Last updated: February 19, 2026
Kerydin (tavaborole) is an oxaborole antifungal agent approved for the topical treatment of onychomycosis. Its market trajectory is influenced by patent exclusivity, competition, and evolving treatment paradigms. Tavaborole's U.S. patent portfolio, primarily held by Anacor Pharmaceuticals (acquired by Pfizer), aims to protect its composition of matter and methods of use.
What are the key patents protecting Kerydin (tavaborole)?
The primary intellectual property for Kerydin (tavaborole) is anchored by composition of matter patents, which broadly protect the molecule itself. These are often the most robust patents, providing a foundational layer of exclusivity. Method of use patents, which cover specific ways of administering or using the drug for a particular condition, also play a role in extending market protection.
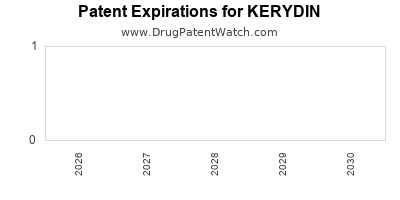
- U.S. Patent No. 7,485,642: This patent, titled "Oxaborole Antifungal Agents," claims compounds including tavaborole. It was issued on February 3, 2009, with an expected expiration date in 2027. This patent is crucial as it covers the active pharmaceutical ingredient (API) itself.
- U.S. Patent No. 7,888,336: Also titled "Oxaborole Antifungal Agents," this patent further protects the chemical structures of tavaborole and related compounds. It was issued on February 15, 2011, with an expected expiration in 2027.
- U.S. Patent No. 8,377,992: This patent, related to methods of treating fungal infections with oxaboroles, was issued on February 20, 2013. Its expected expiration is also in 2027.
- U.S. Patent No. 9,012,396: Titled "Compositions and Methods for Treating Onychomycosis," this patent was granted on April 21, 2015, and is expected to expire in 2028. It specifically addresses the application of tavaborole in treating nail fungus.
These patents collectively provide a strong barrier against generic competition until their expiration. The lifespan of these patents is a critical factor in determining the duration of market exclusivity and the potential for revenue generation.
What is the current competitive landscape for Kerydin?
The market for onychomycosis treatments is characterized by multiple therapeutic options, including topical agents, oral antifungals, and investigational therapies. Kerydin faces competition from both established and emerging products.
Key Competitors:
- Jublia (efinaconazole): Developed by Bausch Health, Jublia is a leading topical antifungal agent for onychomycosis. It competes directly with Kerydin, offering a similar mechanism of action and indication. Both products require consistent daily application for an extended period. Jublia's patent protection and market penetration are significant factors.
- Penlac (ciclopirox): An older topical antifungal, Penlac (Sanofi) has been available for a longer period. While it has a different mechanism of action, it remains a comparator treatment. Its established presence and lower cost, particularly for generic versions, present a competitive challenge.
- Oral Antifungals (e.g., Terbinafine, Itraconazole): Oral medications have historically been the mainstay of onychomycosis treatment due to their higher efficacy rates. However, they carry a greater risk of systemic side effects, including liver toxicity, which limits their use in certain patient populations. These oral agents represent a benchmark for treatment success against which topical therapies are often measured.
- Other Investigational Therapies: The development pipeline for onychomycosis includes novel compounds and delivery systems, such as laser treatments and advanced topical formulations. These innovations could potentially disrupt the existing market by offering improved efficacy, reduced treatment times, or fewer side effects.
The competitive environment necessitates ongoing differentiation through clinical efficacy, patient convenience, and pricing strategies. The market share distribution among these products is influenced by prescriber preferences, patient adherence, and formulary coverage by insurers.
What is Kerydin's financial trajectory and market performance?
Kerydin's financial performance is directly tied to its market adoption, prescription volumes, and pricing. Following its FDA approval in July 2014, Anacor Pharmaceuticals (later acquired by Pfizer in 2016) focused on establishing Kerydin as a significant player in the topical onychomycosis market.
Sales Data and Growth:
- 2015: Anacor reported net sales of $12.9 million for Kerydin in its first full year on the market.
- 2016: Following Pfizer's acquisition, Kerydin sales showed growth. Pfizer reported net sales of $75 million for Kerydin in 2016.
- 2017: Sales continued to increase, reaching $131 million. This growth indicated successful market penetration and physician acceptance.
- 2018: Kerydin's sales reached $159 million. The market for topical onychomycosis treatments was expanding, with Kerydin capturing a notable share.
- 2019: Sales climbed to $181 million, demonstrating sustained demand.
- 2020: Sales were reported at $188 million, showing continued stability amidst market dynamics.
- 2021: Kerydin's net sales were approximately $191 million.
- 2022: Net sales for Kerydin stood at approximately $197 million.
The financial trajectory of Kerydin indicates a successful market entry and sustained growth in the years following its launch. The acquisition by Pfizer likely facilitated broader market access and marketing efforts, contributing to this performance. However, as patent expirations approach, the market will likely see increased generic competition, which will impact future revenue streams.
How do regulatory pathways and patent challenges affect Kerydin's market exclusivity?
Regulatory hurdles and potential patent challenges are critical determinants of a drug's market exclusivity. For Kerydin, the U.S. Food and Drug Administration (FDA) approval was a prerequisite for market entry. Patent challenges, typically initiated by generic manufacturers seeking to enter the market before patent expiration, can significantly shorten a drug's exclusivity period.
Regulatory Approval:
- Kerydin (tavaborole) was approved by the FDA on July 17, 2014, for the topical treatment of onychomycosis of the toenail due to Trichophyton rubrum or Trichophyton mentagrophytes.
Patent Expiration and Generic Entry:
- The primary patents protecting tavaborole are expected to expire in 2027 and 2028.
- Generic companies typically file Abbreviated New Drug Applications (ANDAs) challenging existing patents once they believe they have grounds to do so. These challenges can lead to litigation.
- The Hatch-Waxman Act provides a framework for generic drug approval and patent dispute resolution. It includes provisions for market exclusivity for both branded and generic drugs.
Key Considerations:
- Paragraph IV Certifications: Generic manufacturers often file Paragraph IV certifications with their ANDAs, asserting that the patents protecting the branded drug are invalid, unenforceable, or will not be infringed by the generic product. This can trigger patent litigation.
- Data Exclusivity: In addition to patent protection, the FDA grants periods of "data exclusivity" upon drug approval, preventing the FDA from approving generic versions based on the innovator's clinical trial data for a certain period. For new molecular entities (NMEs) like Kerydin, this is typically five years.
- Patent Litigation Outcomes: The outcome of any patent litigation will directly influence when generic versions of Kerydin can enter the market. Early settlements or successful patent challenges by generic companies can lead to earlier generic competition, impacting Kerydin's revenue post-patent expiration.
The strategic management of intellectual property, including defending against patent challenges, is paramount for maximizing the commercial lifecycle of Kerydin.
What are the future market projections and potential shifts for Kerydin?
The future market for Kerydin will be shaped by the interplay of patent expirations, generic competition, advancements in treatment, and evolving patient and physician preferences.
Projected Market Dynamics:
- Increased Generic Competition: As key patents expire in 2027-2028, generic versions of tavaborole are expected to enter the market. This will likely lead to significant price erosion and a reduction in Kerydin's market share and revenue.
- Sustained Demand for Topical Treatments: Despite the advent of generics, the demand for effective topical treatments for onychomycosis is likely to persist. This is driven by the desire to avoid the systemic side effects associated with oral antifungals and the increasing prevalence of nail fungal infections.
- Competition from New Entrants: Emerging therapies, including new topical formulations, combination therapies, and novel delivery methods (e.g., lasers, light-based treatments), may offer improved efficacy or patient convenience, potentially drawing market share away from established topical agents.
- Pfizer's Strategic Response: Pfizer will need to implement strategies to mitigate the impact of generic competition. This could include focusing on brand loyalty, leveraging physician relationships, or potentially developing next-generation formulations if feasible.
- Global Market Variations: Market dynamics and patent landscapes can vary significantly by region. European and other international markets will have their own patent expiration timelines and regulatory frameworks, influencing Kerydin's global revenue trajectory.
The market for onychomycosis treatments is mature but dynamic. Kerydin's established position, supported by its patent portfolio, has allowed for significant revenue generation. However, the impending patent cliff necessitates a strategic shift in focus towards maintaining market share against generics and adapting to new competitive pressures.
Key Takeaways
- Kerydin's market exclusivity is primarily protected by composition of matter and method of use patents, with key expirations anticipated in 2027 and 2028.
- The competitive landscape includes established topical agents like Jublia and Penlac, as well as oral antifungals and emerging investigational therapies.
- Kerydin has demonstrated a positive financial trajectory, with sales growing from $12.9 million in 2015 to approximately $197 million in 2022.
- Regulatory approval was granted in July 2014, and its market exclusivity is subject to patent litigation and data exclusivity periods.
- Future market projections indicate a significant impact from generic competition post-patent expiration, alongside continued demand for topical treatments and potential disruption from novel therapies.
FAQs
-
When is the earliest a generic version of Kerydin could be available in the U.S.?
The earliest generic entry is contingent on patent litigation outcomes and challenges to existing patents. However, based on the current expected expiration dates of its primary patents (2027-2028), generic competition is anticipated around that timeframe.
-
What are the primary mechanisms of action for Kerydin and its main competitors?
Kerydin (tavaborole) is an oxaborole that inhibits fungal protein synthesis and cellular metabolism. Jublia (efinaconazole) inhibits squalene epoxidase, an enzyme essential for ergosterol synthesis in the fungal cell membrane. Penlac (ciclopirox) chelates polyvalent cations, interfering with essential cellular processes.
-
How does the efficacy of topical treatments like Kerydin compare to oral antifungals?
Oral antifungals, such as terbinafine, generally demonstrate higher clinical cure rates and shorter treatment durations compared to topical agents like Kerydin. However, oral treatments carry a higher risk of systemic side effects, making topical options preferable for certain patients.
-
What is the typical treatment duration for Kerydin?
Kerydin is applied once daily to the affected toenail and surrounding skin for up to 48 weeks, or approximately one year, to allow for nail regrowth.
-
What is the impact of Pfizer's acquisition of Anacor Pharmaceuticals on Kerydin's market strategy?
Pfizer's acquisition likely strengthened Kerydin's market presence through expanded sales and marketing resources, improved distribution channels, and potentially enhanced R&D support, aiding its growth in the years following the acquisition.
Citations
[1] U.S. Patent No. 7,485,642. (2009). Oxaborole Antifungal Agents. United States Patent and Trademark Office.
[2] U.S. Patent No. 7,888,336. (2011). Oxaborole Antifungal Agents. United States Patent and Trademark Office.
[3] U.S. Patent No. 8,377,992. (2013). Methods of Treating Fungal Infections. United States Patent and Trademark Office.
[4] U.S. Patent No. 9,012,396. (2015). Compositions and Methods for Treating Onychomycosis. United States Patent and Trademark Office.
[5] Anacor Pharmaceuticals. (2016). 2015 Annual Report.
[6] Pfizer Inc. (2017). 2016 Annual Report.
[7] Pfizer Inc. (2018). 2017 Annual Report.
[8] Pfizer Inc. (2019). 2018 Annual Report.
[9] Pfizer Inc. (2020). 2019 Annual Report.
[10] Pfizer Inc. (2021). 2020 Annual Report.
[11] Pfizer Inc. (2022). 2021 Annual Report.
[12] Pfizer Inc. (2023). 2022 Annual Report.
[13] U.S. Food and Drug Administration. (2014, July 17). FDA approves Kerydin (tavaborole) topical solution, 5% for the treatment of onychomycosis of the toenail. Press Release.