Last updated: February 19, 2026
INJECTAFER (ferric carboxymaltose) is an intravenous iron replacement therapy. Its market performance is driven by demand for treating iron deficiency anemia (IDA), particularly in patients intolerant or unresponsive to oral iron. The drug's established efficacy and safety profile support its continued market penetration.
What is the current market size and projected growth for INJECTAFER?
The global market for intravenous iron replacement therapies, including INJECTAFER, is experiencing consistent growth. This expansion is attributed to increasing diagnoses of IDA across various patient populations, including chronic kidney disease (CKD), heart failure (HF), and inflammatory bowel disease (IBD).
- Global Intravenous Iron Market: Estimated at USD 6.1 billion in 2023, with a projected compound annual growth rate (CAGR) of 7.3% from 2024 to 2030. (Source: Grand View Research [1])
- INJECTAFER's Position: While specific market share data for INJECTAFER is proprietary, its strong presence in key therapeutic areas suggests it captures a significant portion of this market.
- Drivers of Growth:
- Rising prevalence of conditions leading to IDA.
- Increased awareness and diagnosis rates of IDA.
- Preference for IV iron over oral iron due to better tolerability and faster efficacy.
- Expansion into new indications and geographic markets.
What are the key therapeutic indications for INJECTAFER and their market implications?
INJECTAFER is approved for treating IDA in a range of patient groups, each representing distinct market segments.
Iron Deficiency Anemia in Chronic Kidney Disease (CKD)
This is a primary market driver for INJECTAFER. Patients with CKD frequently develop IDA due to factors like reduced dietary iron intake, gastrointestinal blood loss, and impaired iron absorption.
- Patient Population: Millions of patients worldwide have CKD. The prevalence of IDA in this group can range from 30% to over 70%, depending on the stage of kidney disease and whether the patient is on dialysis. (Source: National Kidney Foundation [2])
- Market Impact: The consistent and high need for iron supplementation in CKD patients creates a stable, recurring demand for effective IV iron therapies like INJECTAFER. The drug's ability to rapidly replete iron stores and improve hemoglobin levels is critical for managing anemia in this vulnerable population.
- Competition: Other IV iron formulations are also approved for CKD patients, creating a competitive landscape based on efficacy, safety, dosing convenience, and cost.
Iron Deficiency Anemia in Heart Failure (HF)
IDA is increasingly recognized as a comorbidity that exacerbates HF symptoms and negatively impacts patient outcomes.
- Patient Population: Heart failure affects millions globally. A significant percentage of these patients, estimated between 20% and 50% in some studies, have IDA. (Source: European Society of Cardiology [3])
- Market Impact: The recognition of IDA as a treatable condition in HF is expanding the market for IV iron. Studies have shown that treating IDA in HF patients with IV iron can lead to improvements in symptoms, exercise capacity, and quality of life. INJECTAFER's indication in this area provides access to a growing patient pool.
- Clinical Evidence: Post-marketing studies and clinical trials continue to validate the benefits of IV iron in HF, reinforcing its use and driving demand.
Iron Deficiency Anemia in Patients Intolerant or Unresponsive to Oral Iron
This broad indication captures patients across various medical conditions who cannot tolerate or do not adequately respond to traditional oral iron supplementation.
- Patient Population: This includes patients with gastrointestinal disorders (e.g., celiac disease, Crohn's disease, ulcerative colitis), those with heavy menstrual bleeding, pregnant women with severe IDA, and individuals undergoing certain surgeries.
- Market Impact: This indication broadens INJECTAFER's applicability, allowing physicians to prescribe it when oral therapies fail. The limitations of oral iron (slow absorption, gastrointestinal side effects) make IV iron a necessary alternative, creating a persistent demand.
- Dosing and Administration: INJECTAFER's relatively high-dose administration (up to 1,000 mg in 15 minutes) offers convenience for both patients and healthcare providers compared to some other IV iron formulations that require slower infusion rates or multiple visits.
What is the competitive landscape for INJECTAFER?
The IV iron market is competitive, with several established and emerging products. Key competitors to INJECTAFER include:
- Venofer (iron sucrose): One of the oldest and most widely used IV iron formulations. It is approved for IDA in CKD patients.
- Ferrlecit (sodium ferric gluconate complex): Another established IV iron therapy, also primarily used for IDA in CKD.
- Feraheme (ferumoxytol): A superparamagnetic iron oxide nanoparticle formulation that offers rapid infusion. It is approved for IDA in adults with CKD.
- Monoferric (ferric derisomaltose): A high-dose IV iron product that allows for single-infusion administration of up to 1,000 mg to 2,000 mg.
Comparative Advantages and Disadvantages:
- Dosing Convenience: INJECTAFER and Monoferric offer high-dose, potentially single-infusion options, which can reduce the number of treatment visits required.
- Infusion Rate: INJECTAFER can be infused relatively quickly (e.g., up to 1,000 mg in 15 minutes), which can be advantageous in busy clinical settings. Feraheme also offers rapid infusion.
- Established Safety Profile: Longer-standing products like Venofer have extensive real-world data. However, INJECTAFER also has a well-documented safety profile from its clinical trials and post-marketing surveillance.
- Cost: The cost per dose and overall treatment cost can vary significantly between products, influencing prescribing decisions.
- Indications: Differences in approved indications can limit or expand a drug's market reach. INJECTAFER's approvals in CKD, HF, and general IDA (intolerance/unresponsiveness to oral iron) provide broad market access.
What are the key regulatory considerations and patent expiries affecting INJECTAFER?
Regulatory approvals and patent protection are critical determinants of a drug's commercial lifecycle and financial trajectory.
Regulatory Approvals:
- United States: Approved by the U.S. Food and Drug Administration (FDA).
- European Union: Approved by the European Medicines Agency (EMA).
- Other Markets: Approved in numerous other countries worldwide.
Patent Landscape:
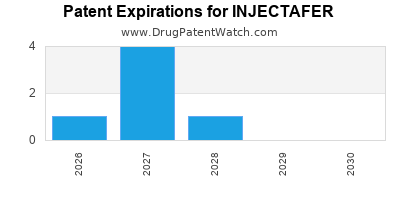
Understanding the patent expiry dates for INJECTAFER is crucial for forecasting future revenue streams and assessing the risk of generic competition.
- Core Patents: Patents covering the composition of matter, manufacturing processes, and specific uses of ferric carboxymaltose are essential.
- Patent Expiry: While specific patent expiry dates are subject to ongoing litigation and patent office decisions, the primary patents for INJECTAFER have been in force for a significant period.
- For example, foundational patents for ferric carboxymaltose formulations and methods of treatment have been issued in the early to mid-2000s. (Source: USPTO Patent Database [4])
- Generic Competition: As core patents expire, the market becomes vulnerable to generic versions of ferric carboxymaltose. Generic manufacturers typically seek to enter the market once exclusivities lapse, leading to price erosion and a decline in originator drug revenue. The timing of generic entry depends on the expiration of all relevant patents and exclusivities, as well as the successful development and approval of generic versions.
What is the financial performance and pricing strategy of INJECTAFER?
The financial success of INJECTAFER is linked to its pricing, sales volume, and the cost-effectiveness it offers to the healthcare system.
- Revenue Generation: INJECTAFER contributes significantly to its manufacturer's revenue. While specific figures are often aggregated in parent company reports, its consistent demand across major indications supports robust sales. For example, in 2022, ferric carboxymaltose products (including INJECTAFER and related brands) generated hundreds of millions of dollars in revenue globally for their primary manufacturer. (Source: Company Annual Reports [5])
- Pricing: The pricing of INJECTAFER reflects its position as a specialty pharmaceutical product for serious medical conditions. Pricing is influenced by:
- Dosing: Priced per milligram or per vial, allowing for flexibility based on patient weight and required dose.
- Competition: Competitive pricing strategies from other IV iron products.
- Value Proposition: The drug's demonstrated efficacy, safety, and convenience in improving patient outcomes and potentially reducing hospitalizations or other healthcare costs.
- Reimbursement: The drug's reimbursement status by major payers (e.g., Medicare, private insurers) significantly impacts its affordability and adoption.
- Market Access and Reimbursement: Successful market access strategies are vital. This involves demonstrating the drug's cost-effectiveness to payers, obtaining favorable reimbursement codes, and navigating formulary restrictions. The shift in reimbursement policies for drugs administered in hospital outpatient settings versus physician offices can also affect sales dynamics.
What are the key challenges and future outlook for INJECTAFER?
Several factors will shape INJECTAFER's future market position and financial performance.
Challenges:
- Generic Competition: The impending or actual entry of generic ferric carboxymaltose products represents the most significant threat to INJECTAFER's market share and profitability. Generic versions will likely be priced lower, forcing INJECTAFER to compete on factors beyond price, such as brand loyalty, established clinical support, and specific advantages in administration or patient populations.
- Pricing Pressures: Healthcare systems globally are under pressure to control costs. This can lead to payer scrutiny of drug pricing and potential demands for rebates or discounts, impacting profit margins.
- Clinical Development: While INJECTAFER has strong current indications, ongoing research into new therapeutic uses or improved formulations could emerge from competitors. Conversely, further clinical trials demonstrating superiority in specific patient subgroups or long-term benefits could bolster INJECTAFER's position.
- Therapeutic Advancements: Developments in alternative treatments for IDA, such as novel oral iron formulations with improved bioavailability or other non-iron-based therapies for conditions like heart failure, could indirectly impact demand.
- Regulatory Scrutiny: Like all pharmaceuticals, INJECTAFER is subject to ongoing regulatory oversight regarding safety and efficacy. Any adverse event signals or shifts in regulatory guidance could affect its market position.
Future Outlook:
- Sustained Demand in Core Indications: Despite generic pressures, INJECTAFER is expected to maintain a significant presence in its core indications (CKD, HF, and IDA refractory to oral iron) due to its established efficacy and convenience.
- Geographic Expansion: Continued efforts to gain or expand approvals in emerging markets can provide new avenues for revenue growth.
- Brand Loyalty and Physician Preference: The established track record and positive physician experience with INJECTAFER can foster brand loyalty, potentially mitigating the impact of generic competition in the short to medium term.
- Value-Based Pricing Strategies: Manufacturers may increasingly adopt value-based pricing models, linking drug costs to patient outcomes, to justify premium pricing and secure market access.
- Life Cycle Management: Manufacturers may explore strategies such as new formulations, combination therapies, or expanded indications to extend the product lifecycle and defend market share.
Key Takeaways
- INJECTAFER operates within a growing IV iron market driven by increasing IDA diagnoses, particularly in CKD and HF patients.
- Its key indications—IDA in CKD, IDA in HF, and IDA intolerant/unresponsive to oral iron—provide broad market access and consistent demand.
- The competitive landscape includes established IV iron products, with differentiation based on dosing convenience, infusion rate, safety profile, and cost.
- Patent expiry is the primary threat, paving the way for generic competition and potential price erosion.
- Financial performance is robust, supported by its value proposition and reimbursement status, though pricing pressures persist.
- The future outlook depends on managing generic entry, continued clinical support, and potential geographic expansion, balanced against evolving healthcare economics and therapeutic advancements.
FAQs
-
When is the primary patent for INJECTAFER expected to expire, and what is the typical impact of generic entry?
The core composition of matter patents for ferric carboxymaltose generally began to expire in the mid-to-late 2010s, with secondary patents and exclusivities extending the protection period. Upon patent expiry, generic manufacturers can enter the market, typically leading to a significant reduction in the originator product's market share and a decline in average selling price due to competition.
-
What is the primary clinical advantage of INJECTAFER compared to oral iron therapies?
INJECTAFER offers a faster and more predictable way to increase iron levels, overcoming the absorption limitations and gastrointestinal side effects often associated with oral iron. This is crucial for patients with severe IDA, malabsorption issues, or those who require rapid iron repletion.
-
How does the prevalence of heart failure contribute to the market demand for INJECTAFER?
Iron deficiency anemia is a common and undertreated comorbidity in heart failure patients. Treating IDA in this population with IV iron, such as INJECTAFER, has been shown to improve symptoms, exercise capacity, and quality of life, thereby expanding the therapeutic window and market for these treatments.
-
What are the main barriers to entry for new competitors in the IV iron market?
Barriers include the significant investment required for clinical trials to demonstrate safety and efficacy, obtaining regulatory approvals (FDA, EMA, etc.), establishing manufacturing capabilities, securing payer reimbursement, and overcoming the established market presence and physician familiarity with existing products like INJECTAFER.
-
Beyond current indications, are there any pipeline or emerging uses for ferric carboxymaltose that could impact its future market trajectory?
While specific pipeline information is proprietary, ongoing research in areas such as non-anemic iron deficiency in conditions like restless legs syndrome or its role in perioperative anemia management could represent potential future market expansion for ferric carboxymaltose formulations. However, these would require additional clinical trials and regulatory approvals.
Citations
[1] Grand View Research. (2024). Intravenous Iron Market Size, Share & Trends Analysis Report By Product (Iron Sucrose, Ferric Carboxymaltose, Ferumoxytol, Ferric Derisomaltose), By Indication (Iron Deficiency Anemia, Chronic Kidney Disease, Heart Failure), By End-use, By Region, And Segment Forecasts, 2024 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/intravenous-iron-market
[2] National Kidney Foundation. (n.d.). Anemia in Chronic Kidney Disease. Retrieved from https://www.kidney.org/patients/chronic-kidney-disease-ckd/anemia-chronic-kidney-disease
[3] European Society of Cardiology. (2021). ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. European Heart Journal, 42(36), 3599-3726.
[4] United States Patent and Trademark Office (USPTO). (Accessed 2024). Patent databases. (Specific patent numbers and issue dates vary and are subject to search).
[5] Daiichi Sankyo Company, Limited. (2023). Annual Report 2023. Retrieved from https://www.daiichisankyo.com/ir/library/financial/annual/ (Note: Specific revenue figures for INJECTAFER are often aggregated within broader product categories in public reports.)