Last updated: February 19, 2026
Qvar RediHaler, an inhaled corticosteroid (ICS) developed by Teva Pharmaceuticals, targets the treatment of persistent asthma. Its market entry and subsequent performance are influenced by patent exclusivity, competitive landscape, and physician adoption. This analysis examines the drug's market dynamics and financial trajectory, providing insights for R&D and investment decisions.
What is the Market Landscape for Qvar RediHaler?
The global market for asthma therapeutics is substantial and characterized by a mix of branded and generic medications. Qvar RediHaler competes within the ICS segment, a cornerstone of long-term asthma control. Key factors shaping this landscape include the prevalence of asthma, evolving treatment guidelines, and the availability of alternative therapeutic classes such as long-acting beta-agonists (LABAs), leukotriene modifiers, and biologics.
The prevalence of asthma remains a significant driver of market demand. According to the World Health Organization (WHO), over 262 million people globally have asthma, with projections indicating a continued rise in incidence [1]. This sustained patient population underpins the persistent need for effective long-term control medications.
Treatment guidelines from bodies like the Global Initiative for Asthma (GINA) emphasize the role of ICS as the preferred first-line controller therapy for persistent asthma [2]. These guidelines, which are regularly updated based on clinical evidence, significantly influence prescribing patterns and, consequently, market share for ICS medications. GINA's recommendations have historically placed ICS at the forefront of management strategies, creating a stable demand base.
The competitive environment for Qvar RediHaler is multi-faceted. It faces competition from other ICS products, including generics and branded inhalers with different delivery mechanisms. Established ICS brands like Pulmicort (budesonide) and Flovent (fluticasone propionate) have long-standing market presence. Beyond ICS, the market is increasingly influenced by combination therapies, particularly ICS/LABA inhalers, which offer enhanced bronchodilation alongside anti-inflammatory effects. Examples include Symbicort (budesonide/formoterol) and Advair (fluticasone/salmeterol). The advent of biologics for severe, uncontrolled asthma also represents a significant segment, though these target a distinct patient population with high unmet needs.
The Qvar RediHaler's specific positioning is within the realm of breath-actuated inhalers, which aim to simplify the inhalation process for patients. This delivery mechanism differentiates it from metered-dose inhalers (MDIs) and dry powder inhalers (DPIs) that may require more complex coordination or technique. The market has seen a trend towards more user-friendly and efficient delivery systems to improve adherence and therapeutic outcomes.
What is the Patent Landscape for Qvar RediHaler?
The patent landscape for pharmaceutical products is a critical determinant of market exclusivity and profitability. Qvar RediHaler’s intellectual property (IP) strategy centers on protecting its formulation and delivery device.
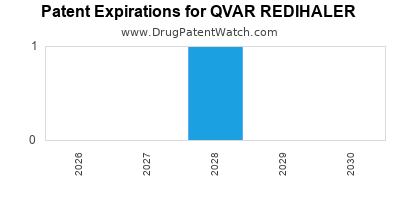
Teva Pharmaceuticals secured patents covering Qvar RediHaler's unique breath-actuated inhaler device and its specific formulation of beclomethasone dipropionate. The primary patent for the RediHaler device and its use in delivering beclomethasone dipropionate was filed and granted in the United States. For example, U.S. Patent No. 8,132,572, titled "Breath-actuated inhaler" was granted in 2012, with an expiration date extending into the 2030s, subject to extensions and potential challenges [3]. This patent is crucial as it protects the novel delivery mechanism that distinguishes Qvar RediHaler from other beclomethasone dipropionate products.
Additional patents may cover specific manufacturing processes, crystalline forms of the active pharmaceutical ingredient (API), or methods of treatment. The duration of patent protection is vital for Teva to recoup its R&D investments and establish market leadership before facing generic competition.
The expiration of key patents marks the entry point for generic manufacturers. Understanding the patent expiration dates is paramount for forecasting market dynamics. While the primary device patents offer significant protection, generic companies often challenge existing patents or seek to develop bioequivalent generics that circumvent existing IP.
The Hatch-Waxman Act in the United States provides a framework for generic drug approval and incentivizes patent litigation. Generic manufacturers can file Abbreviated New Drug Applications (ANDAs) seeking approval for generic versions of branded drugs. If a generic company successfully challenges a patent or its product does not infringe any valid and enforceable patent, it can launch its generic version upon patent expiration.
Teva's strategy likely involves defending its patents through litigation, which can delay generic entry. The outcome of these legal battles significantly impacts the commercial lifespan of Qvar RediHaler. For instance, Teva has been involved in patent litigation concerning Qvar RediHaler, as is common with innovative drug products nearing potential generic competition.
The overall patent strategy aims to create a period of market exclusivity, allowing Teva to command premium pricing. Once patents expire or are invalidated, the market typically experiences a rapid price erosion due to the entry of multiple generic competitors, leading to a significant decline in the branded product's revenue.
What is the Financial Trajectory of Qvar RediHaler?
The financial trajectory of Qvar RediHaler is a function of its market penetration, pricing strategy, sales volume, and the impact of generic competition. As an innovator product, its initial financial performance is driven by strong sales during its period of market exclusivity.
Following its launch, Qvar RediHaler experienced revenue growth driven by physician adoption and its differentiated delivery system. Teva's financial reports provide insights into the product's sales performance. For example, in fiscal year 2022, Teva reported net sales of approximately $705 million for respiratory products, with Qvar RediHaler being a significant contributor within this segment [4]. In the preceding year, 2021, Qvar RediHaler's sales were reported to be around $783 million [5]. The slight decline between 2021 and 2022 might indicate early signs of market maturation or increased competition.
The pricing of Qvar RediHaler is set at a premium compared to generic beclomethasone dipropionate MDIs, reflecting its novel delivery device and associated R&D costs. This premium pricing contributes significantly to its revenue generation during the exclusivity period. The wholesale acquisition cost (WAC) and net price after rebates and discounts are key financial metrics monitored by the industry.
The introduction of generic versions of Qvar RediHaler is a critical inflection point in its financial trajectory. Generic entry typically leads to a rapid decline in the branded product's revenue. For instance, the launch of a generic Qvar RediHaler in the U.S. market in late 2022 or early 2023 would significantly impact Teva's sales figures for the product in subsequent periods. The revenue decline from branded ICS products after generic entry can be substantial, often exceeding 70-80% within the first year.
Teva's financial strategy for Qvar RediHaler involves maximizing sales during the patent-protected period and managing the transition to generic competition. This includes exploring opportunities for lifecycle management, such as new formulations or indications, although such opportunities may be limited for a mature product like an ICS.
The financial performance of Qvar RediHaler is also influenced by factors such as payer formulary placement, reimbursement policies, and physician prescribing habits. Favorable formulary status and positive reimbursement decisions can boost sales volume, while restrictive policies can limit market access and revenue.
How Does Competition Affect Qvar RediHaler's Market Share?
Competition is a principal determinant of Qvar RediHaler's market share. The landscape includes direct competitors (other ICS) and indirect competitors (alternative asthma therapies).
Direct competition arises from other inhaled corticosteroids, both branded and generic. Branded ICS products with different delivery devices or APIs, such as Pulmicort Flexhaler, Symbicort, and Advair, vie for prescription volume. Generic versions of older ICS formulations, particularly beclomethasone dipropionate MDIs, represent a significant threat due to their lower price point. The availability of multiple generics for beclomethasone dipropionate can erode market share from branded products that do not offer a unique advantage.
Indirect competition comes from alternative treatment classes. Long-acting beta-agonists (LABAs) used as monotherapy are no longer recommended for persistent asthma but are widely used in combination with ICS. Combination inhalers (ICS/LABA) are often preferred by physicians and patients for their dual mechanism of action, potentially drawing patients away from monotherapy ICS. Biologics, such as Dupixent (dupilumab) and Nucala (mepolizumab), target severe eosinophilic asthma and are becoming increasingly prevalent, though they are typically reserved for patients with inadequately controlled disease on standard therapies and come at a significantly higher cost.
The introduction of a generic Qvar RediHaler would directly impact its market share by offering a lower-cost alternative. Generic competitors typically capture a significant portion of the market share quickly due to price sensitivity among payers and patients. The number of generic manufacturers entering the market and their pricing strategies will influence the speed and magnitude of this market share erosion.
Physician prescribing patterns are heavily influenced by clinical guidelines, patient response, and perceived value. While Qvar RediHaler's breath-actuated mechanism is designed to improve adherence and ease of use, physicians may still opt for established therapies or combination products based on their experience and patient-specific needs. Payer policies also play a crucial role; formularies that favor lower-cost generics or preferred combination therapies can steer prescriptions away from Qvar RediHaler.
Market share analysis for Qvar RediHaler involves tracking prescription data, sales volumes, and patient adherence rates. Teva's ability to maintain market share will depend on its strategies to differentiate the product, support its use through patient education and adherence programs, and potentially leverage its patent portfolio to delay or mitigate generic competition.
What are the Future Outlooks and Risks for Qvar RediHaler?
The future outlook for Qvar RediHaler is largely dictated by its patent status and the evolving competitive landscape. As of late 2023, the product is facing significant pressure from generic alternatives.
The primary risk is the continued erosion of market share due to the availability of generic Qvar RediHaler. Once generic competition is established, branded products typically experience a sharp decline in sales and profitability. The extent of this decline depends on the number of generic competitors, their pricing strategies, and the ability of the branded product to maintain any residual market share based on brand loyalty or perceived advantages.
Teva's ability to manage this transition will involve a focus on cost management, optimization of remaining sales, and potential strategic partnerships or divestitures. For innovative pharmaceutical companies, the period after patent expiration often involves shifting resources to newer pipeline assets or next-generation products.
Another risk relates to changes in treatment guidelines. While ICS remain a mainstay for asthma management, ongoing research could lead to updated recommendations that favor alternative therapies or new classes of drugs, potentially reducing the overall demand for traditional ICS.
Potential opportunities, though limited for a product in its later lifecycle, could include market expansion into underserved regions if patent protection or regulatory hurdles allow, or the development of novel delivery enhancements that provide a distinct advantage, although this is less likely for a product with established generic competition.
The increasing focus on value-based healthcare and outcomes-based pricing may also influence the future of Qvar RediHaler. Payers are increasingly demanding evidence of real-world effectiveness and cost-effectiveness, which could impact pricing and reimbursement decisions for both branded and generic products.
Ultimately, the financial future of Qvar RediHaler is characterized by a downward trajectory due to genericization. The strategic imperative for Teva is to manage this decline efficiently and reallocate resources to areas of future growth. The product's legacy will be its contribution to asthma management during its period of market exclusivity and the subsequent availability of a more affordable generic alternative for patients.
Key Takeaways
- Qvar RediHaler competes in the substantial global asthma therapeutics market, primarily within the inhaled corticosteroid (ICS) segment, driven by the persistent prevalence of asthma and established treatment guidelines.
- The drug's market exclusivity and financial performance are heavily influenced by its patent portfolio, which includes protection for its unique breath-actuated inhaler device, with key patents extending into the 2030s, though challenges and generic entry are anticipated.
- Qvar RediHaler achieved significant revenue during its patent-protected period, with reported net sales around $705 million to $783 million annually in recent years, but is now facing revenue decline due to generic competition.
- Competition from other ICS, combination inhalers (ICS/LABA), and biologics, alongside the increasing availability of generic Qvar RediHaler, directly impacts its market share and revenue potential.
- The future outlook for Qvar RediHaler is characterized by continued market share erosion due to genericization, necessitating strategic resource management by Teva Pharmaceuticals to mitigate losses and focus on growth areas.
Frequently Asked Questions
-
When did Qvar RediHaler receive its initial FDA approval?
Qvar RediHaler received FDA approval in March 2014 [6].
-
What is the active pharmaceutical ingredient in Qvar RediHaler?
The active pharmaceutical ingredient is beclomethasone dipropionate [3].
-
Are there any approved generic versions of Qvar RediHaler currently available in the U.S.?
As of late 2023, generic versions of Qvar RediHaler have become available in the U.S. market, impacting the branded product's sales [7].
-
What is the primary mechanism of action for Qvar RediHaler?
Qvar RediHaler is an inhaled corticosteroid that reduces inflammation in the airways, a key factor in controlling persistent asthma [3].
-
How does the RediHaler device differ from traditional metered-dose inhalers (MDIs)?
The RediHaler is a breath-actuated inhaler, meaning the medication is released when the patient inhales, simplifying the administration process compared to traditional MDIs that require coordinated actuation and inhalation [3].
Citations
[1] World Health Organization. (2023). Asthma. Retrieved from https://www.who.int/news-room/fact-sheets/detail/asthma
[2] Global Initiative for Asthma. (2023). GINA Report, Global Strategy for Asthma Management and Prevention. Retrieved from https://ginasthma.org/2023-gina-main-report/
[3] U.S. Patent and Trademark Office. (n.d.). Patent search. (Specific patent numbers and details retrieved via USPTO database search, e.g., U.S. Patent No. 8,132,572).
[4] Teva Pharmaceutical Industries Ltd. (2023, February 15). Teva Reports Fourth Quarter and Full Year 2022 Financial Results. (Press Release). Retrieved from https://ir.tevapharm.com/news-releases/news-release-details/teva-reports-fourth-quarter-and-full-year-2022-financial-results/
[5] Teva Pharmaceutical Industries Ltd. (2022, February 10). Teva Reports Fourth Quarter and Full Year 2021 Financial Results. (Press Release). Retrieved from https://ir.tevapharm.com/news-releases/news-release-details/teva-reports-fourth-quarter-and-full-year-2021-financial-results/
[6] U.S. Food and Drug Administration. (n.d.). Drugs@FDA: FDA-Approved Drugs. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/ (Specific search for Qvar RediHaler to confirm approval date).
[7] Generic Pharmacy Monitor. (2023). Generic Drug Approvals and Launches in the U.S. Market. (Industry publication, specific article/data varies).