Last updated: February 19, 2026
AVEED (testosterone undecanoate) is an injectable androgen therapy used for treating testosterone deficiency (hypogonadism) in adult men. Its market trajectory is shaped by patent expiry, regulatory scrutiny, and competition from established and emerging therapies.
What is AVEED and its Indication?
AVEED is a long-acting injectable formulation of testosterone undecanoate developed by Besins Healthcare and previously marketed by Endo Pharmaceuticals in the United States. It is administered intramuscularly and designed to provide sustained testosterone levels, reducing the frequency of injections compared to shorter-acting ester forms of testosterone. The primary indication for AVEED is the treatment of hypogonadism in adult men, a condition characterized by the testes' inability to produce sufficient testosterone. This can lead to symptoms such as fatigue, low libido, erectile dysfunction, and decreased muscle mass.
How does AVEED work?
AVEED functions as a direct replacement for endogenous testosterone. Upon intramuscular injection, testosterone undecanoate is slowly absorbed from the injection depot into the bloodstream. In the body, the undecanoate ester is cleaved by esterases, releasing free testosterone. This free testosterone then binds to androgen receptors, mimicking the action of naturally produced testosterone. It mediates its effects on various tissues, including the skeletal muscle, bone, brain, and reproductive organs, thereby restoring physiological testosterone levels and alleviating hypogonadal symptoms.
What is the Patent Landscape for AVEED?
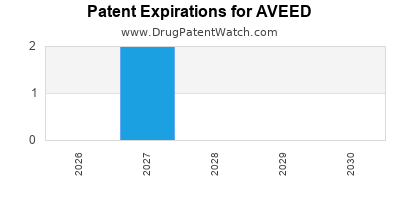
The patent landscape for AVEED is a critical factor influencing its market exclusivity and the potential for generic competition. The primary patent protecting AVEED in the United States was U.S. Patent No. 6,670,359, which covers the testosterone undecanoate formulation and its use for testosterone replacement therapy. This patent was originally set to expire in September 2021. However, patent term extensions (PTEs) and other potential patent strategies can alter the effective market exclusivity period.
- U.S. Patent No. 6,670,359: Filed on March 29, 2002, and issued on December 30, 2003.
- Original Expiration Date: September 20, 2021.
- Patent Term Extension (PTE): Applicable due to regulatory review delays. The PTE for this patent added approximately 1,111 days (about 3 years) to its term, extending its protection.
- Estimated Effective Expiration: This extension pushed the effective expiration of the core patent beyond the original date. A precise calculation would depend on specific FDA approval dates and calculation methodologies.
- Other Potential Patents: Besins Healthcare and its licensees may hold or have filed for secondary patents covering manufacturing processes, new formulations, or specific uses, which could further extend market exclusivity or create barriers to entry for generic manufacturers.
- Generic Entry: The expiration of key patents, particularly the '359 patent and any applicable PTE, opens the door for generic manufacturers to seek U.S. Food and Drug Administration (FDA) approval for their own versions of testosterone undecanoate injections. Companies often file Abbreviated New Drug Applications (ANDAs) to demonstrate bioequivalence.
What are the Regulatory Considerations for AVEED?
AVEED has faced significant regulatory scrutiny, particularly concerning its safety profile and the risk of serious adverse events.
- Boxed Warning: AVEED carries a boxed warning, the FDA's strongest warning, regarding the risk of serious cardiorespiratory events, including anaphylaxis and pulmonary oil embolism, associated with the injection process itself. This risk is particularly elevated with rapid injection rates or excessive volumes.
- Risk Evaluation and Mitigation Strategy (REMS): To mitigate these risks, AVEED is subject to a REMS program. This program requires healthcare providers to be specially certified and to administer AVEED only in a healthcare setting where patients can be monitored for at least 30 minutes post-injection. The REMS also mandates specific administration techniques to minimize the risk of adverse events.
- FDA Actions: The FDA has previously taken actions related to AVEED's safety. In 2018, Endo Pharmaceuticals voluntarily withdrew AVEED from the U.S. market due to concerns about the REMS program's effectiveness in ensuring safe administration and patient monitoring, and the resulting commercial viability. While the drug was initially withdrawn, the patent landscape and potential for reintroduction or licensing by other entities remain relevant.
- Labeling Updates: Throughout its market history, AVEED's labeling has been updated to reflect new safety information and to reinforce the critical aspects of the REMS program.
Who are the Key Competitors in the Testosterone Replacement Therapy Market?
The market for testosterone replacement therapy (TRT) is competitive, with a variety of formulations and delivery methods available. AVEED's competitors can be categorized by their delivery systems and pharmacokinetics.
- Short-Acting Injectables:
- Testosterone Cypionate (e.g., Depo-Testosterone, generic formulations): Intramuscular injection, typically administered every 1-2 weeks. This is a long-established and widely used option.
- Testosterone Enanthate (e.g., Xyosted, generic formulations): Similar to cypionate, administered intramuscularly every 1-2 weeks. Xyosted offers a needle-free auto-injector option.
- Long-Acting Injectables:
- Nebido (Testosterone Undecanoate, marketed outside the US by Bayer): This is the international counterpart to AVEED, also a long-acting intramuscular injectable testosterone undecanoate, often administered every 10-14 weeks after an initial loading phase.
- Transdermal Gels and Patches:
- AndroGel (testosterone gel): Applied daily to the skin.
- Testim (testosterone gel): Applied daily to the skin.
- Fortesta (testosterone gel): Applied daily to the skin.
- Androderm (testosterone patch): Applied daily to the skin.
- These are popular due to ease of use but can have issues with transference to partners and inconsistent absorption.
- Other Formulations:
- Testosterone Pellets (e.g., Testopel): Subcutaneous implants inserted every 3-6 months.
- Intranasal Gels (e.g., Natesto): Applied three times daily into the nasal cavity.
AVEED's key differentiator was its long-acting injectable profile, aiming for less frequent dosing. However, the associated safety concerns and REMS requirements have significantly impacted its market penetration and competitive standing compared to other established and less burdensome TRT options.
What is the Financial Trajectory of AVEED?
The financial trajectory of AVEED has been characterized by initial market entry challenges, followed by a significant withdrawal from the U.S. market, impacting its revenue generation.
- Initial Launch and Sales Performance: Following its FDA approval in 2014, AVEED was launched with the expectation of capturing a segment of the TRT market seeking less frequent injections. However, sales performance was reportedly modest, impacted by the complexities of its REMS program and physician adoption hurdles. Precise historical sales figures are difficult to ascertain post-withdrawal, but pre-withdrawal sales did not meet aggressive growth expectations.
- Market Withdrawal: In March 2018, Endo Pharmaceuticals announced the voluntary withdrawal of AVEED from the U.S. market. The company cited the significant investment required to maintain the REMS program and its limited commercial viability as the primary reasons. This withdrawal effectively halted revenue generation from AVEED in the U.S.
- Post-Withdrawal Landscape: While AVEED is no longer actively marketed in the U.S. by Endo, the underlying patent protection for testosterone undecanoate remains relevant. This could allow for a future reintroduction by a different entity or the development of generic versions once all intellectual property protections expire and regulatory hurdles are addressed. The international market, particularly with Nebido, continues to generate revenue for the originator.
- Generic Potential: The eventual expiry of patents and the resolution of regulatory challenges could lead to the re-entry of testosterone undecanoate into the U.S. market via generic manufacturers. This would likely introduce price competition and potentially increase market access if safety concerns can be adequately managed by the generic products and their associated REMS.
What are the Future Market Prospects for AVEED and Similar Therapies?
The future market prospects for AVEED, and long-acting injectable testosterone undecanoate formulations, are contingent on several factors, including resolution of safety concerns, regulatory pathways, and competitive dynamics.
- Regulatory Re-entry and REMS Burden: For AVEED or a similar formulation to re-enter the U.S. market successfully, a robust and commercially viable REMS program would be essential. The significant costs and logistical challenges associated with the previous REMS contributed to its withdrawal. Future iterations would need to demonstrate improved adherence and effectiveness in mitigating risks.
- Generic Competition: As patent protections expire, the market is likely to see generic testosterone undecanoate injections. The success of these generics will depend on their ability to meet FDA bioequivalence standards, navigate any remaining REMS requirements, and offer a cost-effective alternative to existing TRT options.
- Demand for Long-Acting Formulations: There remains an underlying demand for TRT options that offer less frequent dosing, providing convenience and potentially improving patient adherence. Long-acting injectables, if safely administered, can fulfill this need.
- Advancements in Delivery Systems: Innovation in TRT delivery systems continues. The development of novel long-acting injectables, potentially with improved safety profiles or easier administration, could further shape the competitive landscape and influence the viability of existing or reintroduced therapies.
- Market Size of TRT: The global TRT market is substantial and projected to grow due to an aging male population, increased diagnosis of hypogonadism, and greater awareness of testosterone deficiency symptoms. This overall market growth provides an opportunity for well-positioned therapies.
The success of any future AVEED-like product will depend on its ability to balance efficacy with a manageable and cost-effective safety profile, a challenge that has historically impacted this specific formulation.
Key Takeaways
- AVEED (testosterone undecanoate) is a long-acting injectable TRT with a complex market history marked by regulatory challenges and market withdrawal in the U.S.
- The primary U.S. patent for AVEED has expired or is nearing the end of its extended term, opening the door for generic competition.
- AVEED carries a significant boxed warning for serious cardiorespiratory events and pulmonary oil embolism, necessitating a strict REMS program.
- Endo Pharmaceuticals voluntarily withdrew AVEED from the U.S. market in 2018 due to the commercial burden of its REMS program.
- The competitive TRT market includes various injectables, transdermal systems, and pellets, with AVEED's key differentiator being its long-acting profile.
- Future prospects for AVEED or similar formulations hinge on the ability to navigate regulatory requirements, particularly the REMS, and effectively compete with established and emerging TRT options.
Frequently Asked Questions
- When did AVEED receive FDA approval?
AVEED received U.S. FDA approval on April 4, 2014. [1]
- What was the specific reason for AVEED's withdrawal from the U.S. market?
Endo Pharmaceuticals voluntarily withdrew AVEED in March 2018 due to the significant costs and logistical challenges associated with maintaining the drug's Risk Evaluation and Mitigation Strategy (REMS) and its limited commercial viability. [2]
- Does the withdrawal of AVEED from the U.S. market affect its patent protection?
No, the market withdrawal does not directly affect patent protection. Patents remain in force until their expiration date, and intellectual property can be licensed or utilized by other entities.
- Is testosterone undecanoate still available in the U.S. market in any form?
Testosterone undecanoate is available internationally as Nebido. In the U.S., while AVEED was withdrawn, other forms of testosterone replacement therapy, including different formulations and other testosterone esters, remain available.
- What are the estimated costs associated with the REMS program for AVEED?
Specific detailed financial disclosures on the REMS program costs were not publicly itemized by the manufacturer, but the "significant investment" cited by Endo points to substantial operational, training, and compliance expenses necessary for its implementation and oversight.
Citations
[1] U.S. Food & Drug Administration. (2014, April 4). FDA approves AVEED (testosterone undecanoate) injection, for intramuscular use, for testosterone replacement therapy in adult men. [Press release]. https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/aveed-risk-evaluation-and-mitigation-strategy-rems
[2] Endo International plc. (2018, March 8). Endo Announces Voluntary Withdrawal of AVEED® (testosterone undecanoate) Injection from the U.S. Market. [Press release]. https://www.endo.com/newsroom/press-releases/endo-announces-voluntary-withdrawal-of-aveed-testosterone-undecanoate-injection-from-the-us-market