Expiring Drug Patents Cheat Sheet
We analyse the patents covering drugs in 134 countries and quickly give you the likely loss-of-exclusivity/generic entry date
Russian Federation: These 24 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "Russian Federation: These 24 Drugs Face Patent Expirations and Generic Entry From 2026 - 2027" DrugPatentWatch.com thinkBiotech, 2026 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Russian Federation Patent 2,009,101,082
Patent Title: ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ (PHARMACEUTICAL COMPOSITIONS)
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 19, 2026
Generic Entry Controlled by: Russian Federation Patent 2,445,077
Patent Title: PHARMACEUTICAL COMPOSITIONS
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can EXFORGE HCT (amlodipine besylate; hydrochlorothiazide; valsartan) generic drug versions launch?
Generic name: amlodipine besylate; hydrochlorothiazide; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 27, 2026
Generic Entry Controlled by: Russian Federation Patent 2,009,102,273

This drug has seventy-four patent family members in twenty-three countries. There has been litigation on patents covering EXFORGE HCT
See drug price trends for EXFORGE HCT.
The generic ingredient in EXFORGE HCT is amlodipine besylate; hydrochlorothiazide; valsartan. There are fifty drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the amlodipine besylate; hydrochlorothiazide; valsartan profile page.
When can EXFORGE HCT (amlodipine besylate; hydrochlorothiazide; valsartan) generic drug versions launch?
Generic name: amlodipine besylate; hydrochlorothiazide; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 27, 2026
Generic Entry Controlled by: Russian Federation Patent 2,449,786

This drug has seventy-four patent family members in twenty-three countries. There has been litigation on patents covering EXFORGE HCT
See drug price trends for EXFORGE HCT.
The generic ingredient in EXFORGE HCT is amlodipine besylate; hydrochlorothiazide; valsartan. There are fifty drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the amlodipine besylate; hydrochlorothiazide; valsartan profile page.
When can BYFAVO (remimazolam besylate) generic drug versions launch?
Generic name: remimazolam besylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 10, 2026
Generic Entry Controlled by: Russian Federation Patent 2,009,104,311
Patent Title: БЕНЗОДИАЗЕПИНОВЫЕ СОЛИ КРАТКОВРЕМЕННОГО ДЕЙСТВИЯ И ИХ ПОЛИМОРФНЫЕ ФОРМЫ
BYFAVO is a drug marketed by Acacia. There are eleven patents protecting this drug.
This drug has fifty-three patent family members in twenty-four countries. There has been litigation on patents covering BYFAVO
See drug price trends for BYFAVO.
The generic ingredient in BYFAVO is remimazolam besylate. One supplier is listed for this generic product. Additional details are available on the remimazolam besylate profile page.
When can BYFAVO (remimazolam besylate) generic drug versions launch?
Generic name: remimazolam besylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 10, 2026
Generic Entry Controlled by: Russian Federation Patent 2,470,935
Patent Title: БЕНЗОДИАЗЕПИНОВЫЕ СОЛИ КРАТКОВРЕМЕННОГО ДЕЙСТВИЯ И ИХ ПОЛИМОРФНЫЕ ФОРМЫ (SHORT-TERM ACTION BENZODIAZEPINE SALTS AND POLYMORPHS THEREOF)
BYFAVO is a drug marketed by Acacia. There are eleven patents protecting this drug.
This drug has fifty-three patent family members in twenty-four countries. There has been litigation on patents covering BYFAVO
See drug price trends for BYFAVO.
The generic ingredient in BYFAVO is remimazolam besylate. One supplier is listed for this generic product. Additional details are available on the remimazolam besylate profile page.
When can REZDIFFRA (resmetirom) generic drug versions launch?
Generic name: resmetirom
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 11, 2026
Generic Entry Controlled by: Russian Federation Patent 2,379,295
Patent Title: ПРОИЗВОДНЫЕ ПИРИДАЗИНОНА В КАЧЕСТВЕ АГОНИСТОВ РЕЦЕПТОРА ТИРЕОИДНОГО ГОРМОНА (PYRIDASINONE DERIVATIVES AS AGONISTS OF THYROID HORMONE RECEPTOR)
REZDIFFRA is a drug marketed by Madrigal. There are six patents protecting this drug.
This drug has one hundred and twenty-six patent family members in thirty-seven countries.
See drug price trends for REZDIFFRA.
The generic ingredient in REZDIFFRA is resmetirom. One supplier is listed for this generic product. Additional details are available on the resmetirom profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Russian Federation Patent 2,009,104,949
Patent Title: КОМБИНАЦИЯ АДАПАЛЕНА И ПЕРОКСИДА БЕНЗОИЛА ДЛЯ ЛЕЧЕНИЯ ПОРАЖЕНИЙ АКНЕ
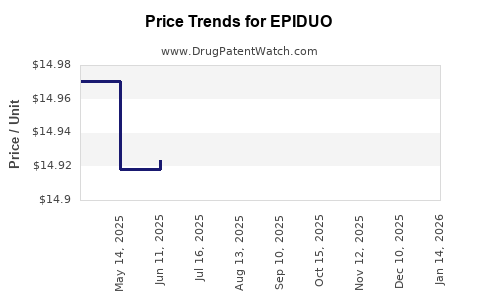
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Russian Federation Patent 2,012,136,952
Patent Title: КОМБИНАЦИЯ АДАПАЛЕНА И ПЕРОКСИДА БЕНЗОИЛА ДЛЯ ЛЕЧЕНИЯ ПОРАЖЕНИЙ АКНЕ

This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Russian Federation Patent 2,012,144,414
Patent Title: КОМБИНАЦИЯ АДАПАЛЕНА И ПЕРОКСИДА БЕНЗОИЛА ДЛЯ ЛЕЧЕНИЯ ПОРАЖЕНИЙ АКНЕ

This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Russian Federation Patent 2,009,104,949
Patent Title: КОМБИНАЦИЯ АДАПАЛЕНА И ПЕРОКСИДА БЕНЗОИЛА ДЛЯ ЛЕЧЕНИЯ ПОРАЖЕНИЙ АКНЕ
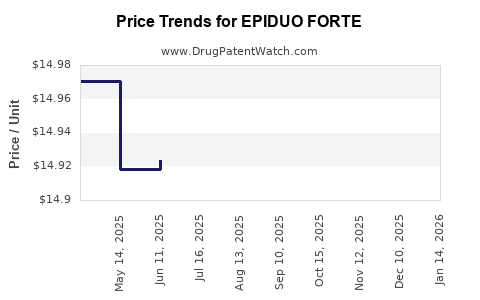
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO FORTE
See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Russian Federation Patent 2,012,136,952
Patent Title: КОМБИНАЦИЯ АДАПАЛЕНА И ПЕРОКСИДА БЕНЗОИЛА ДЛЯ ЛЕЧЕНИЯ ПОРАЖЕНИЙ АКНЕ

This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO FORTE
See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 13, 2026
Generic Entry Controlled by: Russian Federation Patent 2,012,144,414
Patent Title: КОМБИНАЦИЯ АДАПАЛЕНА И ПЕРОКСИДА БЕНЗОИЛА ДЛЯ ЛЕЧЕНИЯ ПОРАЖЕНИЙ АКНЕ

This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO FORTE
See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can RASUVO (methotrexate) generic drug versions launch?
Generic name: methotrexate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 21, 2026
Generic Entry Controlled by: Russian Federation Patent 2,009,106,053
Patent Title: КОНЦЕНТРИРОВАННЫЕ РАСТВОРЫ МЕТОТРЕКСАТА
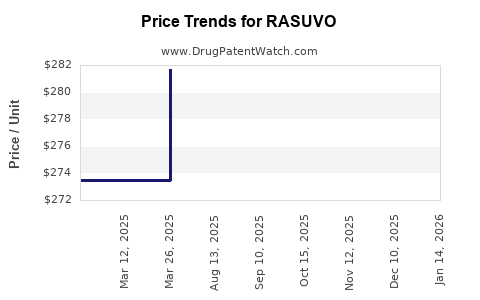
This drug has twenty-nine patent family members in twenty-one countries. There has been litigation on patents covering RASUVO
See drug price trends for RASUVO.
The generic ingredient in RASUVO is methotrexate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the methotrexate profile page.
When can RASUVO (methotrexate) generic drug versions launch?
Generic name: methotrexate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 21, 2026
Generic Entry Controlled by: Russian Federation Patent 2,403,044
Patent Title: КОНЦЕНТРИРОВАННЫЕ РАСТВОРЫ МЕТОТРЕКСАТА (CONCENTRATED METHOTREXATE SOLUTIONS)

This drug has twenty-nine patent family members in twenty-one countries. There has been litigation on patents covering RASUVO
See drug price trends for RASUVO.
The generic ingredient in RASUVO is methotrexate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the methotrexate profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 15, 2026
Generic Entry Controlled by: Russian Federation Patent 2,008,109,651
Patent Title: ЯДРА МИКРОПЕЛЛЕТ ПАНКРЕАТИНА, ПРИГОДНЫЕ ДЛЯ НАНЕСЕНИЯ ЭНТЕРОСОЛЮБИЛЬНОГО ПОКРЫТИЯ
CREON is a drug marketed by
This drug has twenty-nine patent family members in twenty-one countries.
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 15, 2026
Generic Entry Controlled by: Russian Federation Patent 2,440,101
Patent Title: ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ РЕГУЛИРУЕМОГО ВЫСВОБОЖДЕНИЯ НЕСТАБИЛЬНЫХ В КИСЛОЙ СРЕДЕ ЛЕКАРСТВЕННЫХ СРЕДСТВ (CONTROLLED RELEASE PHARMACEUTICAL COMPOSITIONS UNSTABLE IN MEDICINAL ACID MEDIUM)
CREON is a drug marketed by
This drug has twenty-nine patent family members in twenty-one countries.
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 21, 2026
Generic Entry Controlled by: Russian Federation Patent 2,009,104,330
Patent Title: КОМПОЗИЦИИ, ПРИГОДНЫЕ ДЛЯ ПЕРОРАЛЬНОГО ВВЕДЕНИЯ, СОДЕРЖАЩИЕ ПРОИЗВОДНОЕ ТРИАЗОЛО[4,5-D]ПИРИМИДИНА (COMPOSITIONS APPLICABLE FOR ORAL ADMINISTRATION AND CONTAINING TRIAZOLO[4,5-d]PYRIMIDINE DERIVATIVE)
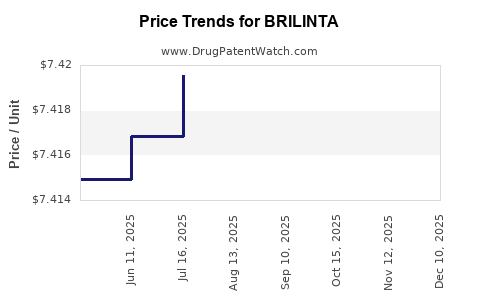
This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 21, 2026
Generic Entry Controlled by: Russian Federation Patent 2,012,153,069
Patent Title: КОМПОЗИЦИИ, ПРИГОДНЫЕ ДЛЯ ПЕРОРАЛЬНОГО ВВЕДЕНИЯ, СОДЕРЖАЩИЕ ПРОИЗВОДНОЕ ТРИАЗОЛО[4,5-d]ПИРИМИДИНА

This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 21, 2026
Generic Entry Controlled by: Russian Federation Patent 2,476,223
Patent Title: КОМПОЗИЦИИ, ПРИГОДНЫЕ ДЛЯ ПЕРОРАЛЬНОГО ВВЕДЕНИЯ, СОДЕРЖАЩИЕ ПРОИЗВОДНОЕ ТРИАЗОЛО[4,5-D]ПИРИМИДИНА (COMPOSITIONS APPLICABLE FOR ORAL ADMINISTRATION AND CONTAINING TRIAZOLO[4,5-d]PYRIMIDINE DERIVATIVE)

This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 29, 2026
Generic Entry Controlled by: Russian Federation Patent 2,009,111,378
Patent Title: ТЕРАПЕВТИЧЕСКИЕ КОМПОЗИЦИИ, СОДЕРЖАЩИЕ СПЕЦИФИЧЕСКИЙ АНТАГОНИСТ РЕЦЕПТОРА ЭНДОТЕЛИНА И ИНГИБИТОРОВ PDE5 (THERAPEUTIC COMPOSITIONS CONTAINING SPECIFIC ENDOTHELIN RECEPTOR ANTAGONIST AND PDE5 INHIBITOR)
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the macitentan profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 29, 2026
Generic Entry Controlled by: Russian Federation Patent 2,462,249
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the macitentan profile page.
When can TYMLOS (abaloparatide) generic drug versions launch?
Generic name: abaloparatide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 03, 2026
Generic Entry Controlled by: Russian Federation Patent 2,506,070
Patent Title: METHOD OF TREATING OSTEOPOROSIS AND COMPOSITION USED THEREIN

This drug has fifty-six patent family members in twenty-nine countries. There has been litigation on patents covering TYMLOS
See drug price trends for TYMLOS.
The generic ingredient in TYMLOS is abaloparatide. One supplier is listed for this generic product. Additional details are available on the abaloparatide profile page.
When can INREBIC (fedratinib hydrochloride) generic drug versions launch?
Generic name: fedratinib hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: Russian Federation Patent 2,008,121,807
INREBIC is a drug marketed by Bristol-myers. There are five patents protecting this drug.
This drug has one hundred and nineteen patent family members in forty-two countries. There has been litigation on patents covering INREBIC
See drug price trends for INREBIC.
The generic ingredient in INREBIC is fedratinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the fedratinib hydrochloride profile page.
When can INREBIC (fedratinib hydrochloride) generic drug versions launch?
Generic name: fedratinib hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: Russian Federation Patent 2,012,103,850
INREBIC is a drug marketed by Bristol-myers. There are five patents protecting this drug.
This drug has one hundred and nineteen patent family members in forty-two countries. There has been litigation on patents covering INREBIC
See drug price trends for INREBIC.
The generic ingredient in INREBIC is fedratinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the fedratinib hydrochloride profile page.
When can INREBIC (fedratinib hydrochloride) generic drug versions launch?
Generic name: fedratinib hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: Russian Federation Patent 2,012,103,851
INREBIC is a drug marketed by Bristol-myers. There are five patents protecting this drug.
This drug has one hundred and nineteen patent family members in forty-two countries. There has been litigation on patents covering INREBIC
See drug price trends for INREBIC.
The generic ingredient in INREBIC is fedratinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the fedratinib hydrochloride profile page.
When can INREBIC (fedratinib hydrochloride) generic drug versions launch?
Generic name: fedratinib hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: Russian Federation Patent 2,448,959
INREBIC is a drug marketed by Bristol-myers. There are five patents protecting this drug.
This drug has one hundred and nineteen patent family members in forty-two countries. There has been litigation on patents covering INREBIC
See drug price trends for INREBIC.
The generic ingredient in INREBIC is fedratinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the fedratinib hydrochloride profile page.
When can INREBIC (fedratinib hydrochloride) generic drug versions launch?
Generic name: fedratinib hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: Russian Federation Patent 2,589,878
INREBIC is a drug marketed by Bristol-myers. There are five patents protecting this drug.
This drug has one hundred and nineteen patent family members in forty-two countries. There has been litigation on patents covering INREBIC
See drug price trends for INREBIC.
The generic ingredient in INREBIC is fedratinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the fedratinib hydrochloride profile page.
When can INREBIC (fedratinib hydrochloride) generic drug versions launch?
Generic name: fedratinib hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 26, 2026
Generic Entry Controlled by: Russian Federation Patent 2,597,364
Patent Title: БИ-АРИЛ-МЕТА-ПИРИМИДИНОВЫЕ ИНГИБИТОРЫ КИНАЗ (BI-ARYL-META-PYRIMIDINE KINASE INHIBITORS)
INREBIC is a drug marketed by Bristol-myers. There are five patents protecting this drug.
This drug has one hundred and nineteen patent family members in forty-two countries. There has been litigation on patents covering INREBIC
See drug price trends for INREBIC.
The generic ingredient in INREBIC is fedratinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the fedratinib hydrochloride profile page.
When can BEVYXXA (betrixaban) generic drug versions launch?
Generic name: betrixaban
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 07, 2026
Generic Entry Controlled by: Russian Federation Patent 2,008,123,055
Patent Title: СОЛИ ИНГИБИТОРА ФАКТОРА Ха, СПОСОБ ИХ ПОЛУЧЕНИЯ, ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ НА ИХ ОСНОВЕ, СПОСОБ ЛЕЧЕНИЯ ТРОМБОЗА И СПОСОБ ИНГИБИРОВАНИЯ КОАГУЛЯЦИИ ОБРАЗЦОВ КРОВИ
BEVYXXA is a drug marketed by Portola Pharms Inc. There are five patents protecting this drug.
This drug has sixty-seven patent family members in twenty-six countries. There has been litigation on patents covering BEVYXXA
See drug price trends for BEVYXXA.
The generic ingredient in BEVYXXA is betrixaban. Additional details are available on the betrixaban profile page.
When can BEVYXXA (betrixaban) generic drug versions launch?
Generic name: betrixaban
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 07, 2026
Generic Entry Controlled by: Russian Federation Patent 2,440,986
Patent Title: СОЛЬ ИНГИБИТОРА ФАКТОРА Ха, СПОСОБ ЕЕ ПОЛУЧЕНИЯ, ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ НА ЕЕ ОСНОВЕ, СОСТОЯЩИЕ ИЗ НАЗВАННОЙ КОМПОЗИЦИИ ТАБЛЕТКА, КАПСУЛА И ЛЕПЕШКА, СПОСОБ ЛЕЧЕНИЯ ТРОМБОЗА И СПОСОБ ИНГИБИРОВАНИЯ КОАГУЛЯЦИИ ОБРАЗЦОВ КРОВИ (FACTOR Xa INHIBITOR SALT, METHOD FOR MAKING IT, BASED PHARMACEUTICAL COMPOSITION, TABLET, CAPSULE AND LOZENGE CONSISTING OF THIS COMPOSITION, METHOD FOR TREATING THROMBOSIS AND METHOD FOR INHIBITING BLOOD SAMPLE COAGULATION)
BEVYXXA is a drug marketed by Portola Pharms Inc. There are five patents protecting this drug.
This drug has sixty-seven patent family members in twenty-six countries. There has been litigation on patents covering BEVYXXA
See drug price trends for BEVYXXA.
The generic ingredient in BEVYXXA is betrixaban. Additional details are available on the betrixaban profile page.
When can ENTRESTO SPRINKLE (sacubitril; valsartan) generic drug versions launch?
Generic name: sacubitril; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 08, 2026
Generic Entry Controlled by: Russian Federation Patent 2,007,123,671
Patent Title: ФАРМАЦЕВТИЧЕСКИЕ КОМБИНАЦИИ АНТАГОНИСТА РЕЦЕПТОРА АНГИОИЕНЗИНА И ИНГИБИТОРА NEP
This drug has one hundred and eight patent family members in thirty-nine countries. There has been litigation on patents covering ENTRESTO SPRINKLE
See drug price trends for ENTRESTO SPRINKLE.
The generic ingredient in ENTRESTO SPRINKLE is sacubitril; valsartan. There are eleven drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the sacubitril; valsartan profile page.
When can ENTRESTO SPRINKLE (sacubitril; valsartan) generic drug versions launch?
Generic name: sacubitril; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 08, 2026
Generic Entry Controlled by: Russian Federation Patent 2,503,668
Patent Title: ФАРМАЦЕВТИЧЕСКИЕ КОМБИНАЦИИ АНТАГОНИСТА РЕЦЕПТОРА АНГИОТЕНЗИНА И ИНГИБИТОРА NEP (PHARMACEUTICAL COMBINATIONS OF ANGIOTENSIN RECEPTOR ANTAGONIST AND NEP INHIBITOR)

This drug has one hundred and eight patent family members in thirty-nine countries. There has been litigation on patents covering ENTRESTO SPRINKLE
See drug price trends for ENTRESTO SPRINKLE.
The generic ingredient in ENTRESTO SPRINKLE is sacubitril; valsartan. There are eleven drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the sacubitril; valsartan profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 10, 2026
Generic Entry Controlled by: Russian Federation Patent 2,009,121,297
Patent Title: СИНТЕТИЧЕСКИЕ ПЕПТИДНЫЕ АМИДЫ (SYNTHETIC PEPTIDE AMIDES)
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 10, 2026
Generic Entry Controlled by: Russian Federation Patent 2,009,121,298
Patent Title: СИНТЕТИЧЕСКИЕ ПЕПТИДНЫЕ АМИДЫ И ИХ ДИМЕРЫ (SYNTHETIC PEPTIDE AMIDES AND THEIR DIMERS)
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 10, 2026
Generic Entry Controlled by: Russian Federation Patent 2,500,685
Patent Title: СИНТЕТИЧЕСКИЕ ПЕПТИДНЫЕ АМИДЫ (SYNTHETIC PEPTIDE AMIDES)
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can AXUMIN (fluciclovine f-18) generic drug versions launch?
Generic name: fluciclovine f-18
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 28, 2026
Generic Entry Controlled by: Russian Federation Patent 2,428,415
Patent Title: СОЕДИНЕНИЕ-ПРЕДШЕСТВЕННИК ОРГАНИЧЕСКОГО СОЕДИНЕНИЯ, МЕЧЕННОГО РАДИОАКТИВНЫМ ГАЛОГЕНОМ (RADIOACTIVE HALOGEN-LABELLED ORGANIC COMPOUND PRECURSOR)
AXUMIN is a drug marketed by Blue Earth. There are eight patents protecting this drug.
This drug has thirty patent family members in sixteen countries. There has been litigation on patents covering AXUMIN
The generic ingredient in AXUMIN is fluciclovine f-18. One supplier is listed for this generic product. Additional details are available on the fluciclovine f-18 profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Russian Federation Patent 2,009,135,038
Patent Title: СТАБИЛЬНЫЕ КОМПОЗИЦИИ ПИЩЕВАРИТЕЛЬНЫХ ФЕРМЕНТОВ (STABLE COMPOSITIONS OF DIGESTIVE ENZYMES)
PANCREAZE is a drug marketed by
This drug has thirty patent family members in sixteen countries.
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Russian Federation Patent 2,445,952
Patent Title: СТАБИЛЬНЫЕ КОМПОЗИЦИИ ПИЩЕВАРИТЕЛЬНЫХ ФЕРМЕНТОВ (STABLE COMPOSITIONS OF DIGESTIVE ENZYMES)
PANCREAZE is a drug marketed by
This drug has thirty patent family members in sixteen countries.
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Russian Federation Patent 2,009,135,038
Patent Title: СТАБИЛЬНЫЕ КОМПОЗИЦИИ ПИЩЕВАРИТЕЛЬНЫХ ФЕРМЕНТОВ (STABLE COMPOSITIONS OF DIGESTIVE ENZYMES)
ZENPEP is a drug marketed by
This drug has thirty patent family members in sixteen countries.
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: February 20, 2027
Generic Entry Controlled by: Russian Federation Patent 2,445,952
Patent Title: СТАБИЛЬНЫЕ КОМПОЗИЦИИ ПИЩЕВАРИТЕЛЬНЫХ ФЕРМЕНТОВ (STABLE COMPOSITIONS OF DIGESTIVE ENZYMES)
ZENPEP is a drug marketed by
This drug has thirty patent family members in sixteen countries.
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can VANFLYTA (quizartinib dihydrochloride) generic drug versions launch?
Generic name: quizartinib dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 16, 2027
Generic Entry Controlled by: Russian Federation Patent 2,441,011
Patent Title: СОЕДИНЕНИЯ ИМИДАЗОЛОТИАЗОЛА ДЛЯ ЛЕЧЕНИЯ ЗАБОЛЕВАНИЙ (IMIDAZOLOTHIAZOL DERIVATIVES FOR TREATING DISEASES)
VANFLYTA is a drug marketed by Daiichi Sankyo Inc. There are eleven patents protecting this drug.
This drug has one hundred and twenty-six patent family members in thirty-two countries.
See drug price trends for VANFLYTA.
The generic ingredient in VANFLYTA is quizartinib dihydrochloride. One supplier is listed for this generic product. Additional details are available on the quizartinib dihydrochloride profile page.
When can XTANDI (enzalutamide) generic drug versions launch?
Generic name: enzalutamide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: March 29, 2027
Generic Entry Controlled by: Russian Federation Patent 2,449,993
Patent Title: ДИАРИЛТИОГИДАНТОИНОВЫЕ СОЕДИНЕНИЯ (DIARYLTHIOHYDATOIC COMPOUNDS)
XTANDI is a drug marketed by Astellas. There are seven patents protecting this drug and one Paragraph IV challenge. Three tentatively approved generics are ready to enter the market.
This drug has one hundred and ninety-two patent family members in thirty-six countries. There has been litigation on patents covering XTANDI
See drug price trends for XTANDI.
The generic ingredient in XTANDI is enzalutamide. There are nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the enzalutamide profile page.
When can INLYTA (axitinib) generic drug versions launch?
Generic name: axitinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 05, 2027
Generic Entry Controlled by: Russian Federation Patent 2,009,136,593
Patent Title: КРИСТАЛЛИЧЕСКИЕ ФОРМЫ 6-[2-(МЕТИЛКАРБАМОИЛ)ФЕНИЛСУЛЬФАНИЛ]-3-Е-[2-(ПИРИДИН-2-ИЛ)ЭТЕНИЛ]ИНДАЗОЛА, ПРИГОДНЫЕ ДЛЯ ЛЕЧЕНИЯ АНОМАЛЬНОГО РОСТА КЛЕТОК У ИЛЕКОПИТАЮШИХ
INLYTA is a drug marketed by Pf Prism Cv. There are four patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has ninety-seven patent family members in thirty countries. There has been litigation on patents covering INLYTA
See drug price trends for INLYTA.
The generic ingredient in INLYTA is axitinib. There are four drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the axitinib profile page.
When can SYMDEKO (COPACKAGED) (ivacaftor; ivacaftor, tezacaftor) generic drug versions launch?
Generic name: ivacaftor; ivacaftor, tezacaftor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 09, 2027
Generic Entry Controlled by: Russian Federation Patent 2,008,144,124
Patent Title: МОДУЛЯТОРЫ АТФ-СВЯЗЫВАЮЩИХ КАССЕТНЫХ ТРАНСПОРТЕРОВ
SYMDEKO (COPACKAGED) is a drug marketed by Vertex Pharms Inc. There are twenty-six patents protecting this drug.
This drug has four hundred and twenty-eight patent family members in forty-one countries. There has been litigation on patents covering SYMDEKO (COPACKAGED)
The generic ingredient in SYMDEKO (COPACKAGED) is ivacaftor; ivacaftor, tezacaftor. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivacaftor; ivacaftor, tezacaftor profile page.
When can SYMDEKO (COPACKAGED) (ivacaftor; ivacaftor, tezacaftor) generic drug versions launch?
Generic name: ivacaftor; ivacaftor, tezacaftor
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: April 09, 2027
Generic Entry Controlled by: Russian Federation Patent 2,451,018
Patent Title: МОДУЛЯТОРЫ АТФ-СВЯЗЫВАЮЩИХ КАССЕТНЫХ ТРАНСПОРТЕРОВ (MODULATORS OF ATP-BINDING CASSETTE TRANSPORTERS)
SYMDEKO (COPACKAGED) is a drug marketed by Vertex Pharms Inc. There are twenty-six patents protecting this drug.
This drug has four hundred and twenty-eight patent family members in forty-one countries. There has been litigation on patents covering SYMDEKO (COPACKAGED)
The generic ingredient in SYMDEKO (COPACKAGED) is ivacaftor; ivacaftor, tezacaftor. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivacaftor; ivacaftor, tezacaftor profile page.
When can TEKTURNA HCT (aliskiren hemifumarate; hydrochlorothiazide) generic drug versions launch?
Generic name: aliskiren hemifumarate; hydrochlorothiazide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 21, 2027
Generic Entry Controlled by: Russian Federation Patent 2,009,101,971
Patent Title: ГАЛЕНОВЫЙ СОСТАВ АЛИСКИРЕНА И ГИДРОХЛОРТИАЗИДА

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering TEKTURNA HCT
See drug price trends for TEKTURNA HCT.
The generic ingredient in TEKTURNA HCT is aliskiren hemifumarate; hydrochlorothiazide. There are four drug master file entries for this API. Additional details are available on the aliskiren hemifumarate; hydrochlorothiazide profile page.
When can TEKTURNA HCT (aliskiren hemifumarate; hydrochlorothiazide) generic drug versions launch?
Generic name: aliskiren hemifumarate; hydrochlorothiazide
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 21, 2027
Generic Entry Controlled by: Russian Federation Patent 2,491,058
Patent Title: ГАЛЕНОВЫЙ СОСТАВ АЛИСКИРЕНА И ГИДРОХЛОРТИАЗИДА (GALENA COMPOSITION OF ALISKIREN AND HYDROCHLOROTHIAZIDE)

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering TEKTURNA HCT
See drug price trends for TEKTURNA HCT.
The generic ingredient in TEKTURNA HCT is aliskiren hemifumarate; hydrochlorothiazide. There are four drug master file entries for this API. Additional details are available on the aliskiren hemifumarate; hydrochlorothiazide profile page.
When can OJEMDA (tovorafenib) generic drug versions launch?
Generic name: tovorafenib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 29, 2027
Generic Entry Controlled by: Russian Federation Patent 2,009,149,214
OJEMDA is a drug marketed by Day One Biopharms. There are two patents protecting this drug.
This drug has fifty patent family members in twenty-seven countries.
The generic ingredient in OJEMDA is tovorafenib. One supplier is listed for this generic product. Additional details are available on the tovorafenib profile page.
When can OJEMDA (tovorafenib) generic drug versions launch?
Generic name: tovorafenib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 29, 2027
Generic Entry Controlled by: Russian Federation Patent 2,492,166
OJEMDA is a drug marketed by Day One Biopharms. There are two patents protecting this drug.
This drug has fifty patent family members in twenty-seven countries.
The generic ingredient in OJEMDA is tovorafenib. One supplier is listed for this generic product. Additional details are available on the tovorafenib profile page.
Russian Federation Branded and Generic Drug Markets: Assessment, Regulatory Opportunities, and Challenges
More… ↓
DrugPatentWatch cited by CNN, NEJM, Nature Journals, and more …

Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
