Share This Page
Drug Price Trends for TWIRLA
✉ Email this page to a colleague
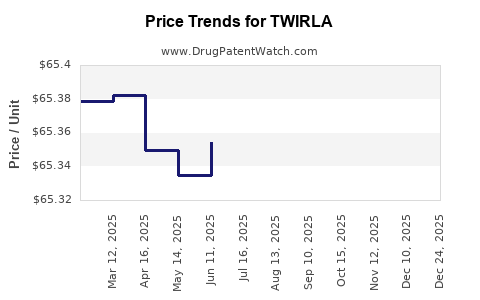
Average Pharmacy Cost for TWIRLA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TWIRLA 120-30 MCG/DAY PATCH | 71671-0100-11 | 67.21328 | EACH | 2026-01-01 |
| TWIRLA 120-30 MCG/DAY PATCH | 71671-0100-03 | 67.21328 | EACH | 2026-01-01 |
| TWIRLA 120-30 MCG/DAY PATCH | 71671-0100-11 | 65.25561 | EACH | 2025-12-17 |
| TWIRLA 120-30 MCG/DAY PATCH | 71671-0100-03 | 65.25561 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Twirla (levonorgestrel/ethinyl estradiol) Market Analysis and Price Projections
Twirla, a transdermal contraceptive patch containing levonorgestrel and ethinyl estradiol, presents a defined market segment within the global female contraception market. Its efficacy and delivery method position it for continued, albeit competitive, uptake. Price projections are influenced by manufacturing costs, competitor pricing, and reimbursement policies.
What is Twirla's Market Position?
Twirla is an extended-cycle transdermal contraceptive patch. It is indicated for the prevention of pregnancy in women ofroductive potential with a total weight of 50 kg (110 lb) or greater. The patch delivers a combination of 1.5 mg levonorgestrel and 30 mcg ethinyl estradiol per 24 hours. [1]
The global female contraception market is substantial and projected for steady growth. Factors driving this growth include increasing awareness of family planning, rising disposable incomes in emerging economies, and the continuous development of new contraceptive methods and improved formulations. Within this broader market, transdermal patches represent a niche but significant segment due to their convenience and consistent hormone delivery compared to daily oral contraceptives.
Twirla competes with other hormonal contraceptive methods, including oral contraceptive pills, vaginal rings, injectable contraceptives, and other transdermal patches. Its primary direct competitor in the transdermal patch category is Xulane (formerly known as Ortho Evra), which has been available for a longer period and possesses established market share. [2] Twirla's development aimed to address certain attributes, potentially including adhesion or regimen adherence, though direct comparative efficacy data often leads to nuanced market positioning rather than drastic shifts.
The specific market share of Twirla is not widely disclosed as a standalone entity in broad market reports, as it is often aggregated with other transdermal contraceptives or hormonal methods. However, industry analyses consistently indicate that oral contraceptives remain the dominant form of female contraception globally. Transdermal patches, while convenient for some users, represent a smaller but stable percentage of the overall market.
What are the Key Clinical and Regulatory Aspects of Twirla?
Twirla is a prescription-only medication. Its approval by regulatory bodies such as the U.S. Food and Drug Administration (FDA) is a prerequisite for market access. The FDA approved Twirla in May 2020. [1]
The prescribing information for Twirla includes a boxed warning regarding the risk of thromboembolic disorders and other vascular events. This warning is standard for combined hormonal contraceptives. [1] The risk is higher in women who are overweight (BMI ≥ 25 kg/m²). This specific cautionary note directly impacts the eligible patient population for Twirla, as indicated by its labeling for women weighing 50 kg or greater. [1]
Regulatory approvals in other major markets, such as the European Union, would be crucial for global expansion, but current market penetration appears primarily focused on the U.S.
The development and ongoing regulatory compliance for Twirla involve significant investment in clinical trials to demonstrate safety and efficacy, post-market surveillance, and adherence to Good Manufacturing Practices (GMP). These regulatory hurdles contribute to the cost of goods sold and influence pricing strategies.
What are the Manufacturing and Supply Chain Considerations?
The manufacturing of a transdermal patch involves complex processes, including drug formulation, lamination, cutting, and packaging. The active pharmaceutical ingredients (APIs) – levonorgestrel and ethinyl estradiol – are synthesized through multi-step chemical processes. The backing layer, adhesive matrix, and release liner are specialized materials.
Manufacturing costs are influenced by:
- API Sourcing: The cost and reliable supply of levonorgestrel and ethinyl estradiol are critical. These are well-established APIs, but quality and consistency are paramount.
- Transdermal Technology: The proprietary adhesive technology and the manufacturing equipment for precise drug loading and controlled release contribute significantly to capital expenditure and operational costs.
- Quality Control: Stringent quality control measures are required at every stage to ensure consistent drug release, adhesion, and absence of contaminants.
- Scale of Production: Larger production volumes can lead to economies of scale, reducing per-unit manufacturing costs.
- Supply Chain Logistics: Ensuring a stable and efficient supply chain from API suppliers to finished product manufacturers and distributors is essential to prevent stockouts and maintain market availability.
The supply chain for pharmaceuticals, particularly for a product with a limited number of manufacturers for specialized components like transdermal adhesives, can be subject to disruption. Geopolitical factors, raw material availability, and unexpected demand shifts can impact the cost and accessibility of components.
What is the Competitive Landscape for Twirla?
Twirla operates in a competitive market for female contraception. Its primary direct competitors are other transdermal contraceptive patches.
Key Competitors:
- Xulane (levonorgestrel/ethinyl estradiol transdermal system): This is Twirla's most direct competitor, utilizing the same API combination. Xulane has a longer history in the market and a more established brand presence. [2]
- Evra (norelgestromin/ethinyl estradiol transdermal system): A different combination of hormones, Evra also competes in the transdermal patch segment.
Indirect Competition:
- Oral Contraceptive Pills (OCPs): The largest segment of the contraceptive market. A vast array of OCPs are available, offering different hormone combinations, dosages, and dosing regimens (daily, extended-cycle).
- Vaginal Rings (e.g., NuvaRing, Annovera): Offer a monthly or annual dosing schedule, providing an alternative to daily pills and weekly patches.
- Injectable Contraceptives (e.g., Depo-Provera): Provide long-acting contraception, typically administered every few months.
- Intrauterine Devices (IUDs): Long-acting reversible contraceptives (LARCs) offering several years of protection.
Twirla's competitive advantage hinges on its specific formulation, adhesion characteristics, and potentially its patient-specific dosing guidelines (e.g., for women of certain weights). However, the established efficacy and widespread familiarity with OCPs and the long-term convenience of LARCs present significant competitive barriers.
Pricing strategies of competitors, particularly Xulane, will directly influence Twirla's market penetration and pricing flexibility. Reimbursement rates from insurance providers and government programs also play a critical role in determining patient out-of-pocket costs and, consequently, adoption rates.
What are the Price Drivers for Twirla?
The pricing of Twirla is determined by a confluence of factors:
- Manufacturing Costs: This includes the cost of APIs, excipients, specialized transdermal patch manufacturing, quality control, and packaging. The proprietary nature of transdermal delivery systems often leads to higher manufacturing costs compared to oral dosage forms.
- Research and Development (R&D) Costs: The initial investment in developing Twirla, including clinical trials to demonstrate safety and efficacy, regulatory submission fees, and post-market studies, is amortized into the product's price.
- Competitive Pricing: The pricing of comparable products, particularly Xulane and other transdermal patches, serves as a benchmark. If Twirla is priced significantly higher without clear clinical advantages, adoption will be limited.
- Reimbursement Policies: The extent to which private insurers and government healthcare programs (e.g., Medicare, Medicaid in the U.S.) cover Twirla, and at what co-payment level for patients, is a primary determinant of its real-world affordability and uptake. Manufacturers often engage in formulary negotiations to secure preferred placement on insurance formularies.
- Marketing and Sales Expenses: Costs associated with educating healthcare providers and patients about Twirla, including sales force expenses, promotional materials, and advertising, are factored into the pricing.
- Target Patient Population: The specific indication for Twirla (women weighing 50 kg or greater) may influence the addressable market size and, therefore, pricing strategies aimed at maximizing revenue from this segment.
- Perceived Value and Patient Adherence: The convenience of a weekly patch, compared to daily pills, can be valued by patients and healthcare providers, potentially supporting a premium price. However, this value must outweigh any perceived drawbacks or higher costs.
- Generic Competition: As of its introduction, Twirla is a branded product. The absence of generic equivalents for transdermal contraceptive patches generally allows for higher pricing. However, the eventual expiration of patent protection and the introduction of generics would significantly impact pricing.
The initial launch price of Twirla was set in the context of prevailing prices for comparable transdermal contraceptives. For instance, the list price for a 3-month supply (3 patches) of Xulane has historically ranged from $150 to $200 USD. [3] Twirla's pricing would likely align with or be slightly differentiated from this range, subject to formulary negotiations.
What are the Price Projections for Twirla?
Predicting precise future pricing is subject to market dynamics, but general trends can be projected:
Short-Term (1-3 years post-launch):
- Stable to Modest Increases: Initial pricing will likely remain relatively stable, with modest increases reflecting inflation and continued R&D amortization.
- Competitive Pressure: Pricing will be heavily influenced by Xulane's price and any new entrants to the transdermal patch market.
- Reimbursement Negotiations: Ongoing negotiations with payers will shape effective pricing and patient out-of-pocket costs. Generic Xulane, if available or approved, would place downward pressure on the entire patch market.
Medium-Term (3-7 years post-launch):
- Potential Price Erosion due to Competition: As the market matures and if generic versions of competing patches enter the market, there could be downward pressure on Twirla's price.
- Patent Expiry Impact: The eventual expiry of Twirla's primary patents will pave the way for generic competition, leading to significant price reductions. The timing of patent expiries is critical. U.S. patent 9,125,708 for the levonorgestrel/ethinyl estradiol transdermal system lists expiration dates well into the 2030s, but this is subject to patent challenges and extensions. [4]
- Market Share Development: Twirla's pricing will also depend on its ability to capture and maintain market share against established competitors.
Long-Term (7+ years post-launch):
- Generic Dominance: The market will likely be dominated by generic versions of Twirla and its competitors, with prices reflecting manufacturing costs and competitive dynamics of generic products.
- Shift in Delivery Methods: Evolving contraceptive technologies could also influence pricing by shifting patient preference and payer coverage towards newer or different delivery methods.
Estimated Price Range: Given the competitive landscape and pricing of Xulane, a monthly supply (approximately 3 patches) of Twirla, at retail, could be expected to range from $150 to $220 USD in its initial branded phase. This range is a projection based on comparable products and market conditions as of its launch and immediate follow-on period. Insurance coverage, co-pay assistance programs, and pharmacy discounts will significantly alter the actual cost to the end-user.
Key Takeaways
- Twirla is a prescription transdermal contraceptive patch competing in a substantial but fragmented global female contraception market.
- Its primary direct competitor is Xulane, with a longer market history. Broader competition includes oral contraceptives, rings, injections, and IUDs.
- Key price drivers include manufacturing complexity, R&D amortization, competitive pricing, and payer reimbursement policies.
- Short-term pricing is expected to be stable to moderately increasing, heavily influenced by direct competitors.
- Medium to long-term pricing will be significantly impacted by patent expirations and the subsequent introduction of generic versions.
- Actual patient cost will be heavily mediated by insurance coverage and co-pay structures.
FAQs
-
What is the primary difference between Twirla and other transdermal contraceptive patches like Xulane? Both Twirla and Xulane contain the same active pharmaceutical ingredients: levonorgestrel and ethinyl estradiol. Differences typically lie in proprietary adhesive technology, patch design, and potentially minor variations in drug release profiles or adhesion characteristics, which may be perceived differently by patients.
-
How does the weight restriction for Twirla impact its market potential? The indication for women weighing 50 kg (110 lb) or greater, alongside a boxed warning about increased risk of thromboembolic disorders in overweight women, narrows the eligible patient population compared to some other contraceptive methods. This can affect market penetration and necessitates targeted marketing.
-
What is the typical cost structure for a transdermal contraceptive patch like Twirla? The cost includes API sourcing, complex transdermal manufacturing, rigorous quality control, packaging, marketing, sales, and R&D amortization. These factors contribute to a higher per-unit cost compared to oral contraceptives.
-
Will Twirla become cheaper once generic versions are available? Yes, as is standard in the pharmaceutical industry, the introduction of generic versions following patent expiration will lead to a significant reduction in the price of Twirla.
-
What role do insurance companies play in determining the affordability of Twirla? Insurance companies are critical. Their formulary placement, co-payment requirements, and coverage decisions directly determine the out-of-pocket cost for patients. Negotiations between the manufacturer and payers heavily influence the effective price and patient access.
Citations
[1] U.S. Food & Drug Administration. (2020, May 22). FDA approves Twirla (levonorgestrel/ethinyl estradiol) transdermal system. https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-twirla-levonorgestrel-ethinyl-estradiol-transdermal-system
[2] Warner, S. (2023, January 10). Twirla (levonorgestrel/ethinyl estradiol) Transdermal Patch. Drugs.com. https://www.drugs.com/mtm/twirla.html (Note: While Drugs.com is a patient-facing resource, it provides summaries of prescribing information and comparative context. Information regarding competitors like Xulane is widely established in market analyses.)
[3] GoodRx. (n.d.). Xulane prices, coupons, and patient assistance programs. Retrieved from https://www.goodrx.com/xulane (Note: GoodRx provides retail pricing data which can serve as an indicator of manufacturer list prices and competitive pricing benchmarks.)
[4] USPTO Patent Full-Text and Image Database. (n.d.). U.S. Patent 9,125,708. Retrieved from USPTO website. (Specific patent number and its expiration data are subject to ongoing legal status and patent office records.)
More… ↓
