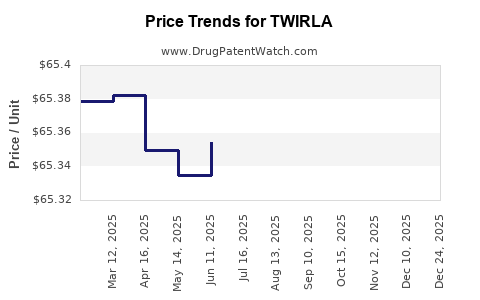
This drug has nineteen patent family members in twelve countries.
See drug price trends for TWIRLA.
The generic ingredient in TWIRLA is ethinyl estradiol; levonorgestrel. There are twenty-six drug master file entries for this API. Twenty-three suppliers are listed for this generic product. Additional details are available on the ethinyl estradiol; levonorgestrel profile page.
Hong Kong Branded and Generic Drug Markets: Assessment and Regulatory Opportunities and Challenges
More… ↓
DrugPatentWatch cited by CNN, NEJM, Nature Journals, and more …

Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
