Recordati Rare Company Profile
✉ Email this page to a colleague
What is the competitive landscape for RECORDATI RARE
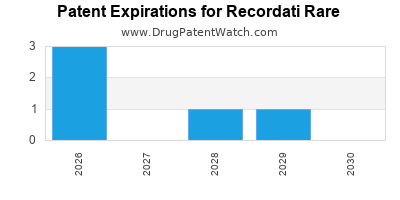
RECORDATI RARE has twelve approved drugs.
There are ten US patents protecting RECORDATI RARE drugs.
There are two hundred and forty-six patent family members on RECORDATI RARE drugs in sixty countries and forty-nine supplementary protection certificates in twenty countries.
Summary for Recordati Rare
| International Patents: | 246 |
| US Patents: | 10 |
| Tradenames: | 12 |
| Ingredients: | 12 |
| NDAs: | 12 |
Drugs and US Patents for Recordati Rare
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Recordati Rare | ISTURISA | osilodrostat phosphate | TABLET;ORAL | 212801-002 | Mar 6, 2020 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Recordati Rare | SIGNIFOR LAR KIT | pasireotide pamoate | FOR SUSPENSION;INTRAMUSCULAR | 203255-002 | Dec 15, 2014 | RX | Yes | No | 7,473,761 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Recordati Rare | CYSTADROPS | cysteamine hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 211302-001 | Aug 19, 2020 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | |||||
| Recordati Rare | SIGNIFOR | pasireotide diaspartate | SOLUTION;SUBCUTANEOUS | 200677-001 | Dec 14, 2012 | RX | Yes | No | 7,473,761 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Recordati Rare | ISTURISA | osilodrostat phosphate | TABLET;ORAL | 212801-002 | Mar 6, 2020 | RX | Yes | No | 9,434,754 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Recordati Rare | ISTURISA | osilodrostat phosphate | TABLET;ORAL | 212801-003 | Mar 6, 2020 | DISCN | Yes | No | 10,143,680 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Recordati Rare | CARBAGLU | carglumic acid | TABLET, FOR SUSPENSION;ORAL | 022562-001 | Mar 18, 2010 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Recordati Rare
Paragraph IV (Patent) Challenges for RECORDATI RARE drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Injection | 10 mg/mL, 2 mL vial | ➤ Subscribe | 2010-10-01 |
International Patents for Recordati Rare Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Spain | 2761227 | ⤷ Start Trial |
| Ecuador | SP066565 | ⤷ Start Trial |
| Spain | 2417498 | ⤷ Start Trial |
| Norway | 337172 | ⤷ Start Trial |
| Peru | 20070517 | ⤷ Start Trial |
| Hungary | E046490 | ⤷ Start Trial |
| Denmark | 3166596 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Recordati Rare Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2523731 | 400 50015-2020 | Slovakia | ⤷ Start Trial | PRODUCT NAME: OSILODROSTAT VO VSETKYCH FORMACH CHRANENYCH ZAKLADNYM PATENTOM; REGISTRATION NO/DATE: EU/1/19/1407 20200113 |
| 1686964 | PA2015009 | Lithuania | ⤷ Start Trial | PRODUCT NAME: PASIREOTIDUM; REGISTRATION NO/DATE: EU/1/12/753/13 - EU/1/12/753/17 20141119 |
| 1307486 | 2012C/026 | Belgium | ⤷ Start Trial | PRODUCT NAME: PASIREOTIDE OU UN DE SES SELS PHARMACEUTIQUEMENT ACCEPTABLES; AUTHORISATION NUMBER AND DATE: EU/1/12/753/001 20120426 |
| 1686964 | 122015000015 | Germany | ⤷ Start Trial | PRODUCT NAME: PASIREOTID ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ DAVON ODER JEGLICHES HYDRAT DAVON; REGISTRATION NO/DATE: EU/1/12/753/013-017 20141119 |
| 2523731 | C202030030 | Spain | ⤷ Start Trial | PRODUCT NAME: OSILODROSTAT O UNA SAL FARMACEUTICAMENTE ACEPTABLE DEL MISMO, INCLUIDO EL OSILODROSTAT DIHIDROGENO FOSFATO.; NATIONAL AUTHORISATION NUMBER: EU/1/19/1407; DATE OF AUTHORISATION: 20200109; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/19/1407; DATE OF FIRST AUTHORISATION IN EEA: 20200109 |
| 1307486 | C 2012 018 | Romania | ⤷ Start Trial | PRODUCT NAME: PASIREOTIDA SI SARURILE ACCEPTABILE FARMACEUTIC ALEACESTEIACICLO[(4R)-4-(2-AMINOETILCARBAMOILOXI)-L-PROLIL-L-FENILGLICIL-D-TRIPTOFIL-L-LISIL-4-O-BENZIL-L-TIROSIL-L-FENILALANIL-); NATIONAL AUTHORISATION NUMBER: RO EU/1/12/753/001 - RO EU/1/12/753/012; DATE OF NATIONAL AUTHORISATION: 20120424; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EMEA EU/1/12/753/001 - EMEA EU/1/12/753/012; DATE OF FIRST AUTHORISATION IN EEA: 20120424 |
| 1686964 | C01686964/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: PASIREOTIDE; REGISTRATION NO/DATE: SWISSMEDIC 65148 25.02.2015 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.