Last updated: February 19, 2026
What is DYANAVEL XR and its Therapeutic Indication?
DYANAVEL XR (amphetamine extended-release) is an extended-release formulation of amphetamine used for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in individuals aged 6 years and older. It is a Schedule II controlled substance. The extended-release mechanism is designed to provide a prolonged therapeutic effect, reducing the need for multiple daily doses.
The active ingredient, amphetamine, is a central nervous system stimulant. It works by increasing the levels of dopamine and norepinephrine in the brain, neurotransmitters that play a role in attention, focus, and impulse control.
Who is the Manufacturer and What is its Market Position?
DYANAVEL XR is manufactured by Takeda Pharmaceutical Company Limited, a global pharmaceutical company. Takeda acquired the drug as part of its acquisition of Shire Pharmaceuticals in 2019. Shire had originally developed and launched DYANAVEL XR.
In the ADHD market, DYANAVEL XR competes with a range of stimulant and non-stimulant medications. Key competitors include:
- Methylphenidate-based stimulants: Ritalin, Concerta, Daytrana, Focalin XR.
- Amphetamine-based stimulants: Adderall XR, Vyvanse, Adzenys XR-ODT, Evekeo.
- Non-stimulants: Strattera, Intuniv, Kapvay.
Takeda positions DYANAVEL XR as a once-daily oral liquid formulation, offering an alternative for patients who have difficulty swallowing pills or capsules. This differentiation is a key aspect of its market strategy, targeting a specific patient need within the broader ADHD landscape.
What is the Patent Landscape for DYANAVEL XR?
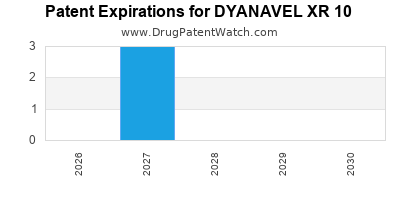
The patent landscape for DYANAVEL XR involves several key patents related to its formulation, manufacturing process, and methods of use. Understanding these patents is critical for assessing market exclusivity and potential for generic competition.
Key patents associated with DYANAVEL XR include:
- U.S. Patent No. 9,119,712: This patent, titled "Amphetamine Extended Release Formulations," is a foundational patent for the DYANAVEL XR extended-release technology. It was issued on August 4, 2015.
- U.S. Patent No. 9,295,579: Another patent related to extended-release formulations of amphetamine, issued on March 29, 2016.
- U.S. Patent No. 9,415,003: This patent, also covering amphetamine extended-release formulations, was issued on August 16, 2016.
- U.S. Patent No. 9,526,760: This patent, relating to methods of treating ADHD with amphetamine extended-release formulations, was issued on December 27, 2016.
- U.S. Patent No. 10,293,150: A more recent patent concerning amphetamine extended-release formulations, issued on May 21, 2019.
These patents, among others, provide Takeda with market exclusivity for DYANAVEL XR. The expiry dates of these patents will determine when generic manufacturers can enter the market. The primary formulation patents are crucial as they protect the core technology.
The earliest expiration dates for key formulation patents are typically considered the most critical for market entry timelines. While specific patent terms can be complex due to extensions, generally, patents filed in the early 2010s for this type of formulation would extend into the late 2020s or early 2030s.
Takeda also engages in patent litigation to defend its intellectual property. Challenges to patent validity or claims of infringement by generic manufacturers are common in the pharmaceutical industry. Any such litigation can significantly impact the timeline for generic market entry and the subsequent revenue trajectory of DYANAVEL XR.
What are the Sales and Revenue Performance Data?
Takeda's financial reporting provides insights into the commercial performance of DYANAVEL XR, often reported within its Neuroscience segment. The drug has demonstrated consistent revenue growth since its launch.
| Year |
Net Sales (USD Millions) |
| 2020 |
$153.7 |
| 2021 |
$209.8 |
| 2022 |
$267.4 |
| 2023 |
$336.5 |
Source: Takeda Pharmaceutical Company Limited Annual Reports
The data shows a compound annual growth rate (CAGR) of approximately 30% between 2020 and 2023, indicating strong market adoption and sales momentum. This growth is driven by increased prescribing, physician familiarity, and patient acceptance of the liquid extended-release formulation.
What are the Key Market Drivers and Challenges?
Market Drivers:
- Increasing ADHD Diagnosis Rates: The prevalence of ADHD continues to be a significant driver, with ongoing efforts to improve diagnosis and treatment access.
- Patient and Physician Preference for Once-Daily Dosing: The convenience of a single daily dose simplifies adherence and improves patient outcomes, a key selling point for DYANAVEL XR.
- Liquid Formulation Advantage: The oral liquid format addresses a specific unmet need for pediatric and adult patients who struggle with swallowing pills or capsules, expanding the addressable patient population.
- Takeda's Commercialization Strength: As a major pharmaceutical company, Takeda possesses the sales force and marketing infrastructure to effectively promote DYANAVEL XR to healthcare providers.
- Broad Age Indication: Approved for individuals aged 6 years and older, the drug has a wide patient demographic.
Market Challenges:
- Intense Competition: The ADHD market is highly saturated with numerous stimulant and non-stimulant treatment options, including established brands and emerging generics.
- Generic Competition Threat: As key patents approach expiry, the threat of generic entry looms. Generic versions of amphetamine and methylphenidate products can significantly reduce market share and pricing power.
- Controlled Substance Status: As a Schedule II controlled substance, DYANAVEL XR faces regulatory scrutiny, prescribing restrictions, and potential for diversion or abuse, which can impact market access and physician willingness to prescribe.
- Reimbursement and Payer Landscape: Navigating insurance formularies and obtaining favorable reimbursement status can be challenging, impacting patient access and out-of-pocket costs.
- Post-Acquisition Integration: While Takeda has integrated Shire's assets, ongoing management and optimization of the DYANAVEL XR portfolio within Takeda's broader therapeutic areas are crucial.
What is the Future Outlook and Financial Trajectory?
The future financial trajectory of DYANAVEL XR will be primarily shaped by the interplay of sustained market growth and the eventual impact of generic competition.
Near-term Outlook (1-3 years):
- Continued Growth: Expect continued revenue growth driven by the aforementioned market drivers. Takeda will likely continue to invest in marketing and patient support programs to maximize prescription volumes before patent expiry.
- Geographic Expansion: Potential for expanded indications or market approvals in regions outside of the U.S., though initial focus has been North America.
- Patent Defense: Takeda will actively defend its intellectual property through patent litigation. The outcome of these legal battles is critical for determining the timeline of generic entry.
Long-term Outlook (3+ years):
- Generic Entry Impact: Once key patents expire and generic versions become available, DYANAVEL XR's market share and pricing power will likely decline significantly, as is typical for branded pharmaceuticals. The speed and extent of this decline will depend on the number of generic entrants and their pricing strategies.
- Lifecycle Management: Takeda may explore lifecycle management strategies such as developing new formulations, combination therapies, or seeking label expansions to maintain revenue streams, though options for stimulant medications are often limited.
- Diversification: Takeda's overall financial performance will increasingly rely on its other product lines and pipeline assets as DYANAVEL XR faces generic erosion.
The financial trajectory of DYANAVEL XR is expected to be characterized by strong growth in the near to medium term, followed by a substantial decline in revenue post-generic entry. The specific timing and severity of this decline are contingent on patent expiry dates and the success of Takeda's patent defense strategies.
Key Takeaways
- DYANAVEL XR is an extended-release amphetamine formulation for ADHD, manufactured by Takeda Pharmaceutical Company Limited.
- The drug has demonstrated robust revenue growth, with net sales increasing from $153.7 million in 2020 to $336.5 million in 2023.
- Key patents protecting its formulation and methods of use extend into the late 2020s and early 2030s, providing market exclusivity.
- Market drivers include increasing ADHD diagnoses, preference for once-daily dosing, and its unique liquid formulation.
- Major challenges are intense market competition and the eventual threat of generic entry.
- The near-term financial outlook is positive, with continued growth anticipated, while the long-term outlook is subject to significant pressure from generic competition post-patent expiry.
Frequently Asked Questions
-
When do the core patents for DYANAVEL XR expire?
Core patents related to the extended-release formulation of DYANAVEL XR are generally expected to expire in the late 2020s to early 2030s, though specific expiry dates can vary based on patent extensions and litigation outcomes.
-
What is Takeda's strategy for defending DYANAVEL XR's market exclusivity?
Takeda's strategy primarily involves active defense of its intellectual property through patent litigation against potential generic challengers.
-
How does the liquid formulation of DYANAVEL XR differentiate it from competitors?
The oral liquid formulation provides an alternative for patients who have difficulty swallowing pills or capsules, addressing a specific patient subgroup within the ADHD market.
-
What is the projected impact of generic competition on DYANAVEL XR's revenue?
Upon the expiry of key patents and the entry of generic versions, DYANAVEL XR is expected to experience a significant decline in market share and revenue, a common trend for branded pharmaceuticals.
-
Are there any plans for expanding DYANAVEL XR's indications or developing new formulations?
While specific plans for label expansions or new formulations are proprietary, pharmaceutical companies often explore lifecycle management strategies to extend a drug's commercial life. Takeda's ongoing R&D pipeline will determine future developments.
Citations
[1] Takeda Pharmaceutical Company Limited. (2021). Takeda Annual Report 2020. https://www.takeda.com/investors/reports/annual-reports/
[2] Takeda Pharmaceutical Company Limited. (2022). Takeda Annual Report 2021. https://www.takeda.com/investors/reports/annual-reports/
[3] Takeda Pharmaceutical Company Limited. (2023). Takeda Annual Report 2022. https://www.takeda.com/investors/reports/annual-reports/
[4] Takeda Pharmaceutical Company Limited. (2024). Takeda Annual Report 2023. https://www.takeda.com/investors/reports/annual-reports/
[5] United States Patent and Trademark Office. (n.d.). Patent Search. Retrieved from https://patft.uspto.gov/