VELTASSA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Veltassa, and when can generic versions of Veltassa launch?
Veltassa is a drug marketed by Vifor Pharma and is included in one NDA. There are seven patents protecting this drug and one Paragraph IV challenge.
This drug has two hundred and six patent family members in twenty-seven countries.
The generic ingredient in VELTASSA is patiromer sorbitex calcium. One supplier is listed for this compound. Additional details are available on the patiromer sorbitex calcium profile page.
DrugPatentWatch® Generic Entry Outlook for Veltassa
Veltassa was eligible for patent challenges on October 21, 2019.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be May 29, 2030. This may change due to patent challenges or generic licensing.
There have been two patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for VELTASSA?
- What are the global sales for VELTASSA?
- What is Average Wholesale Price for VELTASSA?
Summary for VELTASSA
| International Patents: | 206 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 13 |
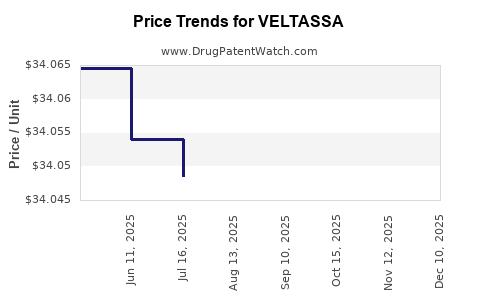
| Drug Prices: | Drug price information for VELTASSA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for VELTASSA |
| What excipients (inactive ingredients) are in VELTASSA? | VELTASSA excipients list |
| DailyMed Link: | VELTASSA at DailyMed |
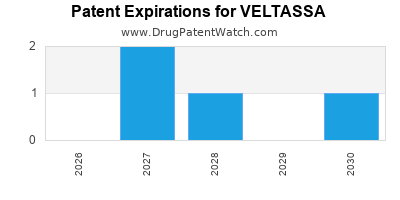
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for VELTASSA
Generic Entry Date for VELTASSA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
POWDER;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for VELTASSA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of California, Irvine | Phase 4 |
| University of Glasgow | Phase 4 |
| NHS Greater Glasgow and Clyde | Phase 4 |
Pharmacology for VELTASSA
| Drug Class | Potassium Binder |
| Mechanism of Action | Potassium Ion Binding Activity |
Paragraph IV (Patent) Challenges for VELTASSA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| VELTASSA | for Oral Suspension | patiromer sorbitex calcium | 8.4 g, 16.8 g and 25.2 g | 205739 | 2 | 2019-10-21 |
US Patents and Regulatory Information for VELTASSA
VELTASSA is protected by seven US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of VELTASSA is ⤷ Start Trial.
This potential generic entry date is based on patent 8,337,824.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vifor Pharma | VELTASSA | patiromer sorbitex calcium | POWDER;ORAL | 205739-003 | Oct 21, 2015 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Vifor Pharma | VELTASSA | patiromer sorbitex calcium | POWDER;ORAL | 205739-003 | Oct 21, 2015 | DISCN | Yes | No | 8,216,560 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Vifor Pharma | VELTASSA | patiromer sorbitex calcium | POWDER;ORAL | 205739-003 | Oct 21, 2015 | DISCN | Yes | No | 11,123,363 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Vifor Pharma | VELTASSA | patiromer sorbitex calcium | POWDER;ORAL | 205739-004 | Oct 2, 2023 | RX | Yes | No | 8,216,560 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Vifor Pharma | VELTASSA | patiromer sorbitex calcium | POWDER;ORAL | 205739-002 | Oct 21, 2015 | RX | Yes | Yes | 9,492,476 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Vifor Pharma | VELTASSA | patiromer sorbitex calcium | POWDER;ORAL | 205739-002 | Oct 21, 2015 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for VELTASSA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Vifor Pharma | VELTASSA | patiromer sorbitex calcium | POWDER;ORAL | 205739-003 | Oct 21, 2015 | 8,475,780 | ⤷ Start Trial |
| Vifor Pharma | VELTASSA | patiromer sorbitex calcium | POWDER;ORAL | 205739-003 | Oct 21, 2015 | 8,889,115 | ⤷ Start Trial |
| Vifor Pharma | VELTASSA | patiromer sorbitex calcium | POWDER;ORAL | 205739-001 | Oct 21, 2015 | 8,778,324 | ⤷ Start Trial |
| Vifor Pharma | VELTASSA | patiromer sorbitex calcium | POWDER;ORAL | 205739-003 | Oct 21, 2015 | 8,287,847 | ⤷ Start Trial |
| Vifor Pharma | VELTASSA | patiromer sorbitex calcium | POWDER;ORAL | 205739-004 | Oct 2, 2023 | 10,485,821 | ⤷ Start Trial |
| Vifor Pharma | VELTASSA | patiromer sorbitex calcium | POWDER;ORAL | 205739-002 | Oct 21, 2015 | 8,778,324 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for VELTASSA
When does loss-of-exclusivity occur for VELTASSA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 09282721
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0917853
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 35058
Estimated Expiration: ⤷ Start Trial
China
Patent: 2202670
Estimated Expiration: ⤷ Start Trial
Patent: 3919792
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0150955
Estimated Expiration: ⤷ Start Trial
Patent: 0181961
Estimated Expiration: ⤷ Start Trial
Patent: 0230091
Estimated Expiration: ⤷ Start Trial
Patent: 0250774
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 16850
Estimated Expiration: ⤷ Start Trial
Patent: 20930
Estimated Expiration: ⤷ Start Trial
Patent: 18002
Estimated Expiration: ⤷ Start Trial
Patent: 19002
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 65988
Estimated Expiration: ⤷ Start Trial
Patent: 57286
Estimated Expiration: ⤷ Start Trial
Patent: 31094
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 65988
Estimated Expiration: ⤷ Start Trial
Patent: 57286
Estimated Expiration: ⤷ Start Trial
Patent: 31094
Estimated Expiration: ⤷ Start Trial
Patent: 01408
Estimated Expiration: ⤷ Start Trial
Finland
Patent: 31094
Estimated Expiration: ⤷ Start Trial
Germany
Patent: 2009002063
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 84675
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 25934
Estimated Expiration: ⤷ Start Trial
Patent: 61320
Estimated Expiration: ⤷ Start Trial
Patent: 71829
Estimated Expiration: ⤷ Start Trial
Patent: 800004
Estimated Expiration: ⤷ Start Trial
Patent: 900003
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 56403
Estimated Expiration: ⤷ Start Trial
Patent: 83471
Estimated Expiration: ⤷ Start Trial
Patent: 32468
Estimated Expiration: ⤷ Start Trial
Patent: 33583
Estimated Expiration: ⤷ Start Trial
Patent: 90116
Estimated Expiration: ⤷ Start Trial
Patent: 12500806
Estimated Expiration: ⤷ Start Trial
Patent: 14144977
Estimated Expiration: ⤷ Start Trial
Patent: 14144978
Estimated Expiration: ⤷ Start Trial
Patent: 16145256
Estimated Expiration: ⤷ Start Trial
Patent: 17218454
Estimated Expiration: ⤷ Start Trial
Patent: 19065030
Estimated Expiration: ⤷ Start Trial
Patent: 20172537
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 957286
Estimated Expiration: ⤷ Start Trial
Patent: 2018004
Estimated Expiration: ⤷ Start Trial
Patent: 2018016
Estimated Expiration: ⤷ Start Trial
Patent: 57286
Estimated Expiration: ⤷ Start Trial
Patent: 31094
Estimated Expiration: ⤷ Start Trial
Luxembourg
Patent: 0094
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 4398
Estimated Expiration: ⤷ Start Trial
Patent: 9664
Estimated Expiration: ⤷ Start Trial
Patent: 11001893
Estimated Expiration: ⤷ Start Trial
Netherlands
Patent: 0962
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 18004
Estimated Expiration: ⤷ Start Trial
Patent: 18041
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 65988
Estimated Expiration: ⤷ Start Trial
Patent: 57286
Estimated Expiration: ⤷ Start Trial
Patent: 31094
Estimated Expiration: ⤷ Start Trial
Patent: 01408
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 65988
Estimated Expiration: ⤷ Start Trial
Patent: 57286
Estimated Expiration: ⤷ Start Trial
Patent: 31094
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01500207
Estimated Expiration: ⤷ Start Trial
Patent: 01800660
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 65988
Estimated Expiration: ⤷ Start Trial
Patent: 57286
Estimated Expiration: ⤷ Start Trial
Patent: 31094
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1825113
Estimated Expiration: ⤷ Start Trial
Patent: 1928973
Estimated Expiration: ⤷ Start Trial
Patent: 2150184
Estimated Expiration: ⤷ Start Trial
Patent: 2178208
Estimated Expiration: ⤷ Start Trial
Patent: 2300471
Estimated Expiration: ⤷ Start Trial
Patent: 110063647
Estimated Expiration: ⤷ Start Trial
Patent: 180014845
Estimated Expiration: ⤷ Start Trial
Patent: 180133561
Estimated Expiration: ⤷ Start Trial
Patent: 200029614
Estimated Expiration: ⤷ Start Trial
Patent: 200128600
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 45887
Estimated Expiration: ⤷ Start Trial
Patent: 99494
Estimated Expiration: ⤷ Start Trial
Patent: 39021
Estimated Expiration: ⤷ Start Trial
Patent: 34535
Estimated Expiration: ⤷ Start Trial
United Kingdom
Patent: 75657
Estimated Expiration: ⤷ Start Trial
Patent: 95426
Estimated Expiration: ⤷ Start Trial
Patent: 95427
Estimated Expiration: ⤷ Start Trial
Patent: 95428
Estimated Expiration: ⤷ Start Trial
Patent: 1104561
Estimated Expiration: ⤷ Start Trial
Patent: 1222749
Estimated Expiration: ⤷ Start Trial
Patent: 1222750
Estimated Expiration: ⤷ Start Trial
Patent: 1222752
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering VELTASSA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| European Patent Office | 2916851 | AGENTS DE LIAISON AU POTASSIUM POUR TRAITER L'HYPERTENSION ET L'HYPERKALIÉMIE (POTASSIUM-BINDING AGENTS FOR TREATING HYPERTENSION AND HYPERKALEMIA) | ⤷ Start Trial |
| Brazil | 112015007749 | métodos para tratamento de hipertensão , de hipercaliemia , e de doença renal crônica. | ⤷ Start Trial |
| Australia | 2005231424 | Ion binding polymers and uses thereof | ⤷ Start Trial |
| Netherlands | 300962 | ⤷ Start Trial | |
| China | 1980645 | Method and composition for treating ionic equilibrium disorder | ⤷ Start Trial |
| Spain | 2720612 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VELTASSA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1732523 | 2018C/004 | Belgium | ⤷ Start Trial | PRODUCT NAME: PATIROMER OF EENDER WELK ZOUT OF AFGELEIDE ERVAN; AUTHORISATION NUMBER AND DATE: EU/1/17/1179 20170721 |
| 1732523 | C01732523/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: PATIROMER; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 66411 22.12.2017 |
| 2957286 | 122018000145 | Germany | ⤷ Start Trial | PRODUCT NAME: PATIROMER SORBITEX CALCIUM; REGISTRATION NO/DATE: EU/1/17/1179 20170719 |
| 2957286 | SPC/GB19/003 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: PATIROMER SORBITEX CALCIUM; REGISTERED: UK EU/1/17/1179/001(NI) 20170721; UK EU/1/17/1179/002(NI) 20170721; UK EU/1/17/1179/003(NI) 20170721; UK EU/1/17/1179/004(NI) 20170721; UK EU/1/17/1179/005(NI) 20170721; UK EU/1/17/1179/006(NI) 20170721; UK EU/1/17/1179/007(NI) 20170721; UK EU/1/17/1179/008(NI) 20170721; UK EU/1/17/1179/009(NI) 20170721; UK PLGB 50784/0002-0001 20170721; UK PLGB 50784/0003-0001 20170721; UK PLGB 50784/0004-0001 20170721 |
| 2957286 | CR 2018 00043 | Denmark | ⤷ Start Trial | PRODUCT NAME: PATIROMER SORBITEX CALCIUM; REG. NO/DATE: EU/1/17/1179/001-009 20170721 |
| 2957286 | 2018/046 | Ireland | ⤷ Start Trial | PRODUCT NAME: PATIROMER SORBITEX CALCIUM; REGISTRATION NO/DATE: EU/1/17/1179 20170719 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
VELTASSA: Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
