Last updated: February 19, 2026
What is Vertex Pharmaceuticals Inc.'s Core Business and Market Focus?
Vertex Pharmaceuticals Inc. is a global biotechnology company focused on the discovery, development, and commercialization of transformative medicines for serious diseases. The company's primary therapeutic area is cystic fibrosis (CF), where it has established a dominant market position with its portfolio of CFTR modulators. Beyond CF, Vertex is actively developing novel therapies for other significant unmet medical needs, including sickle cell disease, beta-thalassemia, pain, and type 1 diabetes.
How Has Vertex Pharmaceuticals Inc. Achieved Its Dominant Position in Cystic Fibrosis?
Vertex's leadership in CF is a direct result of its sustained investment in understanding the disease at a molecular level and developing targeted therapies. The company’s strategy has focused on addressing the underlying cause of CF, a defective or missing cystic fibrosis transmembrane conductance regulator (CFTR) protein.
Key Milestones in Vertex's CF Portfolio:
- Kalydeco (ivacaftor): Approved in 2012, Kalydeco was the first medicine to treat the underlying cause of CF in patients with specific CFTR gene mutations. It targets the gating defect in the CFTR protein.
- Orkambi (lumacaftor/ivacaftor): Approved in 2015, Orkambi was the first combination therapy to address the most common CF-causing mutation, F508del, by also correcting protein folding and trafficking.
- Symdeko (tezacaftor/ivacaftor): Approved in 2018, Symdeko offered an improved option for certain F508del mutation carriers, building on the foundation of Orkambi.
- Trikafta (elexacaftor/tezacaftor/ivacaftor) and Kaftrio (in Europe): Approved in 2019 in the U.S. and 2020 in Europe, Trikafta is a triple-combination therapy that addresses approximately 90% of CF patients with at least one F508del mutation. This drug has significantly expanded Vertex's market reach and revenue generation.
Market Penetration and Revenue Generation:
Trikafta has been the primary driver of Vertex's recent revenue growth. Its broad applicability has led to high prescription rates and market penetration among eligible CF patients. In the first nine months of 2023, Vertex reported net revenue of $7.8 billion, with Trikafta accounting for the majority of this figure. The company has guided for 2023 full-year Trikafta sales between $10.5 billion and $10.7 billion. This strong performance highlights the significant unmet need and the efficacy of Vertex's differentiated treatment approach.
What are Vertex Pharmaceuticals Inc.'s Key Strengths?
Vertex's competitive advantages are rooted in its scientific expertise, product pipeline, intellectual property, and strategic partnerships.
Scientific and R&D Expertise:
Vertex possesses deep expertise in understanding disease biology at the molecular level, particularly for genetic diseases. This has enabled the company to identify and develop targeted therapies that address the root causes of disease, rather than just managing symptoms. Their sustained focus on CFTR biology has allowed for iterative improvements in their therapeutic approaches, leading to more effective and broadly applicable treatments.
Diversified Product Pipeline:
While CF remains the company's stronghold, Vertex is actively diversifying its portfolio into other high-unmet-need areas.
- Sickle Cell Disease (SCD) and Beta-Thalassemia (BT): Vertex is advancing its gene-editing therapies for SCD and BT, in collaboration with CRISPR Therapeutics. Exa-cel (Casgevy) has received regulatory approvals in the UK and US for treating these conditions, marking a significant milestone in gene therapy for inherited blood disorders. This represents a major expansion beyond CF into other genetic diseases.
- Pain: The company is developing non-opioid pain treatments, focusing on Nav1.8 inhibitors for moderate-to-severe acute and neuropathic pain. This addresses a critical public health concern by offering alternatives to addictive pain medications.
- Type 1 Diabetes (T1D): Vertex is pursuing a novel approach to T1D by developing stem cell-derived therapies to restore insulin production. This aims to offer a functional cure for T1D patients.
Intellectual Property Portfolio:
Vertex maintains a robust portfolio of patents protecting its key medicines and underlying technologies. This IP provides a significant barrier to entry for competitors and ensures market exclusivity for its innovative therapies, allowing for continued reinvestment in R&D. The company actively defends its patents, ensuring its market leadership is sustained.
Strategic Partnerships and Collaborations:
Vertex has a history of forming strategic alliances to accelerate development and broaden its therapeutic reach.
- CRISPR Therapeutics: The collaboration on exa-cel for SCD and BT is a prime example of leveraging cutting-edge gene-editing technology. This partnership has led to the first approved gene-editing therapies for these diseases.
- Moderna: Vertex has partnered with Moderna to develop a mRNA vaccine targeting the cause of CF. This represents an exploration of novel delivery mechanisms.
What are the Key Competitive Threats and Challenges for Vertex Pharmaceuticals Inc.?
Despite its strong market position, Vertex faces several competitive threats and challenges that require strategic navigation.
Competition in Cystic Fibrosis:
While Vertex dominates the CF market, other companies are developing therapies that target CF or its complications.
- AbbVie: AbbVie has been exploring its own CFTR modulators, though Vertex's current portfolio and pipeline offer significant advantages in terms of efficacy and patient coverage.
- Genentech (Roche): Genentech is developing therapies that target inflammation and mucus clearance in CF, addressing aspects of the disease that are not directly handled by CFTR modulators.
- Emerging Therapies: While not yet significant competitors, the potential for novel approaches to CF, including gene therapy or other small molecule therapies, poses a long-term threat if they offer superior efficacy or broader applicability.
Pipeline Execution and Regulatory Hurdles:
Vertex's expansion into new therapeutic areas, particularly with complex modalities like gene editing and cell therapy, carries inherent risks.
- Gene Therapy Risks: The development and commercialization of gene therapies, such as exa-cel, involve complex manufacturing processes, stringent regulatory requirements, and potential long-term safety monitoring. Any setbacks in clinical trials or manufacturing issues could delay market entry or impact commercial adoption.
- Regulatory Scrutiny: As Vertex brings novel therapies to market, it faces intense regulatory review. The approval pathways for gene therapies are still evolving, and unexpected hurdles could arise.
- Clinical Trial Success: The success of future pipeline assets in ongoing clinical trials is critical. Failure to demonstrate efficacy or safety in key indications could significantly impact future revenue streams and diversification efforts.
Pricing and Market Access:
The high cost of Vertex's CFTR modulators has led to scrutiny and challenges related to pricing and market access.
- Reimbursement Pressures: Payers globally are increasingly scrutinizing the cost-effectiveness of high-priced specialty drugs. Vertex must continue to demonstrate the value proposition of its therapies to secure favorable reimbursement.
- Global Market Access: Expanding access to its therapies in lower and middle-income countries presents a challenge, requiring innovative pricing strategies and distribution models.
Drug Discovery and Innovation Risks:
While Vertex has a strong track record, the inherent unpredictability of drug discovery means that not all research programs will yield successful therapies. Sustaining a pipeline of innovative drugs is a continuous challenge.
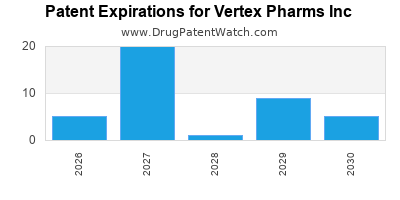
Patent Expirations and Generic Competition:
As the patents for Vertex's existing drugs expire, the company will face competition from generic manufacturers. While current CFTR modulators are protected by strong patents for many years, the long-term strategy must account for eventual patent cliffs.
What are Vertex Pharmaceuticals Inc.'s Strategic Priorities and Future Outlook?
Vertex's strategic priorities focus on maintaining leadership in CF while aggressively pursuing diversification and innovation in new therapeutic areas.
Maintaining CF Leadership:
Vertex aims to maximize the impact of its current CF portfolio by expanding patient access globally and continuing to monitor for potential improvements or new formulations. The company is also exploring therapies for the small percentage of CF patients who do not benefit from current CFTR modulators.
Advancing New Therapeutic Areas:
The successful development and commercialization of therapies in SCD, BT, pain, and T1D are critical for Vertex's long-term growth and diversification.
- SCD/BT Expansion: Following the initial approvals of exa-cel, Vertex will focus on expanding its label, improving manufacturing and delivery, and increasing patient access worldwide.
- Pain and T1D Development: Continued progress in clinical trials for its pain and T1D programs is essential. Achieving regulatory approval in these large, underserved markets would significantly expand Vertex's therapeutic footprint.
Investing in Next-Generation Technologies:
Vertex continues to invest in cutting-edge scientific platforms, including gene editing, cell therapy, and mRNA technology, to fuel its future pipeline. The company also maintains a strong commitment to its small molecule discovery engine.
Operational Excellence and Global Expansion:
Vertex is focused on optimizing its manufacturing capabilities, supply chains, and commercial operations to support its growing global presence and product launches. This includes building out its infrastructure in new geographic markets.
Financial Performance:
Vertex is well-positioned financially to fund its R&D initiatives and commercial expansion. The strong revenue generated from its CF portfolio provides a stable foundation for investing in its pipeline. The company's financial projections indicate continued revenue growth, driven primarily by Trikafta and the successful launch of its pipeline assets.
Key Takeaways
- Vertex Pharmaceuticals Inc. dominates the cystic fibrosis market with its CFTR modulator therapies, particularly Trikafta, which treats approximately 90% of CF patients with at least one F508del mutation.
- The company is aggressively diversifying its pipeline into sickle cell disease, beta-thalassemia, pain, and type 1 diabetes, leveraging cutting-edge technologies like gene editing and cell therapy.
- Key strengths include deep scientific expertise in genetic diseases, a robust IP portfolio, and strategic partnerships, notably with CRISPR Therapeutics for gene-editing therapies.
- Challenges include potential competition in CF, execution risks in developing complex new modalities, pricing pressures, and the inherent unpredictability of drug discovery.
- Vertex's strategy prioritizes maintaining CF leadership while successfully launching and expanding its diversified pipeline, supported by strong financial performance and investment in next-generation technologies.
Frequently Asked Questions
-
What is the primary therapeutic area where Vertex Pharmaceuticals Inc. generates the majority of its revenue?
Vertex Pharmaceuticals Inc. generates the majority of its revenue from its portfolio of cystic fibrosis (CF) treatments, with Trikafta (elexacaftor/tezacaftor/ivacaftor) being the leading contributor.
-
Which company is Vertex collaborating with on gene-editing therapies for sickle cell disease and beta-thalassemia?
Vertex Pharmaceuticals Inc. is collaborating with CRISPR Therapeutics on gene-editing therapies for sickle cell disease and beta-thalassemia, which have led to the development of exa-cel (Casgevy).
-
What is Vertex's strategy for addressing the portion of the cystic fibrosis patient population not covered by its current CFTR modulators?
Vertex is exploring novel approaches to address the small percentage of CF patients who do not benefit from its current CFTR modulators, indicating ongoing research into alternative or complementary therapies for this segment.
-
Beyond cystic fibrosis and blood disorders, what other therapeutic areas is Vertex Pharmaceuticals Inc. actively developing treatments for?
Vertex Pharmaceuticals Inc. is actively developing treatments for pain (specifically non-opioid pain relief) and type 1 diabetes (through stem cell-derived therapies to restore insulin production).
-
What is the expected impact of patent expirations on Vertex's future revenue streams, and how is the company preparing for this?
While Vertex's current CFTR modulators are protected by strong patents for many years, the company is preparing for eventual patent cliffs by investing in and diversifying its pipeline into new therapeutic areas and next-generation technologies, aiming to create new revenue streams that will succeed its currently patented drugs.
Citations
[1] Vertex Pharmaceuticals Incorporated. (2023). Third Quarter 2023 Results & Webcast. Retrieved from https://investors.vrtx.com/events-and-presentations/events/event-details/2023/Q3-2023-Results-Webcast/default.aspx (Specific financial data and guidance cited herein are derived from the materials presented during this event or publicly available SEC filings referenced by Vertex.)
[2] Vertex Pharmaceuticals Incorporated. (n.d.). Our Pipeline. Retrieved from https://www.vrtx.com/pipeline
[3] U.S. Food and Drug Administration. (2023). FDA Approves First Gene Therapy for Patients with Sickle Cell Disease. Retrieved from https://www.fda.gov/news-events/press-announcements/fda-approves-first-gene-therapy-patients-sickle-cell-disease
[4] U.S. Food and Drug Administration. (2019). FDA Approves Trikafta (elexacaftor/tezacaftor/ivacaftor) for Patients with Cystic Fibrosis. Retrieved from https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-trikafta-elexacaftortezacaftorivacaftor-patients-cystic-fibrosis