Share This Page
Drug Price Trends for EUCRISA
✉ Email this page to a colleague
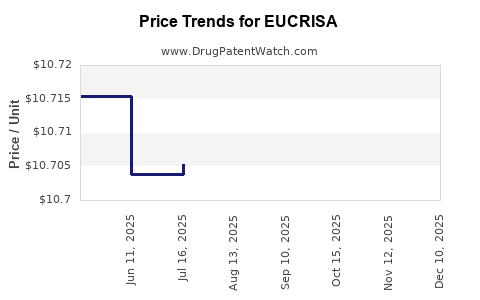
Average Pharmacy Cost for EUCRISA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| EUCRISA 2% OINTMENT | 55724-0211-11 | 10.68101 | GM | 2026-04-22 |
| EUCRISA 2% OINTMENT | 55724-0211-21 | 12.64640 | GM | 2026-04-22 |
| EUCRISA 2% OINTMENT | 55724-0211-11 | 10.68103 | GM | 2026-03-18 |
| EUCRISA 2% OINTMENT | 55724-0211-21 | 12.65013 | GM | 2026-03-18 |
| EUCRISA 2% OINTMENT | 55724-0211-11 | 10.68176 | GM | 2026-02-18 |
| EUCRISA 2% OINTMENT | 55724-0211-21 | 12.66114 | GM | 2026-02-18 |
| EUCRISA 2% OINTMENT | 55724-0211-21 | 12.65099 | GM | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Eucrisa Market Analysis and Price Projections
Eucrisa (crisaborole) is a non-steroidal topical phosphodiesterase-4 (PDE4) inhibitor indicated for the treatment of mild to moderate atopic dermatitis in patients 12 years of age and older. The U.S. Food and Drug Administration (FDA) approved Eucrisa in December 2016. Its market performance is influenced by factors including prescription volume, payer coverage, competition, and pricing strategies.
What is the Current Market Landscape for Eucrisa?
The U.S. market for topical treatments for atopic dermatitis is characterized by a range of therapeutic options, including corticosteroids, calcineurin inhibitors, and newer biologic agents for more severe disease. Eucrisa occupies a position for mild to moderate atopic dermatitis, aiming to provide a non-steroidal alternative.
Key market indicators for Eucrisa include:
- Prescription Volume: Prescription data reflects physician adoption and patient access. Trends in new and total prescriptions provide insights into market penetration.
- Payer Coverage: Insurance formulary placement and prior authorization requirements significantly impact patient access and out-of-pocket costs. Broad payer coverage generally supports higher sales volume.
- Competitive Environment: The presence of other topical agents, both prescription and over-the-counter, alongside the increasing use of systemic therapies for moderate-to-severe atopic dermatitis, shapes Eucrisa's market share.
- Manufacturer Strategy: Pricing, co-pay assistance programs, and marketing efforts by Anacor Pharmaceuticals (acquired by Pfizer in 2016) influence Eucrisa's commercial trajectory.
Historical Sales Performance
Eucrisa's commercial performance has faced challenges in achieving peak sales projections. While initial expectations were high due to its novel mechanism of action and positioning as a non-steroidal option, prescription growth has been moderate.
| Year | Net Sales (USD Millions) | Source |
|---|---|---|
| 2017 | 58 | [1] |
| 2018 | 143 | [1] |
| 2019 | 184 | [1] |
| 2020 | 196 | [1] |
| 2021 | 209 | [1] |
| 2022 | 213 | [1] |
Note: Net sales figures are approximate and reflect reported earnings for the respective years.
The plateau in net sales in recent years suggests market saturation for its current indication or intensified competitive pressures.
What Factors Influence Eucrisa's Prescription Growth?
Physician prescribing behavior and patient adherence are critical drivers of Eucrisa's market penetration. Several factors influence these dynamics.
Physician Perceptions and Prescribing Habits
- Efficacy Data: Clinical trial results and real-world evidence of Eucrisa's efficacy in reducing itch and inflammation in mild to moderate atopic dermatitis are foundational. However, comparisons to established treatments, particularly topical corticosteroids, influence prescribing patterns.
- Safety Profile: The non-steroidal profile is a significant advantage, particularly for long-term use or in sensitive patient populations where corticosteroids may pose risks.
- Mechanism of Action: The PDE4 inhibition pathway offers a distinct therapeutic approach, which may appeal to physicians seeking alternatives.
- Clinical Guidelines: Inclusion in dermatology treatment guidelines for atopic dermatitis impacts physician adoption.
Patient Access and Affordability
- Insurance Formulary Status: Eucrisa's placement on insurance formularies, including tier status and prior authorization requirements, directly affects its accessibility. Higher formulary tiers or significant prior authorization hurdles can limit uptake.
- Co-pay Assistance Programs: Pfizer offers co-pay assistance programs to reduce out-of-pocket costs for eligible commercially insured patients. The structure and generosity of these programs influence patient affordability and adherence.
- Out-of-Pocket Costs: For patients with high deductibles or limited insurance coverage, the out-of-pocket cost of Eucrisa can be a barrier to treatment.
Competition
The atopic dermatitis market is highly competitive.
- Topical Corticosteroids: These remain the first-line treatment for many patients with atopic dermatitis due to their broad efficacy and low cost. While Eucrisa offers a non-steroidal alternative, it must demonstrate a clear benefit or comparable efficacy with a superior safety profile to displace corticosteroid use consistently.
- Topical Calcineurin Inhibitors (TCIs): Drugs like tacrolimus and pimecrolimus are also non-steroidal options, primarily used for facial or intertriginous areas, or when corticosteroids are contraindicated.
- Newer Systemic and Biologic Therapies: For moderate-to-severe atopic dermatitis, therapies such as dupilumab (Dupixent) and JAK inhibitors have transformed treatment paradigms. While Eucrisa targets mild to moderate disease, the severity spectrum of atopic dermatitis means patients may progress to these more potent agents, potentially limiting the long-term market for Eucrisa if disease control is not maintained.
- Emerging Topical Agents: Ongoing research and development in dermatology could introduce new topical therapies with novel mechanisms of action or improved efficacy profiles, further intensifying competition.
What is Eucrisa's Current Pricing Structure?
Eucrisa's pricing is a critical factor influencing its market position and commercial viability. Pharmaceutical pricing in the U.S. is complex, involving list prices, net prices after rebates, and patient out-of-pocket costs.
- List Price: The manufacturer's published price for Eucrisa is a benchmark. For example, the list price for a 60-gram tube of Eucrisa has historically been in the range of $650 to $700 USD [2, 3].
- Net Price: The actual revenue realized by Pfizer after accounting for rebates, discounts, and chargebacks to payers is the net price. Net price is generally lower than the list price.
- Payer Rebates: Pharmaceutical companies negotiate rebates with pharmacy benefit managers (PBMs) and health plans to secure favorable formulary placement. The size of these rebates can significantly impact the net price.
- Patient Co-pay: This is the amount a patient pays for Eucrisa after their insurance has paid its share. Co-pay amounts can range from nominal amounts (e.g., $0 to $25) for commercially insured patients with good coverage and co-pay assistance, to substantial amounts for uninsured or underinsured individuals.
Comparison with Competitors (Pricing Context)
Direct price comparisons are complex due to varying indications, dosages, and treatment durations. However, within the topical treatment landscape for atopic dermatitis:
- Topical Corticosteroids: Generally available as generics at significantly lower price points, often in the range of tens of dollars for a tube.
- Topical Calcineurin Inhibitors: Generic versions of tacrolimus and pimecrolimus are also available at lower costs than branded Eucrisa.
Eucrisa's higher price point is justified by its novel mechanism of action and non-steroidal profile, but this necessitates strong perceived clinical value and broad market access to overcome cost sensitivities.
What are the Price Projections for Eucrisa?
Projecting Eucrisa's future pricing involves considering several market dynamics, including competition, payer negotiations, and potential label expansions or market shifts.
Factors Influencing Future Pricing
- Sustained Market Share: If Eucrisa maintains or grows its market share for mild to moderate atopic dermatitis, the manufacturer may have leverage to maintain current pricing levels or implement modest annual increases, typical of the pharmaceutical industry in the U.S.
- Competitive Pricing Pressure: The introduction of new, effective, and potentially lower-cost topical or oral therapies for atopic dermatitis could force Eucrisa's pricing to become more competitive. If alternative treatments demonstrate superior efficacy, safety, or cost-effectiveness, payers may demand lower prices or favor competing products.
- Payer Negotiations: As PBMs and health plans face increasing cost pressures, they may demand larger rebates from Eucrisa's manufacturer or shift Eucrisa to higher formulary tiers with increased patient cost-sharing. This would effectively lower the net price received by the manufacturer.
- Patent Expiration: While Eucrisa was approved in 2016, specific patent protection timelines are critical. The onset of generic competition following patent expiry would lead to a significant decline in Eucrisa's price and market share. The earliest U.S. patent for crisaborole is expected to expire around 2030, with some data suggesting potential generic entry as early as 2027-2028 depending on specific patent challenges and settlements [4].
- Label Expansions: If Eucrisa were to receive approval for new indications (e.g., for younger age groups or different dermatological conditions), this could broaden its market and potentially influence its pricing strategy, though market dynamics for new indications would need to be assessed independently.
Price Projection Scenarios
- Scenario 1: Stable Market Position (Moderate Price Growth)
- Assumption: Eucrisa maintains its current role for mild to moderate atopic dermatitis, with steady but not dramatic prescription growth. Payer relationships remain relatively stable.
- Projection: Annual price increases consistent with pharmaceutical industry norms (e.g., 4-7% annually) may occur. The list price could reach approximately $800-$900 USD per 60g tube by 2028, with net prices adjusted by rebates.
- Scenario 2: Increased Competitive Pressure (Price Stabilization or Decline)
- Assumption: New, highly effective competitors emerge, or payers aggressively negotiate for lower prices. Generic alternatives to other topical therapies become more prominent.
- Projection: Price increases would be significantly curtailed, potentially leading to price stabilization or even modest declines in net price to maintain market share. List prices might remain flat or see minimal increases (e.g., 1-3% annually).
- Scenario 3: Pre-Generic Erosion (Declining Market Share and Price)
- Assumption: Approaching patent expiry, with anticipation of generic entry. Payers may begin to shift towards more cost-effective alternatives.
- Projection: This scenario is more relevant in the late 2020s. As generic crisaborole becomes available, the price of branded Eucrisa would likely decline significantly to compete, and its market share would diminish rapidly. Generic prices would typically fall by 70-90% from the branded product's peak price shortly after launch.
Current Price Range (as of late 2023/early 2024):
- List Price (60g tube): Approximately $680 - $720 USD [2, 3]
- Estimated Net Price (after rebates): Varies widely based on payer contracts, but generally significantly lower than list price.
- Patient Co-pay (with assistance): Often $10 - $30 USD for eligible commercially insured patients [3].
Projected List Price Range (5-year outlook, nominal growth):
- By 2028: $750 - $850 USD (assuming moderate annual price adjustments and stable market position) [2, 3].
Note: These projections are based on current market conditions and historical trends. Actual pricing may deviate based on unforeseen market events, regulatory changes, and competitive dynamics.
What is the Future Market Potential for Eucrisa?
Eucrisa's future market potential is largely tied to its ability to maintain its positioning for mild to moderate atopic dermatitis against evolving competition and pricing pressures, and the timing of potential generic entry.
- U.S. Market: The U.S. remains the primary market for Eucrisa. Its potential here hinges on continued physician and patient acceptance for its approved indication. The prevalence of atopic dermatitis in the U.S. is significant, estimated to affect millions of individuals, particularly children and adolescents [5]. However, the definition of "mild to moderate" and the accessibility of alternative treatments will constrain its addressable market.
- International Markets: Expansion into international markets would require separate regulatory approvals and tailored market access strategies, considering varying healthcare systems and competitive landscapes. Pfizer's global presence could facilitate this, but penetration would depend on local formulary adoption and pricing.
- Pipeline and Label Expansion: Any successful expansion of Eucrisa's label to new indications or patient populations could significantly enhance its market potential. However, to date, such expansions have not materialized for Eucrisa.
- Genericization Impact: The most significant threat to Eucrisa's market potential is the eventual arrival of generic competition. The exact timing and impact will depend on the strength of patent protection and the success of any legal challenges. Once generics are available, the market for branded Eucrisa will likely contract sharply.
Estimated U.S. Market Size for Topical Atopic Dermatitis Treatments (Mild-to-Moderate Segment):
- This segment represents a significant portion of the overall atopic dermatitis market, estimated to be worth several billion dollars annually in the U.S. Eucrisa's share of this segment is currently modest but has potential for growth if current trends continue and competitive pressures do not intensify dramatically before patent expiry.
Key Considerations for Future Market Access
- Demonstrating Value: Ongoing efforts to demonstrate Eucrisa's long-term value proposition, including potential reduction in the need for more expensive systemic therapies for patients who respond well, will be crucial for payer engagement.
- Real-World Evidence: Generating and disseminating robust real-world evidence on Eucrisa's effectiveness and safety in diverse patient populations can strengthen its market position and support payer negotiations.
- Patient Support Programs: The continued effectiveness of patient support programs in mitigating out-of-pocket costs will be essential for maintaining patient access and adherence, especially in a market where cost is a significant barrier for many.
Eucrisa's projected market potential will likely be characterized by a gradual increase in net sales leading up to potential patent challenges and generic entry, followed by a sharp decline once generic competition commences. The period between now and approximately 2027-2028 represents the window for maximizing its market penetration and revenue generation for the branded product.
Key Takeaways
- Eucrisa, a non-steroidal topical PDE4 inhibitor for mild to moderate atopic dermatitis, generated approximately $213 million in net sales in the U.S. in 2022.
- Its market performance is influenced by prescription volume, payer coverage, and competition from topical corticosteroids, calcineurin inhibitors, and newer systemic therapies.
- Eucrisa's list price for a 60-gram tube is around $680-$720 USD, with significant payer rebates and patient co-pay assistance programs impacting net revenue and patient affordability.
- Price projections anticipate moderate annual increases for the branded product in the near term, potentially reaching $750-$850 USD (list price) by 2028, assuming a stable market position.
- The market potential for Eucrisa is significantly influenced by the anticipated expiration of key patents around 2027-2028, which will likely lead to the introduction of generic competition and a substantial decline in branded product sales and pricing.
Frequently Asked Questions
1. What is the primary indication for Eucrisa?
Eucrisa is indicated for the topical treatment of mild to moderate atopic dermatitis in patients 12 years of age and older.
2. What is the mechanism of action of Eucrisa?
Eucrisa is a phosphodiesterase-4 (PDE4) inhibitor. By inhibiting PDE4, it is believed to reduce the release of pro-inflammatory cytokines and chemokines, thereby decreasing inflammation and itching associated with atopic dermatitis.
3. What is the typical out-of-pocket cost for patients using Eucrisa?
For eligible commercially insured patients, out-of-pocket costs with manufacturer co-pay assistance programs are often in the range of $10 to $30 per prescription. This can vary based on individual insurance plans and the terms of the assistance program.
4. When is generic Eucrisa expected to become available?
While specific patent expiry dates can be complex and subject to legal challenges, key patents for crisaborole are generally expected to expire around 2027-2028, which could allow for the introduction of generic versions of Eucrisa around that time.
5. How does Eucrisa compare in price to topical corticosteroids?
Eucrisa is priced significantly higher than most topical corticosteroids. Generic topical corticosteroids are typically available at a fraction of Eucrisa's price, often ranging from tens of dollars to under one hundred dollars per tube, depending on the specific steroid and formulation.
Cited Sources
- Pfizer Inc. (2017-2023). Annual Reports (10-K Filings). U.S. Securities and Exchange Commission.
- GoodRx. (n.d.). Eucrisa Prices, Coupons & Savings. Retrieved from https://www.goodrx.com/eucrisa
- SingleCare. (n.d.). Eucrisa Prices, Coupons & Savings Cards. Retrieved from https://www.singlecare.com/prescription/eucrisa
- Patent Litigation Data. (Various Sources, including analysis of Orange Book and patent dockets).
- National Eczema Association. (n.d.). Atopic Dermatitis Statistics. Retrieved from https://nationaleczema.org/eczema-statistics/
More… ↓
