Loxapine - Generic Drug Details
✉ Email this page to a colleague
What are the generic drug sources for loxapine and what is the scope of freedom to operate?
Loxapine
is the generic ingredient in five branded drugs marketed by Nova Pneuma, Teva Branded Pharm, Actavis Labs Ut Inc, Elite Labs Inc, Lannett Co Inc, Rising, and Watson Labs, and is included in eleven NDAs. There is one patent protecting this compound. Additional information is available in the individual branded drug profile pages.Loxapine has twenty-one patent family members in six countries.
There are eight drug master file entries for loxapine.
Summary for loxapine
| International Patents: | 21 |
| US Patents: | 1 |
| Tradenames: | 5 |
| Applicants: | 7 |
| NDAs: | 11 |
| Drug Master File Entries: | 8 |
| Raw Ingredient (Bulk) Api Vendors: | 52 |
| Clinical Trials: | 26 |
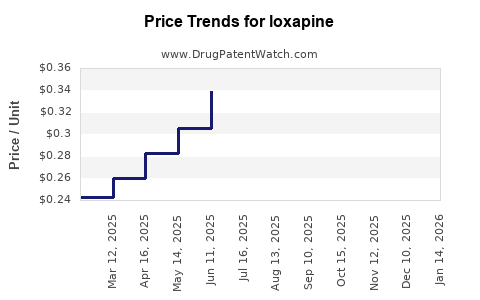
| Drug Prices: | Drug price trends for loxapine |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for loxapine |
| What excipients (inactive ingredients) are in loxapine? | loxapine excipients list |
| DailyMed Link: | loxapine at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for loxapine
Generic Entry Date for loxapine*:
Constraining patent/regulatory exclusivity:
Dosage:
POWDER;INHALATION |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for loxapine
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University Hospital, Strasbourg, France | PHASE2 |
| Assistance Publique - Hôpitaux de Paris | Phase 3 |
| Lariboisière-Saint Louis clinical research unit | Phase 3 |
Medical Subject Heading (MeSH) Categories for loxapine
Anatomical Therapeutic Chemical (ATC) Classes for loxapine
US Patents and Regulatory Information for loxapine
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lannett Co Inc | LOXAPINE SUCCINATE | loxapine succinate | CAPSULE;ORAL | 090695-002 | Sep 26, 2011 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Teva Branded Pharm | LOXITANE | loxapine succinate | TABLET;ORAL | 017525-008 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Teva Branded Pharm | LOXITANE | loxapine succinate | CAPSULE;ORAL | 017525-001 | Approved Prior to Jan 1, 1982 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Elite Labs Inc | LOXAPINE SUCCINATE | loxapine succinate | CAPSULE;ORAL | 076868-003 | Aug 4, 2005 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for loxapine
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Nova Pneuma | ADASUVE | loxapine | POWDER;INHALATION | 022549-001 | Dec 21, 2012 | ⤷ Start Trial | ⤷ Start Trial |
| Nova Pneuma | ADASUVE | loxapine | POWDER;INHALATION | 022549-001 | Dec 21, 2012 | ⤷ Start Trial | ⤷ Start Trial |
| Nova Pneuma | ADASUVE | loxapine | POWDER;INHALATION | 022549-001 | Dec 21, 2012 | ⤷ Start Trial | ⤷ Start Trial |
| Nova Pneuma | ADASUVE | loxapine | POWDER;INHALATION | 022549-001 | Dec 21, 2012 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for loxapine
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Ferrer Internacional S.A. | Adasuve | loxapine | EMEA/H/C/002400Adasuve is indicated for the rapid control of mild-to-moderate agitation in adult patients with schizophrenia or bipolar disorder. Patients should receive regular treatment immediately after control of acute agitation symptoms. | Authorised | no | no | no | 2013-02-20 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for loxapine
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Japan | 2007516403 | ⤷ Start Trial | |
| Canada | 2526478 | PROCEDE DE REGULATION THERMIQUE UNIFORME D'UN SUBSTRAT ; UNITE DE CHAUFFAGE INTEGREE ET UNITE D'ADMINISTRATION DE MEDICAMENTS RECOURANT A CE PROCEDE (METHODS OF CONTROLLING UNIFORMITY OF SUBSTRATE TEMPERATURE AND SELF-CONTAINED HEATING UNIT AND DRUG-SUPPLY UNIT EMPLOYING SAME) | ⤷ Start Trial |
| European Patent Office | 1625334 | UNITÉ DE CHAUFFAGE AUTONOME ALLUMÉE PAR PERCUSSION (PERCUSSIVELY IGNITED SELF-CONTAINED HEATING UNIT) | ⤷ Start Trial |
| Canada | 2526475 | UNITE DE CHAUFFAGE AUTONOME A ALLUMAGE OPTIQUE OU ELECTRIQUE, ET UNITE D'ADMINISTRATION DE MEDICAMENT UTILISANT CETTE UNITE DE CHAUFFAGE (OPTICALLY IGNITED OR ELECTRICALLY IGNITED SELF-CONTAINED HEATING UNIT AND DRUG-SUPPLY UNIT EMPLOYING SAME) | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for loxapine
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1389098 | 2013C/054 | Belgium | ⤷ Start Trial | PRODUCT NAME: LOXAPINE; AUTHORISATION NUMBER AND DATE: EU/1/13/823/001 20130225 |
| 1389098 | 300609 | Netherlands | ⤷ Start Trial | PRODUCT NAME: LOXAPINE; REGISTRATION NO/DATE: EU/1/13/823/001-002 20130220 |
| 1389098 | SPC/GB13/055 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: LOXAPINE; REGISTERED: UK EU/1/13/823/001 20130220; UK EU/1/13/823/002 20130220 |
| 1389098 | C300609 | Netherlands | ⤷ Start Trial | PRODUCT NAME: LOXAPINE; REGISTRATION NO/DATE: EU/1/13/823/001-002 20130220 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for Loxapine
More… ↓