FARXIGA Drug Patent Profile
✉ Email this page to a colleague
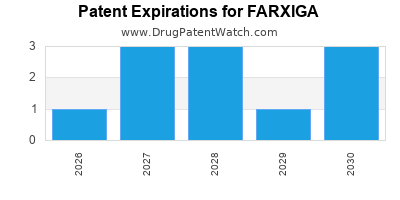
When do Farxiga patents expire, and when can generic versions of Farxiga launch?
Farxiga is a drug marketed by Astrazeneca Ab and is included in one NDA. There are seventeen patents protecting this drug and one Paragraph IV challenge.
This drug has four hundred and twenty-five patent family members in fifty-two countries.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this compound. Twenty-two suppliers are listed for this compound. Additional details are available on the dapagliflozin profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Farxiga
A generic version of FARXIGA was approved as dapagliflozin by AIZANT on April 6th, 2026.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for FARXIGA?
- What are the global sales for FARXIGA?
- What is Average Wholesale Price for FARXIGA?
Summary for FARXIGA
| International Patents: | 425 |
| US Patents: | 17 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 6 |
| Raw Ingredient (Bulk) Api Vendors: | 2 |
| Clinical Trials: | 50 |
| Patent Applications: | 1 |
| Drug Prices: | Drug price information for FARXIGA |
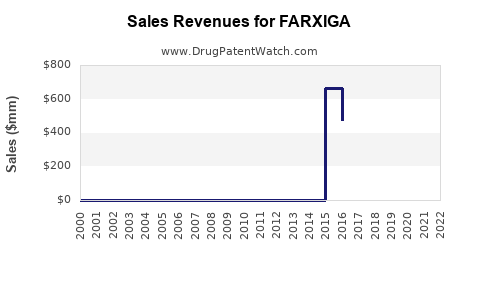
| Drug Sales Revenues: | Drug sales revenues for FARXIGA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for FARXIGA |
| What excipients (inactive ingredients) are in FARXIGA? | FARXIGA excipients list |
| DailyMed Link: | FARXIGA at DailyMed |
Recent Clinical Trials for FARXIGA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Han Xu, M.D., Ph.D., FAPCR, Sponsor-Investigator, IRB Chair | PHASE2 |
| UnitedHealthcare | PHASE2 |
| The University of Hong Kong | Phase 2 |
Pharmacology for FARXIGA
| Drug Class | Sodium-Glucose Cotransporter 2 Inhibitor |
| Mechanism of Action | Sodium-Glucose Transporter 2 Inhibitors |
Paragraph IV (Patent) Challenges for FARXIGA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| FARXIGA | Tablets | dapagliflozin | 5 mg and 10 mg | 202293 | 20 | 2018-01-08 |
US Patents and Regulatory Information for FARXIGA
FARXIGA is protected by seventeen US patents and four FDA Regulatory Exclusivities.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-002 | Jan 8, 2014 | AB | RX | Yes | Yes | 12,213,988*PED | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-002 | Jan 8, 2014 | AB | RX | Yes | Yes | 11,826,376*PED | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-002 | Jan 8, 2014 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-002 | Jan 8, 2014 | AB | RX | Yes | Yes | 11,903,955*PED | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-001 | Jan 8, 2014 | AB | RX | Yes | No | 8,221,786*PED | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-002 | Jan 8, 2014 | AB | RX | Yes | Yes | 7,851,502*PED | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for FARXIGA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-001 | Jan 8, 2014 | 6,936,590 | ⤷ Start Trial |
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-001 | Jan 8, 2014 | 6,495,164 | ⤷ Start Trial |
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-002 | Jan 8, 2014 | 7,741,269 | ⤷ Start Trial |
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-002 | Jan 8, 2014 | 9,238,076 | ⤷ Start Trial |
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-001 | Jan 8, 2014 | 6,414,126 | ⤷ Start Trial |
| Astrazeneca Ab | FARXIGA | dapagliflozin | TABLET;ORAL | 202293-001 | Jan 8, 2014 | 7,563,871 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for FARXIGA
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| AstraZeneca AB | Forxiga | dapagliflozin | EMEA/H/C/002322Type 2 diabetes mellitusForxiga is indicated in adults and children aged 10 years and above for the treatment of insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exerciseas monotherapy when metformin is considered inappropriate due to intolerance.in addition to other medicinal products for the treatment of type 2 diabetes.For study results with respect to combination of therapies, effects on glycaemic control, cardiovascular and renal events, and the populations studied, see sections 4.4, 4.5 and 5.1.Heart failureForxiga is indicated in adults for the treatment of symptomatic chronic heart failure.Chronic kidney diseaseForxiga is indicated in adults for the treatment of chronic kidney disease. | Authorised | no | no | no | 2012-11-11 | |
| AstraZeneca AB | Edistride | dapagliflozin | EMEA/H/C/004161Type 2 diabetes mellitusEdistride is indicated in adults and children aged 10 years and above for the treatment of insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exerciseas monotherapy when metformin is considered inappropriate due to intolerance.in addition to other medicinal products for the treatment of type 2 diabetes.For study results with respect to combination of therapies, effects on glycaemic control, cardiovascular and renal events, and the populations studied, see sections 4.4, 4.5 and 5.1.Heart failureEdistride is indicated in adults for the treatment of symptomatic chronic heart failure.Chronic kidney diseaseEdistride is indicated in adults for the treatment of chronic kidney disease. | Authorised | no | no | no | 2015-11-09 | |
| Viatris Limited | Dapagliflozin Viatris | dapagliflozin | EMEA/H/C/006006Type 2 diabetes mellitusDapagliflozin Viatris is indicated in adults and children aged 10 years and above for the treatment of insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exercise- as monotherapy when metformin is considered inappropriate due to intolerance.- in addition to other medicinal products for the treatment of type 2 diabetes.For study results with respect to combination of therapies, effects on glycaemic control, cardiovascular and renal events, and the populations studied, see sections 4.4, 4.5 and 5.1.Heart failureDapagliflozin Viatris is indicated in adults for the treatment of symptomatic chronic heart failure with reduced ejection fraction.Chronic kidney diseaseDapagliflozin Viatris is indicated in adults for the treatment of chronic kidney disease. | Authorised | yes | no | no | 2023-03-24 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for FARXIGA
When does loss-of-exclusivity occur for FARXIGA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 1730
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 07265246
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2017015106
Estimated Expiration: ⤷ Start Trial
Patent: 2017021516
Estimated Expiration: ⤷ Start Trial
Patent: 0713544
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 53344
Estimated Expiration: ⤷ Start Trial
Patent: 24318
Estimated Expiration: ⤷ Start Trial
Patent: 85797
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 07001915
Estimated Expiration: ⤷ Start Trial
China
Patent: 1479287
Estimated Expiration: ⤷ Start Trial
Patent: 3145773
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 60299
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0141007
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 15738
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 69374
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 8229
Estimated Expiration: ⤷ Start Trial
Patent: 0428
Estimated Expiration: ⤷ Start Trial
Patent: 8259
Estimated Expiration: ⤷ Start Trial
Patent: 5999
Estimated Expiration: ⤷ Start Trial
Patent: 0900066
Estimated Expiration: ⤷ Start Trial
Patent: 1171333
Estimated Expiration: ⤷ Start Trial
Patent: 1490902
Estimated Expiration: ⤷ Start Trial
Patent: 1791254
Estimated Expiration: ⤷ Start Trial
Patent: 2091391
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 69374
Estimated Expiration: ⤷ Start Trial
Patent: 57918
Estimated Expiration: ⤷ Start Trial
Patent: 45466
Estimated Expiration: ⤷ Start Trial
Patent: 63807
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 27359
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 5882
Estimated Expiration: ⤷ Start Trial
Patent: 4180
Estimated Expiration: ⤷ Start Trial
Patent: 4181
Estimated Expiration: ⤷ Start Trial
Patent: 4182
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 13889
Estimated Expiration: ⤷ Start Trial
Patent: 66651
Estimated Expiration: ⤷ Start Trial
Patent: 37187
Estimated Expiration: ⤷ Start Trial
Patent: 09545525
Estimated Expiration: ⤷ Start Trial
Patent: 13209394
Estimated Expiration: ⤷ Start Trial
Patent: 15071636
Estimated Expiration: ⤷ Start Trial
Patent: 16172758
Estimated Expiration: ⤷ Start Trial
Patent: 17222681
Estimated Expiration: ⤷ Start Trial
Patent: 19059779
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 8566
Estimated Expiration: ⤷ Start Trial
Patent: 3930
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 9143
Estimated Expiration: ⤷ Start Trial
Patent: 7155
Estimated Expiration: ⤷ Start Trial
Patent: 08015377
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 4346
Estimated Expiration: ⤷ Start Trial
Patent: 9190
Estimated Expiration: ⤷ Start Trial
Patent: 9195
Estimated Expiration: ⤷ Start Trial
Patent: 9202
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 6828
Estimated Expiration: ⤷ Start Trial
Patent: 7770
Estimated Expiration: ⤷ Start Trial
Patent: 085169
Estimated Expiration: ⤷ Start Trial
Patent: 221233
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 080349
Estimated Expiration: ⤷ Start Trial
Patent: 120776
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 012500168
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (IS)-1,5-ANHYDRO-L-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 69374
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 69374
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 638
Patent: KRISTALNI SOLVATI DERIVATA (1S)-1,5-ANHIDRO-1-C-(3-((FENIL) METIL) FENIL)-D-GLUCITOLA SA ALKOHOLIMA KAO INHIBITORI SGLT2 ZA TRETMAN DIJABETESA (CRYSTALLINE SOLVATES OF (1S)-1,5-ANHYDRO-1-C-(3-((PHENYL) METHYL) PHENYL)-D-GLUCITOL DERIVATIVES WITH ALCOHOLS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201402181S
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (1S) -1, 5-ANHYDRO-1-C- (3- ( (PHENYL) METHYL) PHENYL) -D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Start Trial
Patent: 2741
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (1S) -1, 5-ANHYDRO-1-C- (3- ( (PHENYL) METHYL) PHENYL) -D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 69374
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 0810475
Patent: CRYSTALLINE SOLVATES AND COMPLEXES OF (IS)-1,5-ANHYDRO-L-C-(3-((PHENYL)METHYL)PHENYL)-D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1493102
Estimated Expiration: ⤷ Start Trial
Patent: 090023643
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 21665
Estimated Expiration: ⤷ Start Trial
Patent: 59862
Estimated Expiration: ⤷ Start Trial
Patent: 69130
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 0811127
Patent: Crystal structures of SGLT2 inhibitors and processes for preparing same
Estimated Expiration: ⤷ Start Trial
Patent: 1406743
Patent: Crystal structures of SGLT2 inhibitors and processes for preparing same
Estimated Expiration: ⤷ Start Trial
Patent: 1509927
Patent: Crystal structures of SGLT2 inhibitors and processes for preparing same
Estimated Expiration: ⤷ Start Trial
Patent: 1546054
Patent: Crystal structures of SGLT2 inhibitors and processes for preparing same
Estimated Expiration: ⤷ Start Trial
Patent: 21245
Estimated Expiration: ⤷ Start Trial
Patent: 66876
Estimated Expiration: ⤷ Start Trial
Patent: 19528
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 765
Patent: КРИСТАЛЛИЧЕСКИЕ СОЛЬВАТЫ И КОМПЛЕКСЫ ПРОИЗВОДНЫХ (IS)-1,5-АНГИДРО-L-C-(3-((ФЕНИЛ)МЕТИЛ)ФЕНИЛ)-D-ГЛЮЦИТОЛА С АМИНОКИСЛОТАМИ КАК ИНГИБИТОРЫ БЕЛКА SGLT2, ПРИГОДНЫЕ В ЛЕЧЕНИИ ДИАБЕТА (CRYSTALLINE SOLVATES AND COMPLEXES OF (IS) -1, 5-ANHYDRO-L-C- (3- ((PHENYL) METHYL) PHENYL) -D-GLUCITOL DERIVATIVES WITH AMINO ACIDS AS SGLT2 INHIBITORS FOR THE TREATMENT OF DIABETES)
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering FARXIGA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Mexico | 2009010155 | FORMULACIONES FARMACEUTICAS QUE CONTIENEN HIDRATO DE PROPILENGLICOL DE DAPAGLIFLOZINA. (PHARMACEUTICAL FORMULATIONS CONTAINING DAPAGLIFLOZIN PROPYLENE GLYCOL HYDRATE.) | ⤷ Start Trial |
| Denmark | 1971362 | ⤷ Start Trial | |
| Spain | 2280759 | ⤷ Start Trial | |
| Japan | 5693817 | ⤷ Start Trial | |
| Japan | 2003519667 | ⤷ Start Trial | |
| Cyprus | 1116071 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for FARXIGA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1506211 | C01506211/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: DAPAGLIFLOZIN; REGISTRATION NO/DATE: SWISSMEDIC 65176 19.08.2014 |
| 2498758 | 2020C/509 | Belgium | ⤷ Start Trial | PRODUCT NAME: QTRILMET - METFORMINE OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELLE-CI; SAXAGLIPTINE OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELLE-CI; DAPAGLIFLOZINE OU UN SOLVATE PHARMACEUTIQUEMENT ACCEPTABLE DE CELLE-CI; AUTHORISATION NUMBER AND DATE: EU/1/19/1401 20191113 |
| 1506211 | 18/2013 | Austria | ⤷ Start Trial | PRODUCT NAME: DAPAGLIFLOZIN UND PHARMAZEUTISCH VERTRAEGLICHE SALZE DAVON; REGISTRATION NO/DATE: EU/1/12/795/001 - EU/1/12/795/010 20121112 |
| 1506211 | C20140021 00131 | Estonia | ⤷ Start Trial | PRODUCT NAME: DAPAGLIFLOSIIN/METFORMIIN;REG NO/DATE: K(2014)308 (LOPLIK) 21.01.2014 |
| 0996459 | CA 2007 00034 | Denmark | ⤷ Start Trial | |
| 2139494 | 122020000043 | Germany | ⤷ Start Trial | PRODUCT NAME: SAXAGLIPTIN UND DAPAGLIFLOZIN; REGISTRATION NO/DATE: EU/1/16/1108 20160715 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for FARXIGA (Dapagliflozin)
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.