VYNDAMAX Drug Patent Profile
✉ Email this page to a colleague
When do Vyndamax patents expire, and when can generic versions of Vyndamax launch?
Vyndamax is a drug marketed by Foldrx Pharms and is included in one NDA. There are three patents protecting this drug and one Paragraph IV challenge.
This drug has sixty-six patent family members in twenty-seven countries.
The generic ingredient in VYNDAMAX is tafamidis. One supplier is listed for this compound. Additional details are available on the tafamidis profile page.
DrugPatentWatch® Generic Entry Outlook for Vyndamax
Vyndamax was eligible for patent challenges on May 3, 2023.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be August 31, 2035. This may change due to patent challenges or generic licensing.
There have been four patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for VYNDAMAX?
- What are the global sales for VYNDAMAX?
- What is Average Wholesale Price for VYNDAMAX?
Summary for VYNDAMAX
| International Patents: | 66 |
| US Patents: | 3 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 70 |
| Patent Applications: | 312 |
| Drug Prices: | Drug price information for VYNDAMAX |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for VYNDAMAX |
| DailyMed Link: | VYNDAMAX at DailyMed |
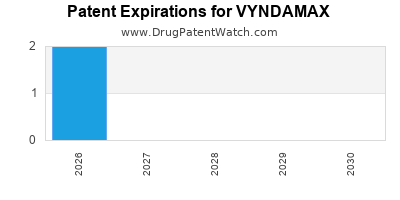

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for VYNDAMAX
Generic Entry Date for VYNDAMAX*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
CAPSULE;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
US Patents and Regulatory Information for VYNDAMAX
VYNDAMAX is protected by three US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of VYNDAMAX is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Foldrx Pharms | VYNDAMAX | tafamidis | CAPSULE;ORAL | 212161-001 | May 3, 2019 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Foldrx Pharms | VYNDAMAX | tafamidis | CAPSULE;ORAL | 212161-001 | May 3, 2019 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Foldrx Pharms | VYNDAMAX | tafamidis | CAPSULE;ORAL | 212161-001 | May 3, 2019 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Foldrx Pharms | VYNDAMAX | tafamidis | CAPSULE;ORAL | 212161-001 | May 3, 2019 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for VYNDAMAX
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Pfizer Europe MA EEIG | Vyndaqel | tafamidis | EMEA/H/C/002294Vyndaqel is indicated for the treatment of transthyretin amyloidosis in adult patients with stage-1 symptomatic polyneuropathy to delay peripheral neurologic impairment. | Authorised | no | no | yes | 2011-11-16 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for VYNDAMAX
When does loss-of-exclusivity occur for VYNDAMAX?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 1778
Patent: FORMAS SÓLIDAS CRISTALINAS DE 6-CARBOXI-2-(3,5-DICLOROFENIL)-BENZOXAZOL
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 15313875
Patent: Crystalline solid forms of 6-carboxy-2-(3,5-dichlorophenyl)-benzoxazole
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2017003421
Patent: formas sólidas cristalinas de 6-carbóxi-2-(3,5-diclorofenil)-benzoxazol
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 03194
Patent: FORMES CRISTALLINES SOLIDES DE 6-CARBOXY-2-(3,5-DICHLOROPHENYL)-BENZOXAZOLE (CRYSTALLINE SOLID FORMS OF 6-CARBOXY-2-(3,5-DICHLOROPHENYL)-BENZOXAZOLE)
Estimated Expiration: ⤷ Start Trial
China
Patent: 6715405
Patent: 6‑羧基‑2‑(3,5‑二氯苯基)苯并噁唑的结晶固体形式 (Crystalline solid forms of 6-carboxy-2-(3,5-dichlorophenyl)-benzoxazole)
Estimated Expiration: ⤷ Start Trial
Patent: 5368313
Patent: 6-羧基-2-(3,5-二氯苯基)苯并噁唑的结晶固体形式 (Crystalline solid forms of 6-carboxy-2-(3, 5-dichlorophenyl) benzoxazole)
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0211834
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 24843
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 91461
Patent: FORMES CRISTALLINES SOLIDES DU 6-CARBOXY-2-(3,5-DICHLOROPHÉNYL)-BENZOXAZOLE (CRYSTALLINE SOLID FORMS OF 6-CARBOXY-2-(3,5-DICHLOROPHENYL)-BENZOXAZOLE)
Estimated Expiration: ⤷ Start Trial
Patent: 77993
Patent: FORMES CRISTALLINES SOLIDES DU 6-CARBOXY-2-(3,5-DICHLOROPHÉNYL)-BENZOXAZOLE POUR L'UTILISATION COMME MÉDICAMENTS (CRYSTALLINE SOLID FORMS OF 6-CARBOXY-2-(3,5-DICHLOROPHENYL)-BENZOXAZOLE FOR USE AS A MEDICAMENT)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 56583
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 57525
Estimated Expiration: ⤷ Start Trial
Patent: 16065042
Patent: 6−カルボキシ−2−(3,5−ジクロロフェニル)−ベンゾオキサゾールの結晶性固体形態 (CRYSTALLINE SOLID FORMS OF 6-CARBOXY-2-(3,5-DICHLOROPHENYL)-BENZOXAZOLE)
Estimated Expiration: ⤷ Start Trial
Patent: 20055832
Patent: 6−カルボキシ−2−(3,5−ジクロロフェニル)−ベンゾオキサゾールの結晶性固体形態 (CRYSTALLINE SOLID FORMS OF 6-CARBOXY-2-(3,5-DICHLOROPHENYL)-BENZOXAZOLE)
Estimated Expiration: ⤷ Start Trial
Patent: 22024014
Patent: 6-カルボキシ-2-(3,5-ジクロロフェニル)-ベンゾオキサゾールの結晶性固体形態
Estimated Expiration: ⤷ Start Trial
Patent: 23134645
Patent: 6-カルボキシ-2-(3,5-ジクロロフェニル)-ベンゾオキサゾールの結晶性固体形態 (CRYSTALLINE SOLID FORMS OF 6-CARBOXY-2-(3,5-DICHLOROPHENYL)-BENZOXAZOLE)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 91461
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 17002954
Patent: FORMAS SOLIDAS CRISTIALINAS DE 6-CARBOXI-2-(3,5-DICLOROFENIL)-BENZ OXAZOL. (CRYSTALLINE SOLID FORMS OF 6-CARBOXY-2-(3,5-DICHLOROPHENYL)-BENZO XAZOLE.)
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 91461
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 91461
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 92252
Patent: КРИСТАЛЛИЧЕСКИЕ ТВЕРДЫЕ ФОРМЫ 6-КАРБОКСИ-2-(3,5-ДИХЛОРФЕНИЛ)БЕНЗОКСАЗОЛА (CRYSTALLINE SOLID FORMS OF 6-CARBOXY-2-(3,5-DICHLOROPHENYL)BENZOXAZOLE)
Estimated Expiration: ⤷ Start Trial
Patent: 17107235
Patent: КРИСТАЛЛИЧЕСКИЕ ТВЕРДЫЕ ФОРМЫ 6-КАРБОКСИ-2-(3,5-ДИХЛОРФЕНИЛ)БЕНЗОКСАЗОЛА
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 634
Patent: ČVRSTI KRISTALNI OBLICI 6-KARBOKSI-2-(3,5-DIHLOROFENIL)-BENZOKSAZOLA (CRYSTALLINE SOLID FORMS OF 6-CARBOXY-2-(3,5-DICHLOROPHENYL)-BENZOXAZOLE)
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201700958Y
Patent: CRYSTALLINE SOLID FORMS OF 6-CARBOXY-2-(3,5-DICHLOROPHENYL)-BENZOXAZOLE
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 91461
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 2004059
Estimated Expiration: ⤷ Start Trial
Patent: 170040314
Patent: 6-카르복시-2--벤족사졸의 결정질 고체 형태 (6--2-35-- CRYSTALLINE SOLID FORMS OF 6-CARBOXY-2-35-DICHLOROPHENYL-BENZOXAZOLE)
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 01785
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 44902
Estimated Expiration: ⤷ Start Trial
Patent: 1613880
Patent: Crystalline solid forms of 6-carboxy-2-(3,5-dichlorophenyl)-benzoxazole
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering VYNDAMAX around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Luxembourg | 91935 | ⤷ Start Trial | |
| Cyprus | 1108467 | ⤷ Start Trial | |
| Portugal | 1988397 | ⤷ Start Trial | |
| Brazil | 112017003421 | formas sólidas cristalinas de 6-carbóxi-2-(3,5-diclorofenil)-benzoxazol | ⤷ Start Trial |
| China | 100448852 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VYNDAMAX
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1587821 | 2012/007 | Ireland | ⤷ Start Trial | PRODUCT NAME: VYNDAQEL- TAFAMIDIS MEGLUMINE; REGISTRATION NO/DATE: EU/1/11/717/001 20111116 |
| 1587821 | C300516 | Netherlands | ⤷ Start Trial | PRODUCT NAME: TAFAMIDIS, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, IN HET BIJZONDER TAFAMIDIS MEGLUMINE ZOUT; REGISTRATION NO/DATE: EU/1/11/717/001 20111116 |
| 1587821 | C 2012 004 | Romania | ⤷ Start Trial | PRODUCT NAME: TAFAMIDISMEGLUMINA; NATIONAL AUTHORISATION NUMBER: EU/1/11/717/001; DATE OF NATIONAL AUTHORISATION: 20111116; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/11/717/001; DATE OF FIRST AUTHORISATION IN EEA: 20111116 |
| 1587821 | 382 | Finland | ⤷ Start Trial | |
| 1587821 | CR 2012 00006 | Denmark | ⤷ Start Trial | PRODUCT NAME: TAFAMIDIS (SOM MEGLUMIN); REG. NO/DATE: EU/1/11/717/001 20111116 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for VYNDAMAX
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
