EVOTAZ Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Evotaz, and when can generic versions of Evotaz launch?
Evotaz is a drug marketed by Bristol and is included in one NDA. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has three hundred and six patent family members in forty-one countries.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the atazanavir sulfate; cobicistat profile page.
DrugPatentWatch® Generic Entry Outlook for Evotaz
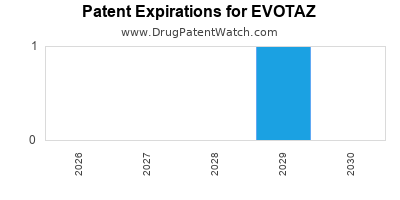
Evotaz was eligible for patent challenges on August 27, 2016.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be September 3, 2029. This may change due to patent challenges or generic licensing.
There have been eight patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for EVOTAZ?
- What are the global sales for EVOTAZ?
- What is Average Wholesale Price for EVOTAZ?
Summary for EVOTAZ
| International Patents: | 306 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 5 |
| Drug Prices: | Drug price information for EVOTAZ |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for EVOTAZ |
| What excipients (inactive ingredients) are in EVOTAZ? | EVOTAZ excipients list |
| DailyMed Link: | EVOTAZ at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for EVOTAZ
Generic Entry Date for EVOTAZ*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for EVOTAZ
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Gilead Sciences | Phase 3 |
| St Stephens Aids Trust | Phase 1 |
| Bristol-Myers Squibb | Phase 1 |
Pharmacology for EVOTAZ
Paragraph IV (Patent) Challenges for EVOTAZ
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| EVOTAZ | Tablets | atazanavir sulfate; cobicistat | 300 mg/150 mg | 206353 | 1 | 2017-09-13 |
US Patents and Regulatory Information for EVOTAZ
EVOTAZ is protected by two US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of EVOTAZ is ⤷ Start Trial.
This potential generic entry date is based on patent 8,148,374.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bristol | EVOTAZ | atazanavir sulfate; cobicistat | TABLET;ORAL | 206353-001 | Jan 29, 2015 | RX | Yes | Yes | 8,148,374 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Bristol | EVOTAZ | atazanavir sulfate; cobicistat | TABLET;ORAL | 206353-001 | Jan 29, 2015 | RX | Yes | Yes | 10,039,718 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for EVOTAZ
When does loss-of-exclusivity occur for EVOTAZ?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
African Regional IP Organization (ARIPO)
Patent: 85
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Patent: 86
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Patent: 15
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Argentina
Patent: 1838
Patent: MODULADORES DE PROPIEDADES FARMACOCINETICAS DE TERAPEUTICOS
Estimated Expiration: ⤷ Start Trial
Patent: 5439
Patent: DERIVADOS DE TIAZOLES 1,3 NO CONDENSADOS,COMPOSICIONES FARMACEUTICAS QUE LOS CONTIENEN Y USOS COMO AGENTES ANTIVIRALES, EN PARTICULAR ANTI HIV.
Estimated Expiration: ⤷ Start Trial
Patent: 7412
Patent: MODULADORES DE PROPIEDADES FARMACEUTICAS DE PRODUCTOS TERAPEUTICOS
Estimated Expiration: ⤷ Start Trial
Patent: 6924
Patent: MODULADORES DE PROPIEDADES FARMACOCINÉTICAS DE TERAPÉUTICOS
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 07275860
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Patent: 08218186
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Patent: 08275744
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0714055
Patent: moduladores de propriedades farmacocinéticas de terapêuticos
Estimated Expiration: ⤷ Start Trial
Patent: 0807581
Patent: MODULADORES DE PROPRIEDADES FARMACOCINÉTICAS DE PRODUTOS TERAPÊUTICOS
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 53374
Patent: MODULATEURS DE PROPRIETES PHARMACOCINETIQUES D'AGENTS THERAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 78907
Patent: MODULATEURS DE PROPRIETES PHARMACOCINETIQUES DE PRODUITS THERAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 92331
Patent: MODULATEURS DES PROPRIETES PHARMACOCINETIQUES DE SUBSTANCES THERAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
China
Patent: 1490023
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Patent: 1679325
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Patent: 1796040
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Patent: 3275033
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Patent: 3435570
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0090077
Patent: MODULATORI FARMAKOKINETIČKIH SVOJSTAVA TERAPEUTIKA (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 0150744
Estimated Expiration: ⤷ Start Trial
Patent: 0161371
Estimated Expiration: ⤷ Start Trial
Patent: 0161428
Estimated Expiration: ⤷ Start Trial
Patent: 0161476
Estimated Expiration: ⤷ Start Trial
Patent: 0161502
Estimated Expiration: ⤷ Start Trial
Patent: 0200349
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 16688
Estimated Expiration: ⤷ Start Trial
Patent: 18219
Estimated Expiration: ⤷ Start Trial
Patent: 18227
Estimated Expiration: ⤷ Start Trial
Patent: 18232
Estimated Expiration: ⤷ Start Trial
Patent: 18292
Estimated Expiration: ⤷ Start Trial
Patent: 22761
Estimated Expiration: ⤷ Start Trial
Patent: 15046
Estimated Expiration: ⤷ Start Trial
Patent: 17001P
Estimated Expiration: ⤷ Start Trial
Patent: 17002
Estimated Expiration: ⤷ Start Trial
Patent: 17003
Estimated Expiration: ⤷ Start Trial
Patent: 20011
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 49506
Estimated Expiration: ⤷ Start Trial
Patent: 87161
Estimated Expiration: ⤷ Start Trial
Patent: 87162
Estimated Expiration: ⤷ Start Trial
Patent: 87163
Estimated Expiration: ⤷ Start Trial
Patent: 87166
Estimated Expiration: ⤷ Start Trial
Patent: 50586
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 9893
Patent: ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ И СПОСОБ ЛЕЧЕНИЯ ВИЧ-ИНФЕКЦИИ (PHARMACEUTICAL COMPOSITION AND METHOD FOR TREATING HIV INFECTION)
Estimated Expiration: ⤷ Start Trial
Patent: 0489
Patent: МОДУЛЯТОРЫ ФАРМАКОКИНЕТИЧЕСКИХ СВОЙСТВ ЛЕКАРСТВЕННЫХ СРЕДСТВ (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 5845
Patent: МОДУЛЯТОРЫ ФАРМАКОКИНЕТИЧЕСКИХ СВОЙСТВ ЛЕКАРСТВЕННЫХ СРЕДСТВ (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 0900155
Patent: МОДУЛЯТОРЫ ФАРМАКОКИНЕТИЧЕСКИХ СВОЙСТВ ЛЕКАРСТВЕННЫХ СРЕДСТВ
Estimated Expiration: ⤷ Start Trial
Patent: 0901155
Patent: МОДУЛЯТОРЫ ФАРМАКОКИНЕТИЧЕСКИХ СВОЙСТВ ТЕРАПЕВТИЧЕСКИХ ПРЕПАРАТОВ (ВАРИАНТЫ) И ИХ ИСПОЛЬЗОВАНИЕ В ТЕРАПИИ
Estimated Expiration: ⤷ Start Trial
Patent: 1270738
Patent: МОДУЛЯТОРЫ ФАРМАКОКИНЕТИЧЕСКИХ СВОЙСТВ ЛЕКАРСТВЕННЫХ СРЕДСТВ
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 49506
Patent: MODULATEURS DE PROPRIÉTÉS PHARMACOCINÉTIQUES D'AGENTS THÉRAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 18082
Patent: MODULATEURS DE PROPRIÉTÉS PHARMACOCINÉTIQUES DE PRODUITS THÉRAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 70851
Patent: MODULATEURS DES PROPRIÉTÉS PHARMACOCINÉTIQUES DE SUBSTANCES THÉRAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 87161
Patent: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)
Estimated Expiration: ⤷ Start Trial
Patent: 87162
Patent: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)
Estimated Expiration: ⤷ Start Trial
Patent: 87163
Patent: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)
Estimated Expiration: ⤷ Start Trial
Patent: 87164
Patent: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)
Estimated Expiration: ⤷ Start Trial
Patent: 87165
Patent: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)
Estimated Expiration: ⤷ Start Trial
Patent: 87166
Patent: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)
Estimated Expiration: ⤷ Start Trial
Patent: 87167
Patent: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)
Estimated Expiration: ⤷ Start Trial
Patent: 87168
Patent: Modulateurs de propriétés pharmacocinétiques d'agents thérapeutiques (Modulators of pharmacokinetic properties of therapeutics)
Estimated Expiration: ⤷ Start Trial
Patent: 12355
Patent: MODULATEURS DE PROPRIÉTÉS PHARMACOCINÉTIQUES D'AGENTS THÉRAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 50586
Patent: MODULATEURS DE PROPRIÉTÉS PHARMACOCINÉTIQUES D'AGENTS THÉRAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 89353
Patent: MODULATEURS DE PROPRIÉTÉS PHARMACOCINÉTIQUES D'AGENTS THÉRAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 96171
Patent: MODULATEURS DE PROPRIÉTÉS PHARMACOCINÉTIQUES D'AGENTS THÉRAPEUTIQUES (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 03221
Estimated Expiration: ⤷ Start Trial
Patent: 66670
Estimated Expiration: ⤷ Start Trial
Finland
Patent: 49506
Estimated Expiration: ⤷ Start Trial
Patent: 87161
Estimated Expiration: ⤷ Start Trial
Patent: 0150052
Estimated Expiration: ⤷ Start Trial
France
Patent: C0078
Estimated Expiration: ⤷ Start Trial
Patent: C1000
Estimated Expiration: ⤷ Start Trial
Patent: C1001
Estimated Expiration: ⤷ Start Trial
Patent: C1002
Estimated Expiration: ⤷ Start Trial
Patent: C1021
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 26485
Patent: 藥動性質治療調節器 (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 37451
Patent: 藥動性質治療的調節器 (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 43144
Patent: MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS
Estimated Expiration: ⤷ Start Trial
Patent: 32217
Patent: 治療劑的藥代動力學特性調節劑 (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 25565
Estimated Expiration: ⤷ Start Trial
Patent: 29842
Estimated Expiration: ⤷ Start Trial
Patent: 29843
Estimated Expiration: ⤷ Start Trial
Patent: 29866
Estimated Expiration: ⤷ Start Trial
Patent: 31707
Estimated Expiration: ⤷ Start Trial
Patent: 600052
Estimated Expiration: ⤷ Start Trial
Patent: 600053
Estimated Expiration: ⤷ Start Trial
Patent: 600054
Estimated Expiration: ⤷ Start Trial
Patent: 000013
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 5488
Patent: תרכובות, תכשירים רוקחיים המכילים אותן ושימושים שלהם (Compounds, pharmaceutical compositions comprising the same and uses thereof)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 81433
Estimated Expiration: ⤷ Start Trial
Patent: 81446
Estimated Expiration: ⤷ Start Trial
Patent: 93665
Estimated Expiration: ⤷ Start Trial
Patent: 54300
Estimated Expiration: ⤷ Start Trial
Patent: 47282
Estimated Expiration: ⤷ Start Trial
Patent: 47388
Estimated Expiration: ⤷ Start Trial
Patent: 92776
Estimated Expiration: ⤷ Start Trial
Patent: 09542696
Estimated Expiration: ⤷ Start Trial
Patent: 10519314
Estimated Expiration: ⤷ Start Trial
Patent: 10532755
Estimated Expiration: ⤷ Start Trial
Patent: 11231132
Patent: MODULATOR OF PHARMACOKINETIC PROPERTY OF THERAPEUTIC
Estimated Expiration: ⤷ Start Trial
Patent: 12051933
Patent: MODULATION OF PHARMACOKINETIC PROPERTIES OF THERAPEUTIC AGENT
Estimated Expiration: ⤷ Start Trial
Patent: 14088441
Patent: ADJUSTMENT OF PHARMACOKINETICS OF CURATIVE MEDICINE
Estimated Expiration: ⤷ Start Trial
Patent: 15078241
Patent: 治療薬の薬物動態特性の調節 (ADJUSTMENT OF PHARMACOKINETICS OF CURATIVE MEDICINE)
Estimated Expiration: ⤷ Start Trial
Patent: 16121193
Patent: 治療薬の薬物動態特性の調節 (ADJUSTMENT OF PHARMACOKINETICS OF CURATIVE MEDICINE)
Estimated Expiration: ⤷ Start Trial
Patent: 16172773
Patent: 治療薬の薬物動態特性の調節 (ADJUSTMENT OF PHARMACOKINETICS OF CURATIVE MEDICINE)
Estimated Expiration: ⤷ Start Trial
Patent: 17171695
Patent: 治療薬の薬物動態特性の調節 (MODULATION OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 19059790
Patent: 治療薬の薬物動態特性の調節 (MODULATION OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 21001236
Patent: 治療薬の薬物動態特性の調節 (MODULATION OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 87161
Estimated Expiration: ⤷ Start Trial
Patent: 87162
Estimated Expiration: ⤷ Start Trial
Patent: 87163
Estimated Expiration: ⤷ Start Trial
Patent: 87166
Estimated Expiration: ⤷ Start Trial
Patent: 50586
Estimated Expiration: ⤷ Start Trial
Patent: 049506
Estimated Expiration: ⤷ Start Trial
Patent: 487162
Estimated Expiration: ⤷ Start Trial
Patent: 487163
Estimated Expiration: ⤷ Start Trial
Patent: 487166
Estimated Expiration: ⤷ Start Trial
Patent: 150586
Estimated Expiration: ⤷ Start Trial
Patent: 2015040
Estimated Expiration: ⤷ Start Trial
Patent: 2016038
Estimated Expiration: ⤷ Start Trial
Patent: 2016039
Estimated Expiration: ⤷ Start Trial
Patent: 2016040
Estimated Expiration: ⤷ Start Trial
Patent: 2020508
Estimated Expiration: ⤷ Start Trial
Luxembourg
Patent: 864
Estimated Expiration: ⤷ Start Trial
Patent: 0156
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 09000234
Patent: MODULADORES DE LAS PROPIEDADES FARMACOCINETICAS DE PRODUCTOS TERAPEUTICOS. (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS.)
Estimated Expiration: ⤷ Start Trial
Patent: 09008935
Patent: MODULADORES DE LAS PROPIEDADES FARMACOCINETICAS DE PRODUCTOS TERAPEUTICOS. (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS.)
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 258
Patent: MODULATORI FARMAKOKINETIČKIH SVOJSTAVA TERAPEUTIKA (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 582
Patent: Modulatori farmakokinetičkih svojstava terapeutika (Modulators of pharmacokinetic properties of therapeutics)
Estimated Expiration: ⤷ Start Trial
Patent: 628
Patent: Modulatori farmakokinetičkih svojstava lekova (Modulators of pharmacokinetic properties of therapeutics)
Estimated Expiration: ⤷ Start Trial
Patent: 631
Patent: Modulatori farmakokinetičkih svojstava lijekova (Modulators of pharmacokinetic properties of therapeutics)
Estimated Expiration: ⤷ Start Trial
Patent: 680
Patent: Modulatori farmakokinetickih svojstava lijekova (Modulators of pharmacokinetic properties of therapeutics)
Estimated Expiration: ⤷ Start Trial
Netherlands
Patent: 0780
Estimated Expiration: ⤷ Start Trial
Patent: 1045
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 3060
Patent: MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS
Estimated Expiration: ⤷ Start Trial
Patent: 9802
Patent: Compounds for improving the efficacy of other drugs
Estimated Expiration: ⤷ Start Trial
Patent: 2093
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 3776
Estimated Expiration: ⤷ Start Trial
Patent: 5343
Estimated Expiration: ⤷ Start Trial
Patent: 5893
Estimated Expiration: ⤷ Start Trial
Patent: 7119
Estimated Expiration: ⤷ Start Trial
Patent: 17001
Estimated Expiration: ⤷ Start Trial
Patent: 17002
Estimated Expiration: ⤷ Start Trial
Patent: 17003
Estimated Expiration: ⤷ Start Trial
Patent: 19027
Estimated Expiration: ⤷ Start Trial
Patent: 19031
Estimated Expiration: ⤷ Start Trial
Patent: 20011
Estimated Expiration: ⤷ Start Trial
Patent: 090593
Estimated Expiration: ⤷ Start Trial
Patent: 190528
Patent: Modulatorer av farmakokinetikkegenskaper til terapeutika
Estimated Expiration: ⤷ Start Trial
Patent: 200578
Patent: Modulatorer av farmakokinetikkegenskaper til terapeutika
Estimated Expiration: ⤷ Start Trial
Patent: 210811
Patent: Modulatorer av farmakokinetikkegenskaper til terapeutika
Estimated Expiration: ⤷ Start Trial
Patent: 230434
Patent: Modulatorer av farmakokinetikkegenskaper til terapeutika
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 49506
Estimated Expiration: ⤷ Start Trial
Patent: 87161
Estimated Expiration: ⤷ Start Trial
Patent: 87162
Estimated Expiration: ⤷ Start Trial
Patent: 87163
Estimated Expiration: ⤷ Start Trial
Patent: 87166
Estimated Expiration: ⤷ Start Trial
Patent: 50586
Estimated Expiration: ⤷ Start Trial
Patent: 96171
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 49506
Estimated Expiration: ⤷ Start Trial
Patent: 18082
Estimated Expiration: ⤷ Start Trial
Patent: 87161
Estimated Expiration: ⤷ Start Trial
Patent: 87162
Estimated Expiration: ⤷ Start Trial
Patent: 87163
Estimated Expiration: ⤷ Start Trial
Patent: 87165
Estimated Expiration: ⤷ Start Trial
Patent: 87166
Estimated Expiration: ⤷ Start Trial
Patent: 50586
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 237
Patent: MODULATORI FARMAKOKINETIČKIH SVOJSTAVA TERAPEUTIKA (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 195
Patent: MODULATORI FARMAKOKINETIČKIH SVOJSTAVA TERAPEUTIKA (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 280
Patent: MODULATORI FARMAKOKINETIČKIH SVOJSTAVA LEKOVA (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 351
Patent: MODULATORI FARMAKOKINETIČKIH SVOJSTAVA LEKOVA (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 459
Patent: MODULATORI FARMAKOKINETIČKIH SVOJSTAVA LEKOVA (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201705929Q
Patent: MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS
Estimated Expiration: ⤷ Start Trial
Patent: 3007
Patent: MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS
Estimated Expiration: ⤷ Start Trial
Patent: 3059
Patent: MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 49506
Estimated Expiration: ⤷ Start Trial
Patent: 87161
Estimated Expiration: ⤷ Start Trial
Patent: 87162
Estimated Expiration: ⤷ Start Trial
Patent: 87163
Estimated Expiration: ⤷ Start Trial
Patent: 87166
Estimated Expiration: ⤷ Start Trial
Patent: 50586
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 0905882
Patent: MODULATOR OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1477822
Estimated Expiration: ⤷ Start Trial
Patent: 1577698
Estimated Expiration: ⤷ Start Trial
Patent: 1636221
Estimated Expiration: ⤷ Start Trial
Patent: 1664154
Estimated Expiration: ⤷ Start Trial
Patent: 090028821
Patent: MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS
Estimated Expiration: ⤷ Start Trial
Patent: 090122261
Patent: MOLATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS
Estimated Expiration: ⤷ Start Trial
Patent: 100040896
Patent: MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS
Estimated Expiration: ⤷ Start Trial
Patent: 140004808
Patent: MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS
Estimated Expiration: ⤷ Start Trial
Patent: 150042869
Patent: 치료제의 약동학 특성의 모듈레이터 (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 150056665
Patent: 치료제의 약동학적 특성의 조절제 (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 38275
Estimated Expiration: ⤷ Start Trial
Patent: 25454
Estimated Expiration: ⤷ Start Trial
Patent: 46378
Estimated Expiration: ⤷ Start Trial
Patent: 01820
Estimated Expiration: ⤷ Start Trial
Patent: 02784
Estimated Expiration: ⤷ Start Trial
Patent: 03617
Estimated Expiration: ⤷ Start Trial
Patent: 03645
Estimated Expiration: ⤷ Start Trial
Patent: 11308
Estimated Expiration: ⤷ Start Trial
Patent: 79826
Estimated Expiration: ⤷ Start Trial
Patent: 96275
Estimated Expiration: ⤷ Start Trial
Patent: 26665
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 0811167
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Patent: 0904419
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Patent: 0909422
Patent: Modulators of pharmacokinetic properties of therapeutics
Estimated Expiration: ⤷ Start Trial
Patent: 1412733
Patent: Modulators for improving pharmacokinetic properties of therapeutics metabolized by cytochrome P450 monooxygenase
Estimated Expiration: ⤷ Start Trial
Patent: 11613
Estimated Expiration: ⤷ Start Trial
Patent: 48457
Estimated Expiration: ⤷ Start Trial
Patent: 96778
Estimated Expiration: ⤷ Start Trial
Patent: 33868
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 112
Patent: МОДУЛЯТОРЫ ФАРМАКОКИНЕТИЧЕСКИХ СВОЙСТВ ЛЕКАРСТВЕННЫХ СРЕДСТВ;МОДУЛЯТОРИ ФАРМАКОКІНЕТИЧНИХ ВЛАСТИВОСТЕЙ ЛІКАРСЬКИХ ЗАСОБІВ (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Patent: 1312
Patent: МОДУЛЯТОРЫ ФАРМАКОКИНЕТИЧЕСКИХ СВОЙСТВ ТЕРАПЕВТИЧЕСКИХ ПРЕПАРАТОВ;МОДУЛЯТОРИ ФАРМАКОКІНЕТИЧНИХ ВЛАСТИВОСТЕЙ ТЕРАПЕВТИЧНИХ ПРЕПАРАТІВ (MODULATORS OF PHARMACOKINETIC PROPERTIES OF THERAPEUTICS)
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering EVOTAZ around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Hungary | S1600053 | ⤷ Start Trial | |
| Poland | 3150586 | ⤷ Start Trial | |
| South Korea | 20140004808 | ⤷ Start Trial | |
| Poland | 329177 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for EVOTAZ
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2049506 | C201530064 | Spain | ⤷ Start Trial | PRODUCT NAME: COBICISTAT O UNA SAL FARMACEUTICAMENTE ACEPTABLE DEL MISMO; NATIONAL AUTHORISATION NUMBER: EU/1/13/830; DATE OF AUTHORISATION: 20130524; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/13/830; DATE OF FIRST AUTHORISATION IN EEA: 20130524 |
| 3150586 | 2020C/515 | Belgium | ⤷ Start Trial | PRODUCT NAME: COBICISTAT OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT OF SOLVAAT HIERVAN, DARUNAVIR OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT OF SOLVAAT HIERVAN, IN HET BIJZONDER DARUNAVIR ETHANOLATE, EN EMTRICITABINE OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT OF SOLVAAT HIERVAN; AUTHORISATION NUMBER AND DATE: EU/1/17/1225 20170925 |
| 2049506 | 300780 | Netherlands | ⤷ Start Trial | PRODUCT NAME: COBICISTAT OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN; REGISTRATION NO/DATE: EU/1/13/830/001-002 20130527 |
| 2049506 | 262 5024-2015 | Slovakia | ⤷ Start Trial | PRODUCT NAME: KOBICISTAT VO VSETKYCH FORMACH CHRANENYCH ZAKLADNYM; REGISTRATION NO/DATE: EU/1/13/830 20130527 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory of EVOTAZ
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.