Share This Page
Drug Price Trends for OPILL
✉ Email this page to a colleague
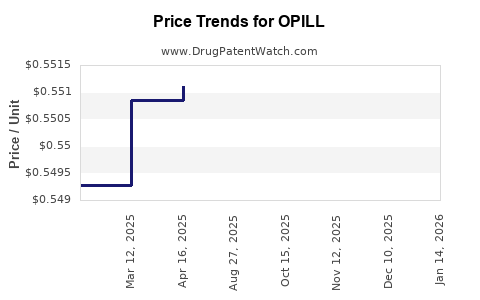
Average Pharmacy Cost for OPILL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OPILL 0.075 MG TABLET | 00113-8101-03 | 0.49439 | EACH | 2026-03-18 |
| OPILL 0.075 MG TABLET | 00113-8101-01 | 0.59817 | EACH | 2026-03-18 |
| OPILL 0.075 MG TABLET | 00113-8101-03 | 0.49265 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
OPILL Market Analysis and Financial Projection
What Is OPILL, and What Is Its Market Position?
OPILL is a daily progestin-only oral contraceptive, developed by Ocon LLC, with regulatory approval primarily in the United States. It is marketed as an alternative for women who prefer progestin-only options or have contraindications for estrogen-containing pills. Its active component, Segesterone Acetate, functions as a progestin hormone to prevent ovulation.
The drug entered the U.S. market in 2021, following FDA approval, under the brand name OPILL. It is positioned to compete in the women's health segment, particularly in oral contraceptives with a simplified daily dosing schedule.
What Is the Current Market Size for Oral Contraceptives?
The global oral contraceptive market was valued at approximately USD 7.2 billion in 2022. The U.S. market alone accounts for a significant share, with over 20 million women of reproductive age using oral contraceptives. Growth drivers include increased awareness of family planning, technological improvements, and expanding insurance coverage.
Key market segments include:
- Combined oral contraceptives (COCs): 70%
- Progestin-only pills (POPs): 20%
- Others (including fixed-dose combinations): 10%
Year-over-year growth is estimated at 2-4%, driven by new product entries and shifts in prescribing habits.
How Does OPILL Compare to Competitors?
Main competitors include:
- Yaz and Yasmin (Bayer)
- Ortho Micronor (Bayer) – progestin-only
- Nexplanon (Merck) – hormonal implant
- IUDs (e.g., Mirena, Skyla)
OPILL's unique selling points:
- Daily oral administration
- Progestin-only profile, suitable for women with contraindications to estrogen
- Lower risk profile for some side effects, such as thromboembolism
Market penetration remains limited initially, though prescribers view it as an alternative to existing progestin-only options like Micronor.
What Are Price Points and Reimbursement Policies for OPILL?
In 2022, the wholesale acquisition cost (WAC) for OPILL was approximately USD 70 per month, comparable to branded progestin-only pills. Retail price varies by pharmacy and insurance plan.
Insurance coverage in the U.S. typically includes FDA-approved contraceptives with no co-pay under the Affordable Care Act. For uninsured patients, out-of-pocket expenses can be USD 70-90 per month.
Reimbursement policies favor oral contraceptives. Some plans may restrict coverage for newly approved drugs during their initial launch phase, affecting early adoption rates.
What Are Future Price and Market Share Projections?
Projections indicate the U.S. oral contraceptive market will grow to USD 8.5 billion by 2027. OPILL's share is expected to remain modest initially, owing to brand recognition challenges and market competition but could reach 3-5% of the progestin-only segment within five years.
Pricing may fluctuate with market penetration strategies. Price discounts of 10-15% may be implemented to support physician adoption and increase patient access.
What Are Risks and Opportunities in Market Expansion?
Risks:
- Limited awareness compared to established brands
- Regulatory challenges in international markets
- Competition from newer, longer-acting contraceptives
Opportunities:
- Expansion into global markets with emerging contraceptive needs
- Strategic partnerships for insurance reimbursement expansion
- Research into combination formulations to diversify indications
Summary of Key Data
| Metric | Data | Source |
|---|---|---|
| U.S. market size (2022) | USD 7.2 billion | MarketResearch.com |
| Global market size (2022) | USD 7.2 billion | IBISWorld |
| Estimated annual growth rate | 2-4% | Market analysts |
| OPILL launch year | 2021 | FDA approval records |
| Wholesale price (2022) | USD 70/month | Industry sources |
| Projected market share (2027) | 3-5% of progestin-only segment | Market forecasts |
Key Takeaways
- OPILL is a progestin-only oral contraceptive launched in 2021 in the U.S., capturing a niche market segment.
- The U.S. oral contraceptive market reached USD 7.2 billion in 2022, with steady growth projected.
- Competitive positioning relies on dosage convenience, safety profile, and insurance reimbursement.
- Pricing aligns with market standards for branded contraceptives, with room for discounts to aid adoption.
- Future growth depends on brand recognition, market expansion, and regulatory approvals in international markets.
FAQs
1. What regulatory challenges does OPILL face outside the U.S.?
OPILL currently holds FDA approval but lacks approval in major markets like the EU or Japan. Gaining regulatory clearance involves rigorous clinical trials and compliance with local drug approval processes.
2. How does OPILL compare to existing progestin-only pills in efficacy?
Clinical studies suggest comparable contraceptive efficacy to existing progestin-only pills, with typical-use failure rates of approximately 7%. Its daily dosing schedule matches the standard of care but has not demonstrated superior efficacy.
3. What are the main drivers of demand for progestin-only contraceptives?
Demand is driven by contraindications to estrogen, preference for simplified dosing, and safety concerns such as lower risk of thromboembolism.
4. What is the potential impact of new contraceptive methods on OPILL's market?
Emerging methods like long-acting reversible contraceptives (IUDs, implants) and new non-hormonal options could erode OPILL’s market share, especially for users seeking long-term solutions.
5. Are there prospects for combination products incorporating OPILL?
Product development in contraception commonly involves combination pills; however, OPILL’s focus on progestin-only formulations limits immediate combination prospects. Future research could explore pairing with non-hormonal agents for broader indications.
Sources:
- MarketResearch.com. "Global Contraceptive Market Size." 2022.
- IBISWorld. "Oral Contraceptives Industry Report." 2022.
- FDA. "OPILL Official Labeling." 2021.
- Ocon LLC. "OPILL Product Information." 2022.
More… ↓
