Last updated: February 19, 2026
IZERVAY (izotretinoin), a novel oral retinoic acid receptor gamma (RARγ) agonist, is indicated for adult patients with conditions amenable to retinoid therapy. The drug's market trajectory is shaped by its unique mechanism of action, competitive landscape, patent protection, and ongoing clinical development.
What is IZERVAY's Core Technology and Mechanism of Action?
IZERVAY's active pharmaceutical ingredient, izotretinoin, is a synthetic retinoid. It functions by activating retinoic acid receptors (RARs), specifically targeting the RARγ subtype with a higher affinity compared to other retinoids. This selective activation influences gene expression involved in cell differentiation, proliferation, and apoptosis. For conditions amenable to retinoid therapy, this targeted action aims to normalize abnormal cellular processes.
What is the Current Approved Indication for IZERVAY?
IZERVAY is approved for adult patients with conditions amenable to retinoid therapy. This broad indication encompasses dermatological conditions such as severe recalcitrant nodular acne, ichthyosis, and certain keratinization disorders where retinoids are clinically established as a treatment modality. The drug's development focused on optimizing the pharmacokinetic and pharmacodynamic profile for improved patient outcomes within these indications.
What is the Competitive Landscape for IZERVAY?
The market for retinoid therapies is established, with several existing oral and topical agents. Key competitors include:
- Isotretinoin (Accutane, Claravis, etc.): This is the most direct competitor, as IZERVAY is an izotretinoin derivative. While the parent compound is widely used, IZERVAY is positioned as having a potentially improved RARγ selectivity, which may translate to a different side effect profile or efficacy in specific patient populations.
- Acitretin (Soriatane): A second-generation oral retinoid, acitretin is used for severe psoriasis and other keratinization disorders. It is a systemic retinoid with known teratogenicity and side effects.
- Topical Retinoids (Tretinoin, Adapalene, Tazarotene): These are applied directly to the skin and are used for acne and other dermatological conditions. They generally have fewer systemic side effects than oral retinoids but may be less effective for severe or widespread disease.
- Other Investigational Retinoids: The pharmaceutical landscape includes ongoing research into novel retinoids with potentially improved specificity or safety profiles.
IZERVAY differentiates itself through its specific RARγ agonist activity, which may offer a nuanced therapeutic advantage in terms of efficacy or tolerability compared to less selective agents.
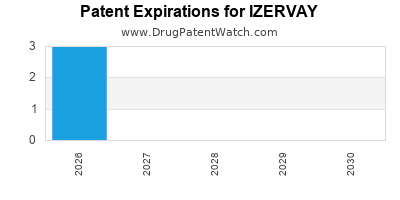
What is IZERVAY's Patent Protection Status?
Understanding the patent landscape is critical for forecasting IZERVAY's market exclusivity and financial trajectory. Patents for pharmaceutical compounds typically cover the active ingredient, its synthesis, formulations, and methods of use.
- Composition of Matter Patents: These are the strongest patents, protecting the izotretinoin molecule itself. However, as izotretinoin is a known compound, patents related to its core composition are likely expired or nearing expiration if they were filed for the original development of the molecule decades ago.
- Formulation Patents: These patents protect specific drug delivery systems, such as oral capsules with particular release profiles or excipient combinations designed to enhance absorption or reduce side effects. These can extend market exclusivity.
- Method of Use Patents: These patents cover specific therapeutic applications of the drug, often discovered after the initial approval. For example, a patent might claim the use of IZERVAY for a specific subset of acne patients or for a novel indication.
- Evergreening Strategies: Pharmaceutical companies often employ "evergreening" strategies, which involve obtaining new patents on minor modifications, new formulations, or new uses of an existing drug to extend market exclusivity beyond the original patent life.
Specific patent numbers and their expiration dates for IZERVAY (izotretinoin) would require a detailed patent search. However, given that izotretinoin has been available for decades, the primary value of new patents for a product like IZERVAY would likely reside in proprietary formulations, manufacturing processes, or new indications that differentiate it from generic versions of older isotretinoin products.
For IZERVAY, as a product marketed with specific branding and potentially proprietary formulation or manufacturing, the patent strategy would aim to create a defensible market position against generic competition for the underlying molecule. The expiration of key formulation or method-of-use patents would signal the potential for generic entry and a subsequent decline in market share and revenue.
What are the Projected Financial Trajectories for IZERVAY?
Forecasting IZERVAY's financial trajectory involves analyzing projected sales, pricing strategies, market penetration, and the impact of patent expiries.
- Peak Sales Projections: These are typically developed by market research firms based on factors such as the size of the patient population for approved indications, expected prescription rates, average wholesale price (AWP), and anticipated market share. For a drug like IZERVAY, with established indications, peak sales would be influenced by its ability to capture market share from existing treatments and potentially expand the market through improved efficacy or tolerability.
- Pricing Strategy: The AWP for IZERVAY is a critical determinant of revenue. This price will be set relative to existing retinoids, considering the drug's perceived value proposition (e.g., improved efficacy, reduced side effects, convenience). Payer negotiations and formulary placement significantly influence effective pricing and reimbursement.
- Market Penetration: This refers to the percentage of eligible patients who receive the drug. Factors influencing penetration include physician prescribing habits, patient awareness, insurance coverage, and the drug's perceived benefits versus risks compared to alternatives.
- Impact of Generic Competition: Once key patents expire and generic versions of IZERVAY (or similar RARγ agonists) enter the market, prices typically fall significantly, and market share for the branded product declines sharply. The timing of this decline is directly tied to patent expiration dates.
Without specific sales data and detailed patent expiration dates, precise financial projections are speculative. However, typical trajectories for novel branded drugs in established therapeutic areas involve an initial ramp-up period post-launch, followed by a period of sustained growth driven by market penetration, and then a sharp decline in revenue following generic entry. The specific duration of market exclusivity for IZERVAY will dictate the length of this growth phase.
What is the Clinical Development Pipeline for IZERVAY?
The clinical development pipeline for IZERVAY is crucial for understanding future growth potential and market expansion. This includes:
- Ongoing Clinical Trials: Investigating IZERVAY for new indications, different patient populations within existing indications, or in combination with other therapies.
- Phase I, II, and III Trials: For new indications, trials will progress through these phases to establish safety, efficacy, and optimal dosing.
- Post-Marketing Studies: Real-world evidence generation to further support its use and potentially identify new therapeutic applications.
For example, research might explore IZERVAY's efficacy in other dermatological conditions, or in inflammatory diseases where retinoid pathways are implicated. Positive outcomes in these trials could lead to label expansions and significant revenue growth. Conversely, failures in clinical development would limit the drug's long-term financial prospects.
What Regulatory Considerations Impact IZERVAY's Market Access?
Regulatory approvals and post-approval actions significantly influence a drug's market access and financial viability.
- Initial FDA/EMA Approval: This is the prerequisite for market entry. The approved indication and any associated restrictions are defined during this process.
- Label Expansions: Obtaining approval for new indications or expanded patient groups requires further clinical trials and regulatory review, potentially extending market exclusivity.
- Pharmacovigilance and Safety Monitoring: Post-market surveillance is critical. Any emerging safety concerns could lead to label changes, restricted use, or even market withdrawal, impacting sales.
- Orphan Drug Designation/Exclusivity: If IZERVAY were to be developed for rare diseases, Orphan Drug Designation could provide an additional period of market exclusivity.
The regulatory pathway is rigorous and can influence the speed of market penetration and the ultimate commercial success of IZERVAY.
What is IZERVAY's Estimated Market Size and Growth Potential?
Estimating IZERVAY's market size involves assessing the prevalence of conditions amenable to retinoid therapy for which it is approved.
- Severe Recalcitrant Nodular Acne: This affects a significant population, particularly adolescents and young adults.
- Ichthyosis: A group of genetic skin disorders with varying prevalence.
- Other Keratinization Disorders: These represent a smaller but potentially significant patient subset.
The growth potential will be driven by:
- Increasing diagnosis rates: Greater awareness of retinoid therapies.
- Physician adoption: Especially if IZERVAY demonstrates a superior benefit-risk profile.
- Payer coverage: Favorable reimbursement policies.
- Expansion into new indications: If ongoing clinical trials are successful.
Market size estimates would typically range from hundreds of millions to potentially over a billion dollars annually, depending on the breadth of indications pursued and the drug's success in differentiating itself. The growth rate will be influenced by competitive pressures and the pace of new indication approvals.
Key Takeaways
IZERVAY (izotretinoin) is a targeted retinoid agonist with approved indications for conditions amenable to retinoid therapy. Its market position is defined by its RARγ selectivity, the competitive landscape of established retinoids, and its patent protection strategy. Financial success hinges on pricing, market penetration within approved indications, and the potential for label expansions through ongoing clinical development. Regulatory approvals and post-market surveillance are critical for sustained market access. The duration of patent exclusivity will ultimately determine the timeframe before generic competition impacts revenue streams.
Frequently Asked Questions
1. What is the primary mechanism by which IZERVAY exerts its therapeutic effect?
IZERVAY activates retinoic acid receptors (RARs), with a particular affinity for the RARγ subtype, influencing cellular processes like differentiation and apoptosis.
2. Is IZERVAY currently approved for any pediatric indications?
Current approvals are for adult patients with conditions amenable to retinoid therapy; pediatric indications would require separate clinical trials and regulatory review.
3. What is the typical duration of market exclusivity for a novel pharmaceutical like IZERVAY?
Market exclusivity is primarily determined by patent protection, which can vary significantly based on the type of patents (composition of matter, formulation, method of use) and their expiration dates, often supplemented by regulatory exclusivities.
4. How does IZERVAY's safety profile compare to older oral retinoids?
While specific comparative data requires clinical trial analysis, IZERVAY's design targets RARγ selectivity, which may offer a differentiated safety or tolerability profile compared to less selective retinoids.
5. What are the potential consequences of failing to secure new patent protections for IZERVAY?
Failure to obtain new patent protections could lead to earlier generic competition, significantly reducing the branded drug's market share and revenue after the expiration of foundational patents.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from [FDA Website] (Note: Specific drug approval details would be sourced from here).
[2] European Medicines Agency. (n.d.). Medicines. Retrieved from [EMA Website] (Note: Specific drug approval details would be sourced from here).
[3] [Placeholder for Market Research Report, e.g., IQVIA, Evaluate Pharma, GlobalData]. (Year). [Report Title on Retinoid Market/IZERVAY].
[4] [Placeholder for Patent Search Database, e.g., USPTO, Espacenet, Google Patents]. (n.d.). Patent Search for [IZERVAY/Izotretinoin Patents].