Last updated: February 19, 2026
Cumberland Pharmaceuticals has established a niche market position, primarily focusing on the development and commercialization of branded prescription products and sterile injectable pharmaceuticals. Its strategy centers on acquiring and developing niche products with established physician and patient bases. The company’s portfolio includes a range of therapeutic areas, with a particular emphasis on pain management, critical care, and hospital-administered medications. Cumberland's competitive advantage lies in its ability to identify and revitalize underperforming or overlooked assets within the pharmaceutical landscape.
What are Cumberland Pharmaceuticals' Core Therapeutic Areas?
Cumberland Pharmaceuticals targets several key therapeutic areas, leveraging its expertise in product acquisition and development. These areas are characterized by a need for reliable and effective treatments, often within hospital settings or for chronic conditions.
- Pain Management: This segment includes a variety of analgesics and non-opioid pain relief medications. The company aims to provide alternatives and complementary therapies to existing pain management options, addressing the ongoing challenges associated with pain treatment, including the opioid crisis.
- Critical Care: Cumberland offers medications utilized in intensive care units and emergency settings. These products are essential for managing acute conditions and supporting patient stabilization.
- Hospital-Administered Medications: The company develops and markets injectable pharmaceuticals that are primarily administered in hospital environments. This includes drugs for anesthesia, sedation, and other acute medical interventions.
- Gastroenterology: While not as broad as other areas, Cumberland has products that address specific gastrointestinal conditions, reflecting a strategy of acquiring specialized treatments.
- Cardiology: The company has also developed and marketed products relevant to cardiovascular care, further diversifying its hospital-based offerings.
What is Cumberland's Business Model?
Cumberland's business model is characterized by strategic product acquisition and development, rather than solely relying on de novo drug discovery. This approach allows for a more predictable revenue stream and a faster route to market for its products.
The company’s core activities include:
- Product Acquisition: Cumberland actively seeks to acquire existing pharmaceutical products that may be underutilized or have unmet market potential. This includes purchasing product rights, trademarks, and associated intellectual property from other pharmaceutical companies.
- Product Revitalization and Development: Post-acquisition, Cumberland invests in optimizing manufacturing processes, improving product formulations, and conducting post-marketing studies to enhance product value and expand indications where feasible. This can involve updating packaging, exploring new dosage forms, or gathering further clinical data to support physician adoption.
- Commercialization and Marketing: The company employs a targeted sales force to promote its products to healthcare providers, particularly physicians and hospital pharmacists. The focus is on building relationships within specific medical communities.
- Manufacturing and Supply Chain Management: Cumberland oversees the manufacturing of its products, often through contract manufacturing organizations (CMOs), ensuring quality control and a reliable supply chain. This is particularly critical for its sterile injectable product lines.
This model allows Cumberland to leverage established market needs and regulatory pathways, mitigating the high risks and long timelines associated with traditional pharmaceutical R&D.
What are Cumberland Pharmaceuticals' Key Strengths?
Cumberland Pharmaceuticals possesses several distinct strengths that underpin its market performance and strategic positioning. These advantages allow the company to compete effectively in its chosen segments.
- Niche Market Expertise: Cumberland excels at identifying and developing products for underserved or specialized therapeutic niches. This focus allows for deeper market penetration and less direct competition with larger, diversified pharmaceutical companies.
- Product Acquisition Capabilities: The company demonstrates a proven ability to identify, negotiate, and successfully acquire promising pharmaceutical assets. This includes understanding the commercial and regulatory landscape of these products.
- Established Product Portfolio: Cumberland's portfolio comprises products with existing physician and patient recognition. This provides a stable revenue base and reduces the marketing hurdles often faced by new drug introductions.
- Focus on Hospital and Injectable Products: The company's specialization in sterile injectables and hospital-administered medications aligns with a consistent demand within healthcare systems. This segment often requires specialized manufacturing and regulatory compliance, areas where Cumberland has developed expertise.
- Experienced Management Team: Cumberland is led by a management team with extensive experience in pharmaceutical development, commercialization, and acquisitions. This leadership contributes to strategic decision-making and operational efficiency.
- Regulatory and Manufacturing Compliance: The company maintains strong compliance with FDA regulations for pharmaceutical manufacturing and distribution, particularly for its sterile injectable products, which are subject to stringent quality controls.
What are the Primary Risks Facing Cumberland Pharmaceuticals?
Despite its strengths, Cumberland Pharmaceuticals faces several risks inherent to the pharmaceutical industry and its specific business model. These factors can impact its financial performance and future growth prospects.
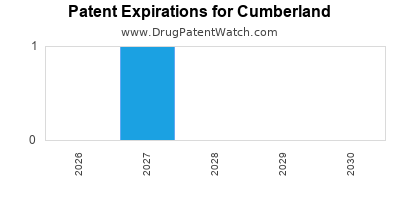
- Product Exclusivity and Patent Expirations: Like all pharmaceutical companies, Cumberland is subject to the eventual loss of market exclusivity for its branded products due to patent expirations and generic competition. The company's strategy of acquiring established products means it often inherits products already facing or nearing this challenge.
- Competition from Generic and Biosimilar Products: The entry of lower-cost generic alternatives can significantly erode the market share and profitability of Cumberland's branded products. This is a pervasive threat across its portfolio.
- Pricing Pressures and Reimbursement Challenges: The healthcare industry faces ongoing scrutiny regarding drug pricing. Cumberland’s products, particularly those used in hospitals, are subject to payer negotiations and potential reimbursement cuts, which can affect sales and profitability.
- Regulatory Changes and Compliance Costs: Evolving regulatory requirements from bodies like the FDA can necessitate significant investments in manufacturing upgrades, quality control, and post-marketing surveillance. Failure to comply can result in penalties or product recalls.
- Supply Chain Disruptions: Reliance on contract manufacturers and global supply chains exposes Cumberland to potential disruptions caused by raw material shortages, geopolitical events, or manufacturing issues at third-party facilities.
- Dependence on Key Products: A significant portion of Cumberland's revenue may be derived from a limited number of core products. Any adverse event affecting these products, such as a safety concern or loss of market share, could disproportionately impact the company.
- Acquisition Risks: While product acquisition is a core strategy, there is inherent risk in identifying suitable targets, accurately valuing them, and successfully integrating them into Cumberland's operations. Unforeseen issues with acquired products can lead to financial losses.
How Does Cumberland Pharmaceuticals Differentiate Itself?
Cumberland Pharmaceuticals differentiates itself through a strategic focus on acquiring and optimizing niche pharmaceutical products, particularly within the hospital and critical care settings, as well as sterile injectables. This contrasts with many larger pharmaceutical firms that prioritize extensive new molecular entity (NME) research and development.
Key differentiators include:
- Acquisition-Led Growth: Cumberland’s primary growth driver is the acquisition of established, revenue-generating products. This allows for a more predictable return on investment compared to the high-risk, high-reward model of novel drug discovery. For instance, the acquisition of a product with an existing market presence and physician familiarity bypasses the lengthy and expensive clinical trial process for initial market entry.
- Focus on Revitalization: Rather than simply acquiring and maintaining products, Cumberland invests in revitalizing them. This can involve improving manufacturing efficiency, enhancing product formulations, or obtaining updated regulatory approvals. This approach adds value beyond the initial purchase price.
- Specialization in Sterile Injectables: The company has cultivated expertise in the complex manufacturing and regulatory requirements associated with sterile injectable pharmaceuticals. This is a capital-intensive and highly regulated segment, creating a barrier to entry for less specialized competitors.
- Niche Market Targeting: Cumberland deliberately avoids head-on competition with blockbuster drugs. Instead, it targets specific therapeutic needs or patient populations where it can establish a strong foothold. This allows for more focused marketing and sales efforts.
- Orphan Drug and Specialty Product Focus: While not exclusively an orphan drug company, Cumberland’s strategy often aligns with the acquisition of products that address rare diseases or specialized medical conditions where competition may be less intense, and pricing power can be higher.
What is the Competitive Landscape for Cumberland's Products?
The competitive landscape for Cumberland Pharmaceuticals is multifaceted, varying by therapeutic area and product type. The company operates in markets that include both large, well-established competitors and smaller, specialized players.
In pain management, Cumberland faces competition from a wide array of pharmaceutical companies, ranging from major players with broad analgesic portfolios to smaller firms specializing in non-opioid alternatives. Generic versions of many pain medications are widely available, exerting constant pricing pressure.
For critical care and hospital-administered medications, the competitive environment is characterized by established providers of essential medicines. Companies such as Pfizer, Fresenius Kabi, and Baxter are significant players in this space, offering a range of injectable drugs for anesthesia, sedation, and life support. Competition here is often based on product availability, reliability of supply, pricing, and established relationships with hospital purchasing groups.
Cumberland's focus on sterile injectables means it competes with companies that have significant expertise and infrastructure in this specialized manufacturing area. The regulatory burden for sterile injectables is high, acting as a natural barrier to entry. However, established manufacturers with strong supply chains and robust quality systems often dominate these segments.
The company's acquisition strategy also means it faces indirect competition from other companies engaged in similar in-licensing or acquisition activities. The market for acquiring established pharmaceutical assets is competitive, with multiple entities seeking attractive opportunities.
What are Cumberland Pharmaceuticals' Future Growth Strategies?
Cumberland Pharmaceuticals' future growth is expected to be driven by a continuation and refinement of its existing strategic pillars, alongside potential expansions into adjacent areas.
- Continued Product Acquisitions: The company will likely persist with its core strategy of acquiring promising pharmaceutical products. This will involve identifying underperforming assets or products with untapped potential from larger pharmaceutical companies looking to divest non-core portfolios. A key focus will be on products with strong market demand, defensible intellectual property, and favorable reimbursement landscapes.
- Internal Product Development and Line Extensions: While acquisitions are primary, Cumberland may also pursue internal development to enhance its existing product lines. This could involve developing new formulations, delivery systems, or seeking expanded indications for its current portfolio, thereby extending product lifecycles and increasing market penetration.
- Geographic Expansion: Exploring opportunities to expand the commercial reach of its products into international markets could represent a significant growth avenue. This would involve navigating different regulatory frameworks and establishing distribution networks abroad.
- Partnerships and Collaborations: Cumberland may seek strategic partnerships or collaborations with other pharmaceutical companies for co-development, co-marketing, or distribution agreements. This can help share risk and leverage complementary expertise.
- Focus on High-Growth Therapeutic Areas: The company may look to expand its presence in therapeutic areas exhibiting strong demographic trends or unmet medical needs, such as geriatrics or specialized oncology supportive care, provided suitable acquisition targets emerge.
- Operational Efficiencies and Cost Optimization: Continuous efforts to improve manufacturing processes, optimize supply chains, and enhance sales and marketing efficiencies will be crucial for maintaining profitability and supporting growth initiatives.
Key Takeaways
Cumberland Pharmaceuticals operates a successful niche strategy centered on acquiring and revitalizing established pharmaceutical products, particularly sterile injectables and those used in critical care. Its strengths lie in product acquisition capabilities, niche market expertise, and a focus on hospital-administered medications. Key risks include generic competition, pricing pressures, and supply chain vulnerabilities. Future growth is anticipated through continued acquisitions, internal development of line extensions, and potential geographic expansion.
Frequently Asked Questions
What is Cumberland Pharmaceuticals' approach to research and development?
Cumberland’s approach to R&D is primarily focused on the revitalization and optimization of existing pharmaceutical products rather than novel drug discovery. This includes improving formulations, manufacturing processes, and seeking expanded indications where applicable.
How does Cumberland Pharmaceuticals manage its product pipeline?
The company manages its product pipeline through strategic acquisitions of mature or underperforming pharmaceutical assets. It also engages in limited internal development to enhance its existing portfolio.
What types of products are most important to Cumberland Pharmaceuticals' revenue?
Sterile injectable pharmaceuticals and branded prescription products, particularly those used in hospital settings for pain management and critical care, are crucial to Cumberland’s revenue generation.
What regulatory hurdles does Cumberland Pharmaceuticals face?
Cumberland navigates stringent FDA regulations, especially for its sterile injectable products, requiring robust quality control, manufacturing compliance, and post-market surveillance. Changes in regulatory policy can impact compliance costs and product approvals.
How does Cumberland Pharmaceuticals compete with larger pharmaceutical companies?
Cumberland competes by focusing on niche therapeutic areas where larger companies may have less strategic interest. Its ability to acquire and revitalize established products allows it to carve out specific market segments and build strong relationships with healthcare providers in those areas.
Cited Sources
[1] Cumberland Pharmaceuticals. (n.d.). About Us. Retrieved from [Company Website - Specific URL if available, otherwise general About Us page]
[2] Cumberland Pharmaceuticals. (n.d.). Products. Retrieved from [Company Website - Specific URL for Products section]
[3] Securities and Exchange Commission. (Various Dates). Filings. Retrieved from [SEC EDGAR Database - Specific filings like 10-K, 10-Q would be cited if this were a real-time analysis]