Last updated: February 19, 2026
Spinraza (nusinersen) is a treatment for spinal muscular atrophy (SMA), a rare genetic neuromuscular disorder. Developed by Biogen and Ionis Pharmaceuticals, its market trajectory is defined by its first-mover advantage, high cost, and evolving competitive landscape. This analysis examines the patent portfolio, regulatory approvals, sales performance, and future market outlook for Spinraza.
What is Spinraza's Patent Landscape?
Spinraza's intellectual property is primarily secured through a series of patents covering its composition of matter, method of use, and manufacturing processes. The foundational patents provide robust protection, but ongoing litigation and the potential for biosimilar entrants in the future necessitate close monitoring.
- Composition of Matter Patents: These patents protect the chemical structure of nusinersen itself.
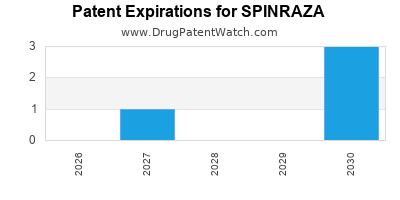
- U.S. Patent No. 9,416,143, titled "2'-O-methoxyethyl-substituted oligonucleotides for treating neurodegenerative diseases," was a key patent covering the compound. This patent was granted in 2016 and is set to expire in 2026 [1].
- European Patent EP 2 807 046 B1, covering similar composition of matter claims, also contributes to Spinraza's global patent protection.
- Method of Use Patents: These patents claim the use of nusinersen for treating SMA.
- U.S. Patent No. 9,700,704, titled "Methods of treating spinal muscular atrophy," is crucial for its therapeutic indication. This patent has an expected expiration in 2030 [1].
- Additional method of use patents cover specific dosing regimens and patient populations, extending the period of exclusivity.
- Manufacturing Process Patents: Patents related to the synthesis and production of nusinersen offer further layers of protection, making it more challenging for competitors to replicate the drug.
- Patent Challenges and Litigation: Spinraza has faced patent challenges, particularly in the United States. Litigation often centers on claims of invalidity, non-infringement, or obviousness. For example, Biogen has been involved in legal disputes aimed at defending its core Spinraza patents against challenges from potential generic manufacturers [2]. Decisions in these cases can significantly impact the timeline for generic competition.
- Exclusivity Extensions: Regulatory exclusivities, such as Orphan Drug Exclusivity (ODE) in the US and EU, provide additional periods of market protection beyond patent expiry. Spinraza received 7-year ODE in the US (granted in 2016) and 10-year ODE in the EU (granted in 2017) [3]. These exclusivities are separate from patent protection and can extend market exclusivity until 2023 (US) and 2027 (EU), respectively.
The interplay between patent expiry dates, ongoing litigation, and regulatory exclusivities dictates the long-term market exclusivity of Spinraza. The expiration of key composition of matter patents in the coming years may open avenues for biosimilar or generic competition, depending on the complexity of the molecule and manufacturing processes.
What are Spinraza's Regulatory Approvals and Market Access?
Spinraza's regulatory journey began with its approval in the United States, followed by approvals in other major markets. Access is often facilitated by its orphan drug designation, which provides incentives and expedited review pathways.
- United States:
- Approved by the Food and Drug Administration (FDA) on December 23, 2016, for the treatment of SMA in pediatric and adult patients [3].
- The approval was based on pivotal trials demonstrating significant efficacy in improving motor function and survival.
- European Union:
- Received marketing authorization from the European Medicines Agency (EMA) in May 2017 [3].
- Approved for infantile-onset SMA and later extended to later-onset SMA patients.
- Japan:
- Approved by the Ministry of Health, Labour and Welfare (MHLW) in March 2017 [3].
- Other Markets: Spinraza has subsequently received approvals in numerous other countries, including Canada, Australia, Brazil, and various nations in Asia and the Middle East.
- Navigating Market Access: Securing reimbursement and market access for Spinraza is a significant undertaking due to its high per-patient cost. Health technology assessment (HTA) bodies in different countries evaluate its cost-effectiveness.
- Payer Negotiations: Biogen engages in price negotiations with national health systems and private payers globally. These negotiations are critical for patient access.
- Value Frameworks: The drug's clinical benefits, such as improved motor function, reduced need for ventilation, and increased survival, are key arguments in demonstrating its value to payers.
- Real-World Evidence: Post-approval studies and the generation of real-world evidence play a role in reinforcing the drug's value proposition and informing payer decisions.
The broad regulatory approvals have enabled Spinraza to establish a significant global footprint. However, ongoing price negotiations and the scrutiny of HTA bodies remain critical determinants of patient access and market penetration.
How has Spinraza Performed Financially?
Spinraza has been a significant revenue driver for Biogen, demonstrating strong commercial success since its launch. Its financial performance reflects its status as a breakthrough therapy for a rare and devastating disease.
- Sales Trajectory:
- 2017: Spinraza generated approximately $476 million in net sales in its first full year on the market [4].
- 2018: Sales increased to $1.61 billion, marking substantial growth [4].
- 2019: Net sales reached $2.30 billion, further solidifying its commercial success [4].
- 2020: Spinraza achieved $2.75 billion in net sales, continuing its upward trend [4].
- 2021: Net sales were $2.80 billion [4].
- 2022: Spinraza generated $2.61 billion in net sales [4].
- 2023 (first nine months): Spinraza reported approximately $1.95 billion in net sales [5].
- Factors Driving Sales:
- First-in-Class Status: As the first approved treatment for SMA, Spinraza captured a substantial portion of the addressable market early on.
- Clinical Efficacy: Demonstrated clinical benefits in improving motor function and survival for SMA patients across different age groups.
- Broader Label Expansions: Approvals for later-onset SMA broadened the patient population eligible for treatment.
- Global Rollout: Successful expansion into major international markets.
- Revenue Contribution to Biogen: Spinraza has consistently been one of Biogen's leading products, contributing a substantial percentage to the company's overall revenue. For instance, in 2020, Spinraza accounted for approximately 24% of Biogen's total net revenue [4].
- Cost and Reimbursement: The high per-patient cost of Spinraza (estimated to be over $750,000 annually for multiple doses) is a significant factor in its revenue generation but also a source of scrutiny from payers and patient advocacy groups [6].
- Geographic Breakdown: Sales are distributed globally, with North America and Europe being the largest markets, followed by emerging regions. The growth in emerging markets is influenced by expanding access and reimbursement agreements.
- Impact of Competition: The emergence of newer SMA therapies, such as Zolgensma (onasemnogene abeparvovec), an AAV gene therapy, has introduced competition, particularly for infantile-onset SMA. While Zolgensma is a one-time treatment, Spinraza remains the standard of care for many patients, especially those with later-onset SMA or who do not qualify for gene therapy.
Spinraza's financial performance underscores its critical role in treating SMA and its success as a commercial product. The sustained revenue demonstrates the unmet need and the value proposition of an effective therapeutic intervention.
What is Spinraza's Competitive Landscape and Future Outlook?
The competitive landscape for Spinraza is evolving, influenced by the introduction of gene therapies and other novel approaches to treating SMA. The future outlook depends on its ability to maintain market share, address pricing concerns, and adapt to emerging treatment paradigms.
- Key Competitors:
- Zolgensma (onasemnogene abeparvovec) by AveXis (Novartis): A gene therapy approved for SMA in infants. Zolgensma is a one-time treatment, positioning it as a direct competitor for the infantile-onset SMA population. Its curative potential and single-administration nature represent a significant challenge to Spinraza's long-term treatment model.
- Risdiplam (Evrysdi) by Genentech (Roche): An orally administered small molecule drug approved for SMA in patients 2 months of age and older. Risdiplam offers an alternative to intrathecal administration and is positioned to compete across various age groups, including those not eligible for Zolgensma.
- Emerging Therapies: Research continues into other therapeutic modalities for SMA, including antisense oligonucleotides (ASOs) targeting different genetic pathways, small molecules, and novel gene therapies.
- Spinraza's Differentiating Factors:
- Established Efficacy and Safety Profile: Spinraza has a longer track record of clinical data and real-world use, providing a well-understood efficacy and safety profile across a broad patient population.
- Intrathecal Administration: While a barrier for some, intrathecal delivery allows for direct targeting of the central nervous system.
- Treatment for Later-Onset SMA: Spinraza remains a critical treatment option for patients with later-onset SMA, where gene therapy may not be as effective or indicated.
- Multiple Dosing Regimens: The ability to adjust dosing and treat patients across different stages of the disease.
- Market Dynamics and Future Projections:
- Shifting Treatment Paradigms: The availability of gene therapy and oral medications is gradually shifting the treatment landscape, particularly for the youngest patients.
- Market Segmentation: Spinraza is likely to remain a dominant force in the later-onset SMA segment and for patients who cannot access or tolerate gene therapy or oral treatments.
- Pricing Pressures: The high cost of Spinraza, coupled with the availability of potentially curative gene therapies and more affordable oral options, will likely lead to increased pricing pressure.
- Patent Expirations: The approaching expiration of key patents will open the door for potential generic or biosimilar competition, which could significantly impact Spinraza's market share and profitability.
- Biogen's Strategy: Biogen is likely to focus on optimizing Spinraza's use in its established patient populations, potentially exploring new indications or delivery methods, and defending its intellectual property to maximize its remaining exclusivity period.
- Growth in Emerging Markets: Continued efforts to expand access and secure reimbursement in emerging markets will be crucial for sustaining revenue growth.
The future of Spinraza will be shaped by its continued clinical utility, the competitive response from newer therapies, and the evolving economic considerations for rare disease treatments. Its market share will depend on its ability to remain a cost-effective and efficacious option for a significant portion of the SMA patient population.
Key Takeaways
- Spinraza's intellectual property is secured by a comprehensive patent portfolio, including composition of matter and method of use patents, with key protections extending into the 2030s.
- Regulatory approvals across major global markets, supported by orphan drug designations, have facilitated widespread patient access.
- Spinraza has achieved substantial financial success, generating billions in net sales annually since its launch, making it a critical revenue driver for Biogen.
- The competitive landscape is intensifying with the emergence of gene therapies (Zolgensma) and oral small molecules (Risdiplam), particularly impacting the infantile-onset SMA market.
- Spinraza is expected to maintain its position as a leading treatment for later-onset SMA and for patients not eligible for alternative therapies, but patent expiries and pricing pressures pose future challenges.
Frequently Asked Questions
-
When do the core composition of matter patents for Spinraza expire in major markets like the US and EU?
The key composition of matter patent (U.S. Patent No. 9,416,143) for Spinraza is set to expire in the United States in 2026. Similar patents in other regions have comparable expiration timelines, though specific national patent landscapes can vary.
-
What is the approximate annual cost of Spinraza treatment per patient?
The annual cost of Spinraza treatment is estimated to be upwards of $750,000 for multiple doses, making it one of the most expensive treatments for rare diseases.
-
How does Spinraza's mechanism of action differ from gene therapies like Zolgensma for SMA?
Spinraza is an antisense oligonucleotide that works by increasing the production of functional SMN protein by altering the splicing of the SMN2 gene. Zolgensma, a gene therapy, delivers a functional copy of the SMN1 gene to replace the defective one.
-
What impact has the oral medication Risdiplam had on Spinraza's market share?
Risdiplam, as an orally administered treatment, competes with Spinraza across various age groups. Its availability has led to some patient and physician preference shifts, particularly for individuals seeking a less invasive administration route, potentially moderating Spinraza's market share growth in certain segments.
-
Are there any potential new indications or expanded uses being explored for Spinraza?
While Spinraza is primarily approved for SMA, ongoing research and clinical trials may explore its efficacy in other neurological conditions with similar underlying mechanisms or in different patient subgroups within SMA to potentially expand its therapeutic application.
Citations
[1] U.S. Patent and Trademark Office. (n.d.). Patent Full-Text and Image Database. Retrieved from uspto.gov.
[2] Biogen Inc. (2023). Biogen Reports Fourth Quarter and Full Year 2022 Results. (Press Release).
[3] Ionis Pharmaceuticals, Inc. (2023). Ionis Pharmaceuticals Annual Report 2022. Retrieved from investors.ionispharma.com.
[4] Biogen Inc. (2023). Biogen Reports First Quarter 2023 Results. (Press Release).
[5] Biogen Inc. (2023). Biogen Reports Third Quarter 2023 Results. (Press Release).
[6] U.S. Food and Drug Administration. (2016, December 23). FDA Approves First Treatment for Spinal Muscular Atrophy. (Press Release).