Expiring Drug Patents Cheat Sheet
We analyse the patents covering drugs in 134 countries and quickly give you the likely loss-of-exclusivity/generic entry date
European Patent Office: These 69 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "European Patent Office: These 69 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027" DrugPatentWatch.com thinkBiotech, 2026 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
When can CONTRAVE (bupropion hydrochloride; naltrexone hydrochloride) generic drug versions launch?
Generic name: bupropion hydrochloride; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 05, 2026
Generic Entry Controlled by: European Patent Office Patent 2,061,448
Patent Title: FORMULATION À LIBÉRATION PROLONGÉE DE NALTRÉXONE (SUSTAINED RELEASE FORMULATION OF NALTREXONE)
CONTRAVE is a drug marketed by Nalpropion. There are eighteen patents protecting this drug and one Paragraph IV challenge.
This drug has two hundred and twenty-seven patent family members in forty-five countries. There has been litigation on patents covering CONTRAVE
See drug price trends for CONTRAVE.
The generic ingredient in CONTRAVE is bupropion hydrochloride; naltrexone hydrochloride. There are thirty-eight drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the bupropion hydrochloride; naltrexone hydrochloride profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 12, 2026
Generic Entry Controlled by: European Patent Office Patent 2,086,930
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 12, 2026
Generic Entry Controlled by: European Patent Office Patent 2,409,967
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 15, 2026
Generic Entry Controlled by: European Patent Office Patent 2,073,795
Patent Title: PRÉPARATION MÉDICAMENTEUSE EMPÊCHANT L'UTILISATION ABUSIVE (ABUSE RESISTANT DRUG FORMULATION)
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 15, 2026
Generic Entry Controlled by: European Patent Office Patent 2,200,593
Patent Title: FORMULATION MÉDICAMENTEUSE CONTRE L'ABUS (ABUSE RESISTANT DRUG FORMULATION)
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 15, 2026
Generic Entry Controlled by: European Patent Office Patent 2,692,341
Patent Title: Préparation médicamenteuse empêchant l'utilisation abusive (Abuse resistant drug formulation)
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 16, 2026
Generic Entry Controlled by: European Patent Office Patent 2,044,043
Patent Title: 1-[2-(2, 4-DIMETHYLPHENYLSULFANYL)-PHENYL]PIPERAZINE COMME COMPOSE PRESENTANT UNE ACTIVITE SUR LA SEROTONINE, 5-HT3 ET 5-HT1A POUR LE TRAITEMENT DU DEFICIT COGNITIF (1- Ý[- (2, 4-DIMETHYLPHENYLSULFANYL) -PHENYL]PIPERAZINE AS A COMPOUND WITH COMBINED SEROTONIN REUPTAKE, 5-HT3 AND 5-HT1A ACTIVITY FOR THE TREATMENT OF COGNITIVE IMPAIRMENT)

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 16, 2026
Generic Entry Controlled by: European Patent Office Patent 2,439,201
Patent Title: COMPOSÉS AVEC ACTIVITÉ COMBINÉ SERT, 5-HT3 ET 5-HT1A (COMPOUNDS WITH COMBINED SERT, 5-HT3 AND 5-HT1A ACTIVITY)

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: European Patent Office Patent 2,034,975
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: European Patent Office Patent 2,484,346
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: European Patent Office Patent 2,526,932
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: European Patent Office Patent 2,719,378
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: European Patent Office Patent 2,029,524

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: European Patent Office Patent 2,474,521

This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can JESDUVROQ (daprodustat) generic drug versions launch?
Generic name: daprodustat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: European Patent Office Patent 2,037,922
JESDUVROQ is a drug marketed by Glaxosmithkline. There are six patents protecting this drug.
This drug has forty-four patent family members in thirty-one countries.
See drug price trends for JESDUVROQ.
The generic ingredient in JESDUVROQ is daprodustat. One supplier is listed for this generic product. Additional details are available on the daprodustat profile page.
When can TEKTURNA HCT (aliskiren hemifumarate; hydrochlorothiazide) generic drug versions launch?
Generic name: aliskiren hemifumarate; hydrochlorothiazide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: European Patent Office Patent 2,034,968

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering TEKTURNA HCT
See drug price trends for TEKTURNA HCT.
The generic ingredient in TEKTURNA HCT is aliskiren hemifumarate; hydrochlorothiazide. There are four drug master file entries for this API. Additional details are available on the aliskiren hemifumarate; hydrochlorothiazide profile page.
When can TEKTURNA HCT (aliskiren hemifumarate; hydrochlorothiazide) generic drug versions launch?
Generic name: aliskiren hemifumarate; hydrochlorothiazide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 23, 2026
Generic Entry Controlled by: European Patent Office Patent 3,391,878

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering TEKTURNA HCT
See drug price trends for TEKTURNA HCT.
The generic ingredient in TEKTURNA HCT is aliskiren hemifumarate; hydrochlorothiazide. There are four drug master file entries for this API. Additional details are available on the aliskiren hemifumarate; hydrochlorothiazide profile page.
When can EXFORGE HCT (amlodipine besylate; hydrochlorothiazide; valsartan) generic drug versions launch?
Generic name: amlodipine besylate; hydrochlorothiazide; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 27, 2026
Generic Entry Controlled by: European Patent Office Patent 2,037,893

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering EXFORGE HCT
See drug price trends for EXFORGE HCT.
The generic ingredient in EXFORGE HCT is amlodipine besylate; hydrochlorothiazide; valsartan. There are fifty drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the amlodipine besylate; hydrochlorothiazide; valsartan profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: European Patent Office Patent 2,069,374

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: European Patent Office Patent 2,457,918

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: European Patent Office Patent 3,045,466

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: European Patent Office Patent 3,363,807

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: European Patent Office Patent 2,069,374

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: European Patent Office Patent 2,457,918

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: European Patent Office Patent 3,045,466

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: European Patent Office Patent 3,363,807

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: European Patent Office Patent 2,069,374

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: European Patent Office Patent 2,457,918

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: European Patent Office Patent 3,045,466

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2026
Generic Entry Controlled by: European Patent Office Patent 3,363,807

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 2,049,506
Patent Title: MODULATEURS DE PROPRIÉTÉS PHARMACOCINÉTIQUES D'AGENTS THÉRAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 2,118,082
Patent Title: MODULATEURS DE PROPRIÉTÉS PHARMACOCINÉTIQUES DE PRODUITS THÉRAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 2,170,851
Patent Title: MODULATEURS DES PROPRIÉTÉS PHARMACOCINÉTIQUES DE SUBSTANCES THÉRAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 2,487,161
Patent Title: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 2,487,162
Patent Title: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 2,487,164
Patent Title: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 2,487,165
Patent Title: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 2,487,166
Patent Title: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 2,487,167
Patent Title: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 2,487,168
Patent Title: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 3,112,355
Patent Title: MODULATEURS DE PROPRIÉTÉS PHARMACOCINÉTIQUES D'AGENTS THÉRAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 3,150,586
Patent Title: MODULATEURS DE PROPRIÉTÉS PHARMACOCINÉTIQUES D'AGENTS THÉRAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 3,689,353
Patent Title: MODULATEURS DE PROPRIÉTÉS PHARMACOCINÉTIQUES D'AGENTS THÉRAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 4,403,221

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 07, 2026
Generic Entry Controlled by: European Patent Office Patent 4,566,670

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can BYFAVO (remimazolam besylate) generic drug versions launch?
Generic name: remimazolam besylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 10, 2026
Generic Entry Controlled by: European Patent Office Patent 2,081,921
Patent Title: SELS DE BENZODIAZÉPINE ET LEURS FORMES POLYMORPHES À ACTION BRÈVE (SHORT-ACTING BENZODIAZEPINE SALTS AND THEIR POLYMORPHIC FORMS)
BYFAVO is a drug marketed by Acacia. There are eleven patents protecting this drug.
This drug has fifty-three patent family members in twenty-four countries. There has been litigation on patents covering BYFAVO
See drug price trends for BYFAVO.
The generic ingredient in BYFAVO is remimazolam besylate. One supplier is listed for this generic product. Additional details are available on the remimazolam besylate profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: European Patent Office Patent 2,046,318
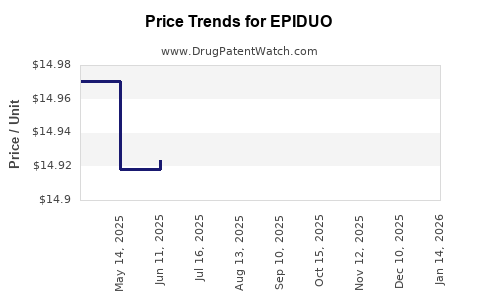
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: European Patent Office Patent 2,450,035

This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: European Patent Office Patent 2,046,318
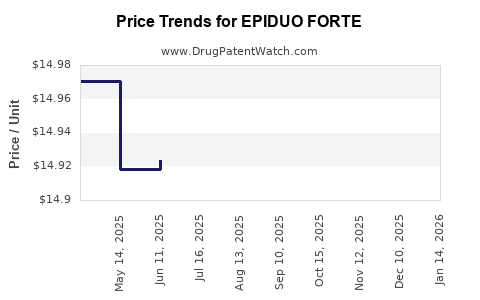
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO FORTE
See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: European Patent Office Patent 2,450,035

This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO FORTE
See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can OLYSIO (simeprevir sodium) generic drug versions launch?
Generic name: simeprevir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 28, 2026
Generic Entry Controlled by: European Patent Office Patent 2,322,516
Patent Title: Composés intermédiaires pour la préparation d'inhibiteurs macrocycliques du virus de l'hépatite C (Intermediates for the preparation of Macrocyclic inhibitors of hepatitis c virus)
OLYSIO is a drug marketed by Janssen Prods. There are eight patents protecting this drug.
This drug has sixty-four patent family members in forty-one countries.
See drug price trends for OLYSIO.
The generic ingredient in OLYSIO is simeprevir sodium. There is one drug master file entry for this API. Additional details are available on the simeprevir sodium profile page.
When can OLYSIO (simeprevir sodium) generic drug versions launch?
Generic name: simeprevir sodium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 28, 2026
Generic Entry Controlled by: European Patent Office Patent 2,937,339
Patent Title: Inhibiteurs macrocycliques du virus de l'hépatite C (Macrocylic inhibitors of hepatitis c virus)
OLYSIO is a drug marketed by Janssen Prods. There are eight patents protecting this drug.
This drug has sixty-four patent family members in forty-one countries.
See drug price trends for OLYSIO.
The generic ingredient in OLYSIO is simeprevir sodium. There is one drug master file entry for this API. Additional details are available on the simeprevir sodium profile page.
When can EPSOLAY (benzoyl peroxide) generic drug versions launch?
Generic name: benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 02, 2026
Generic Entry Controlled by: European Patent Office Patent 1,919,606
Patent Title: REVETEMENT PAR UN OXYDE METALLIQUE D'INGREDIENTS HYDROINSOLUBLES (METAL OXIDE COATING OF WATER INSOLUBLE INGREDIENTS)
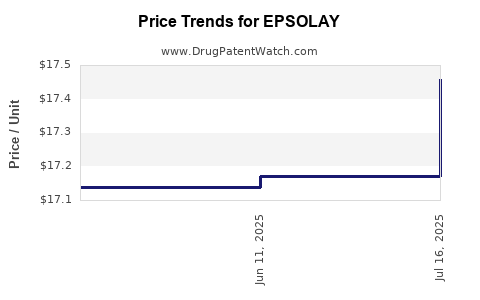
This drug has fifty-one patent family members in fifteen countries. There has been litigation on patents covering EPSOLAY
See drug price trends for EPSOLAY.
The generic ingredient in EPSOLAY is benzoyl peroxide. There are seventeen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the benzoyl peroxide profile page.
When can EPSOLAY (benzoyl peroxide) generic drug versions launch?
Generic name: benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 02, 2026
Generic Entry Controlled by: European Patent Office Patent 2,431,088
Patent Title: Revêtement d'oxyde métallique d'ingrédients insolubles dans l'eau (Metal oxide coating of water insoluble ingredients)

This drug has fifty-one patent family members in fifteen countries. There has been litigation on patents covering EPSOLAY
See drug price trends for EPSOLAY.
The generic ingredient in EPSOLAY is benzoyl peroxide. There are seventeen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the benzoyl peroxide profile page.
When can EPSOLAY (benzoyl peroxide) generic drug versions launch?
Generic name: benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 02, 2026
Generic Entry Controlled by: European Patent Office Patent 2,431,089
Patent Title: Revêtement d'oxyde métallique d'ingrédients insolubles dans l'eau (Metal oxide coating of water insoluble ingredients)

This drug has fifty-one patent family members in fifteen countries. There has been litigation on patents covering EPSOLAY
See drug price trends for EPSOLAY.
The generic ingredient in EPSOLAY is benzoyl peroxide. There are seventeen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the benzoyl peroxide profile page.
When can SALONPAS (menthol; methyl salicylate) generic drug versions launch?
Generic name: menthol; methyl salicylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 04, 2026
Generic Entry Controlled by: European Patent Office Patent 2,047,845
Patent Title: PRÉPARATION ADHÉSIVE (ADHESIVE PREPARATION)
SALONPAS is a drug marketed by Hisamitsu Pharm Co. There are two patents protecting this drug.
This drug has twenty patent family members in sixteen countries.
The generic ingredient in SALONPAS is menthol; methyl salicylate. There are eighteen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the menthol; methyl salicylate profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: European Patent Office Patent 2,049,522

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: European Patent Office Patent 2,385,048

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: European Patent Office Patent 2,784,075

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: European Patent Office Patent 3,042,901

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 11, 2026
Generic Entry Controlled by: European Patent Office Patent 3,321,263

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 21, 2026
Generic Entry Controlled by: European Patent Office Patent 2,056,832
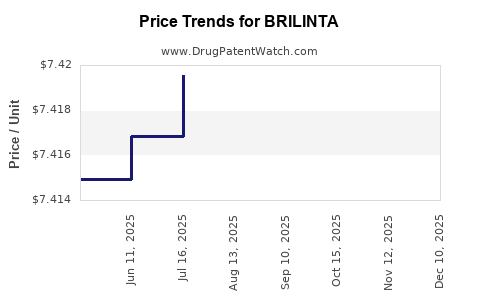
This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 29, 2026
Generic Entry Controlled by: European Patent Office Patent 2,059,246
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the macitentan profile page.
When can LIVDELZI (seladelpar lysine) generic drug versions launch?
Generic name: seladelpar lysine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 13, 2026
Generic Entry Controlled by: European Patent Office Patent 1,937,065
LIVDELZI is a drug marketed by Gilead Sciences Inc. There are six patents protecting this drug.
This drug has one hundred and thirty-eight patent family members in forty-six countries.
The generic ingredient in LIVDELZI is seladelpar lysine. One supplier is listed for this generic product. Additional details are available on the seladelpar lysine profile page.
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 22, 2026
Generic Entry Controlled by: European Patent Office Patent 1,940,364
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 22, 2026
Generic Entry Controlled by: European Patent Office Patent 2,431,025
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can ZUNVEYL (benzgalantamine gluconate) generic drug versions launch?
Generic name: benzgalantamine gluconate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 22, 2026
Generic Entry Controlled by: European Patent Office Patent 1,940,817
Patent Title: AMPLIFICATEURS CHOLINERGIQUES DE PERMÉABILITÉ DE LA BARRIÈRE SANG-CERVEAU AMÉLIORÉE POUR LE TRAITEMENT DE MALADIES ACCOMPAGNÉES D'UNE DÉFICIENCE COGNITIVE (CHOLINERGIC ENHANCERS WITH IMPROVED BLOOD-BRAIN BARRIER PERMEABILITY FOR THE TREATMENT OF DISEASES ACCOMPANIED BY COGNITIVE IMPAIRMENT)
ZUNVEYL is a drug marketed by Alpha Cognition. There are four patents protecting this drug.
This drug has twenty-six patent family members in seventeen countries. There has been litigation on patents covering ZUNVEYL
The generic ingredient in ZUNVEYL is benzgalantamine gluconate. One supplier is listed for this generic product. Additional details are available on the benzgalantamine gluconate profile page.
When can CRESEMBA (isavuconazonium sulfate) generic drug versions launch?
Generic name: isavuconazonium sulfate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 25, 2026
Generic Entry Controlled by: European Patent Office Patent 1,902,708
Patent Title: Compositions pharmaceutiques stabilisées et solides contenant au moins un médicament et procédé d'élaboration (Drug comprising stabilized pharmaceutical solid compositions and processes for their preparation)
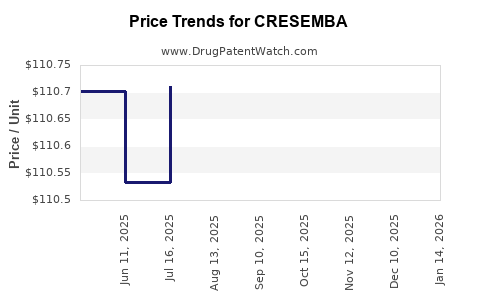
This drug has thirty-two patent family members in nineteen countries. There has been litigation on patents covering CRESEMBA
See drug price trends for CRESEMBA.
The generic ingredient in CRESEMBA is isavuconazonium sulfate. One supplier is listed for this generic product. Additional details are available on the isavuconazonium sulfate profile page.
When can TYMLOS (abaloparatide) generic drug versions launch?
Generic name: abaloparatide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 03, 2026
Generic Entry Controlled by: European Patent Office Patent 2,073,789
Patent Title: COMPOSITION STABLE D'UNE PROTÉINE ANABOLIQUE OSSEUSE QUI EST UN ANALOGUE DE PTHRP, ET SES EMPLOIS (A STABLE COMPOSITION COMPRISING A BONE ANABOLIC PROTEIN, NAMELY A PTHRP ANALOGUE, AND USES THEREOF)

This drug has fifty-six patent family members in twenty-nine countries. There has been litigation on patents covering TYMLOS
See drug price trends for TYMLOS.
The generic ingredient in TYMLOS is abaloparatide. One supplier is listed for this generic product. Additional details are available on the abaloparatide profile page.
When can INREBIC (fedratinib hydrochloride) generic drug versions launch?
Generic name: fedratinib hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: European Patent Office Patent 1,951,684
INREBIC is a drug marketed by Bristol-myers. There are five patents protecting this drug.
This drug has one hundred and nineteen patent family members in forty-two countries. There has been litigation on patents covering INREBIC
See drug price trends for INREBIC.
The generic ingredient in INREBIC is fedratinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the fedratinib hydrochloride profile page.
When can BEVYXXA (betrixaban) generic drug versions launch?
Generic name: betrixaban
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 07, 2026
Generic Entry Controlled by: European Patent Office Patent 1,948,608
Patent Title: SELS PHARMACEUTIQUES ET POLYMORPHES DU N-(5-CHLORO-2-PYRIDINYL)-2-[[4-[(DIMETHYLAMINO)IMINOMETHYL]BENZOYL]AMINO]-5-METHOXY-BENZAMIDE, UN INHIBITEUR DU FACTEUR X (PHARMACEUTICAL SALTS AND POLYMORPHS OF N-(5-CHLORO-2-PYRIDINYL)-2-[[4-[(DIMETHYLAMINO)IMINOMETHYL]BENZOYL]AMINO]-5-METHOXY-BENZAMIDE , A FACTOR XA INHIBITOR)
BEVYXXA is a drug marketed by Portola Pharms Inc. There are five patents protecting this drug.
This drug has sixty-seven patent family members in twenty-six countries. There has been litigation on patents covering BEVYXXA
See drug price trends for BEVYXXA.
The generic ingredient in BEVYXXA is betrixaban. Additional details are available on the betrixaban profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 10, 2026
Generic Entry Controlled by: European Patent Office Patent 2,064,228
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can SOFDRA (sofpironium bromide) generic drug versions launch?
Generic name: sofpironium bromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 13, 2026
Generic Entry Controlled by: European Patent Office Patent 1,948,596
SOFDRA is a drug marketed by Botanix Sb. There are twenty patents protecting this drug.
This drug has one hundred and sixty patent family members in twenty-six countries. There has been litigation on patents covering SOFDRA
The generic ingredient in SOFDRA is sofpironium bromide. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the sofpironium bromide profile page.
When can SOFDRA (sofpironium bromide) generic drug versions launch?
Generic name: sofpironium bromide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 13, 2026
Generic Entry Controlled by: European Patent Office Patent 1,957,451
SOFDRA is a drug marketed by Botanix Sb. There are twenty patents protecting this drug.
This drug has one hundred and sixty patent family members in twenty-six countries. There has been litigation on patents covering SOFDRA
The generic ingredient in SOFDRA is sofpironium bromide. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the sofpironium bromide profile page.
When can AXUMIN (fluciclovine f-18) generic drug versions launch?
Generic name: fluciclovine f-18
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 28, 2026
Generic Entry Controlled by: European Patent Office Patent 1,978,015
Patent Title: COMPOSE PRECURSEUR DE COMPOSE ORGANIQUE MARQUE A L'HALOGENE RADIOACTIF (PRECURSOR COMPOUND OF RADIOACTIVE HALOGEN LABELED ORGANIC COMPOUND)
AXUMIN is a drug marketed by Blue Earth. There are eight patents protecting this drug.
This drug has thirty patent family members in sixteen countries. There has been litigation on patents covering AXUMIN
The generic ingredient in AXUMIN is fluciclovine f-18. One supplier is listed for this generic product. Additional details are available on the fluciclovine f-18 profile page.
When can ZTALMY (ganaxolone) generic drug versions launch?
Generic name: ganaxolone
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 28, 2026
Generic Entry Controlled by: European Patent Office Patent 1,959,966
Patent Title: FORMES GALÉNIQUES DE GANAXOLONE ET PROCÉDÉS DE PREPARATION ET D'UTILISATION DE CELLES-CI (GANAXOLONE FORMULATIONS AND METHODS FOR THE MAKING AND USE THEREOF)
ZTALMY is a drug marketed by Immedica Pharma. There are eleven patents protecting this drug.
This drug has forty-nine patent family members in sixteen countries. There has been litigation on patents covering ZTALMY
See drug price trends for ZTALMY.
The generic ingredient in ZTALMY is ganaxolone. One supplier is listed for this generic product. Additional details are available on the ganaxolone profile page.
When can ZUBSOLV (buprenorphine hydrochloride; naloxone hydrochloride) generic drug versions launch?
Generic name: buprenorphine hydrochloride; naloxone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 04, 2026
Generic Entry Controlled by: European Patent Office Patent 2,101,740
Patent Title: NOUVELLE COMPOSITION PHARMACEUTIQUE NON SUSCEPTIBLE D'ABUS COMPRENANT DES OPIOÏDES (NEW NON-ABUSABLE PHARMACEUTICAL COMPOSITION COMPRISING OPIOIDS)
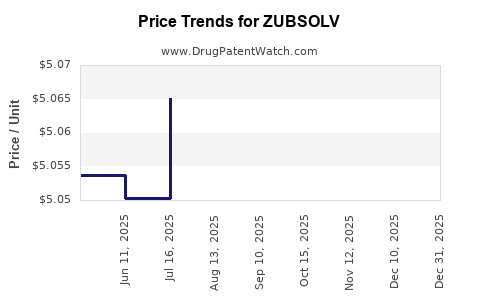
This drug has sixty-two patent family members in thirty-two countries. There has been litigation on patents covering ZUBSOLV
See drug price trends for ZUBSOLV.
The generic ingredient in ZUBSOLV is buprenorphine hydrochloride; naloxone hydrochloride. There are twenty-nine drug master file entries for this API. Twenty-seven suppliers are listed for this generic product. Additional details are available on the buprenorphine hydrochloride; naloxone hydrochloride profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 05, 2026
Generic Entry Controlled by: European Patent Office Patent 2,086,940
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: European Patent Office Patent 2,091,918
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: European Patent Office Patent 2,190,826
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: European Patent Office Patent 2,311,807
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: European Patent Office Patent 2,537,830
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 08, 2026
Generic Entry Controlled by: European Patent Office Patent 3,012,249
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.