Canada: These 79 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
DrugPatentWatch® Estimated Loss of Exclusivity Dates in Canada
Generic Entry Dates in Other Countries
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
Summary: Canada: These 79 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
| Tradename | Ingredient | Estimated Entry Opportunity Date |
|---|---|---|
| QVAR REDIHALER | beclomethasone dipropionate | 2027-04-11 |
| CREON | pancrelipase (amylase;lipase;protease) | 2026-08-15 |
| PANCREAZE | pancrelipase (amylase;lipase;protease) | 2027-02-20 |
| ZENPEP | pancrelipase (amylase;lipase;protease) | 2027-02-20 |
| EMBEDA | morphine sulfate; naltrexone hydrochloride | 2026-06-19 |
| RYTARY | carbidopa; levodopa | 2027-12-28 |
| >Tradename | >Ingredient | >Estimated Entry Opportunity Date |
Details: Canada: These 79 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
When can QVAR REDIHALER (beclomethasone dipropionate) generic drug versions launch?
Generic name: beclomethasone dipropionate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: April 11, 2027
Generic Entry Controlled by: Canada Patent 2,682,528
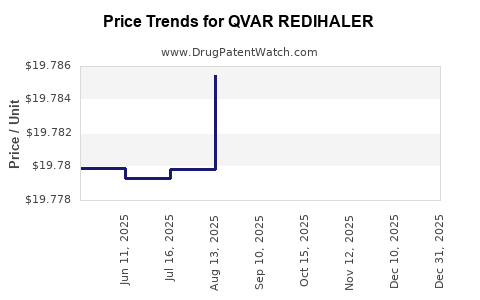
This drug has two hundred and seventy-eight patent family members in twenty-seven countries. There has been litigation on patents covering QVAR REDIHALER
See drug price trends for QVAR REDIHALER.
The generic ingredient in QVAR REDIHALER is beclomethasone dipropionate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the beclomethasone dipropionate profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 15, 2026
Generic Entry Controlled by: Canada Patent 2,619,475
CREON is a drug marketed by
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 15, 2026
Generic Entry Controlled by: Canada Patent 2,619,477
CREON is a drug marketed by
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Canada Patent 2,677,989
PANCREAZE is a drug marketed by
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: Canada Patent 2,677,989
ZENPEP is a drug marketed by
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: Canada Patent 2,655,835
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can RYTARY (carbidopa; levodopa) generic drug versions launch?
Generic name: carbidopa; levodopa
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 28, 2027
Generic Entry Controlled by: Canada Patent 2,711,014
RYTARY is a drug marketed by Impax. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has twenty-five patent family members in twelve countries. There has been litigation on patents covering RYTARY
See drug price trends for RYTARY.
The generic ingredient in RYTARY is carbidopa; levodopa. There are eighteen drug master file entries for this API. Thirty-eight suppliers are listed for this generic product. Additional details are available on the carbidopa; levodopa profile page.
When can CRESEMBA (isavuconazonium sulfate) generic drug versions launch?
Generic name: isavuconazonium sulfate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 14, 2027
Generic Entry Controlled by: Canada Patent 2,663,941
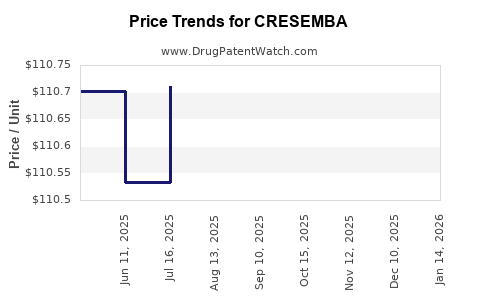
This drug has thirty-two patent family members in nineteen countries. There has been litigation on patents covering CRESEMBA
See drug price trends for CRESEMBA.
The generic ingredient in CRESEMBA is isavuconazonium sulfate. One supplier is listed for this generic product. Additional details are available on the isavuconazonium sulfate profile page.
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 22, 2026
Generic Entry Controlled by: Canada Patent 2,620,380
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can OLYSIO (simeprevir sodium) generic drug versions launch?
Generic name: simeprevir sodium
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 28, 2026
Generic Entry Controlled by: Canada Patent 2,616,580
OLYSIO is a drug marketed by Janssen Prods. There are eight patents protecting this drug.
This drug has sixty-four patent family members in forty-one countries.
See drug price trends for OLYSIO.
The generic ingredient in OLYSIO is simeprevir sodium. There is one drug master file entry for this API. Additional details are available on the simeprevir sodium profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,653,344

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,924,318

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can PICATO (ingenol mebutate) generic drug versions launch?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 18, 2026
Generic Entry Controlled by: Canada Patent 2,634,073

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can SALONPAS (menthol; methyl salicylate) generic drug versions launch?
Generic name: menthol; methyl salicylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 04, 2026
Generic Entry Controlled by: Canada Patent 2,652,220
SALONPAS is a drug marketed by Hisamitsu Pharm Co. There are two patents protecting this drug.
This drug has twenty patent family members in sixteen countries.
The generic ingredient in SALONPAS is menthol; methyl salicylate. There are eighteen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the menthol; methyl salicylate profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Canada Patent 2,702,984

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Canada Patent 2,958,665

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can AMYVID (florbetapir f-18) generic drug versions launch?
Generic name: florbetapir f-18
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 26, 2027
Generic Entry Controlled by: Canada Patent 2,644,530
AMYVID is a drug marketed by Avid Radiopharms Inc. There are two patents protecting this drug.
This drug has fifty-one patent family members in thirty-three countries. There has been litigation on patents covering AMYVID
The generic ingredient in AMYVID is florbetapir f-18. One supplier is listed for this generic product. Additional details are available on the florbetapir f-18 profile page.
When can AXUMIN (fluciclovine f-18) generic drug versions launch?
Generic name: fluciclovine f-18
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 28, 2026
Generic Entry Controlled by: Canada Patent 2,629,227
AXUMIN is a drug marketed by Blue Earth. There are eight patents protecting this drug.
This drug has thirty patent family members in sixteen countries. There has been litigation on patents covering AXUMIN
The generic ingredient in AXUMIN is fluciclovine f-18. One supplier is listed for this generic product. Additional details are available on the fluciclovine f-18 profile page.
When can TRIFERIC (ferric pyrophosphate citrate) generic drug versions launch?
Generic name: ferric pyrophosphate citrate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 20, 2027
Generic Entry Controlled by: Canada Patent 2,638,081
TRIFERIC is a drug marketed by Rockwell Medical Inc. There are two patents protecting this drug.
This drug has thirteen patent family members in eleven countries.
See drug price trends for TRIFERIC.
The generic ingredient in TRIFERIC is ferric pyrophosphate citrate. There are twenty drug master file entries for this API. Additional details are available on the ferric pyrophosphate citrate profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: Canada Patent 2,690,841
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: Canada Patent 2,789,109
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can RASUVO (methotrexate) generic drug versions launch?
Generic name: methotrexate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 20, 2027
Generic Entry Controlled by: Canada Patent 2,659,662
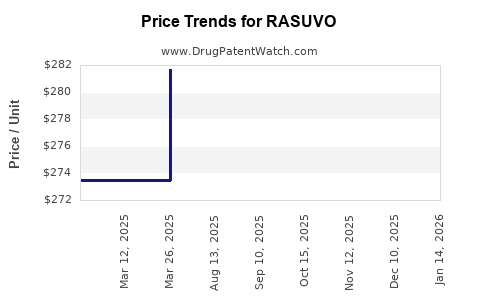
This drug has twenty-nine patent family members in twenty-one countries. There has been litigation on patents covering RASUVO
See drug price trends for RASUVO.
The generic ingredient in RASUVO is methotrexate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the methotrexate profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: Canada Patent 2,700,444
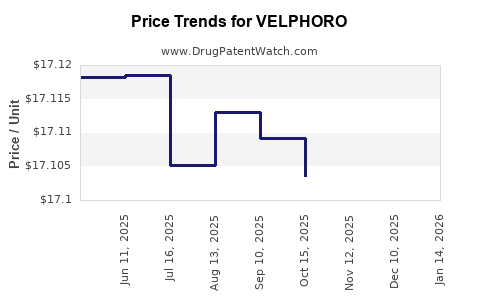
This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 15, 2026
Generic Entry Controlled by: Canada Patent 2,663,172
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 15, 2026
Generic Entry Controlled by: Canada Patent 2,699,142
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can BOSULIF (bosutinib monohydrate) generic drug versions launch?
Generic name: bosutinib monohydrate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,613,053
BOSULIF is a drug marketed by Pf Prism Cv. There are four patents protecting this drug and two Paragraph IV challenges. Three tentatively approved generics are ready to enter the market.
This drug has eighty-one patent family members in thirty countries. There has been litigation on patents covering BOSULIF
See drug price trends for BOSULIF.
The generic ingredient in BOSULIF is bosutinib monohydrate. There are five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the bosutinib monohydrate profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 21, 2026
Generic Entry Controlled by: Canada Patent 2,659,328
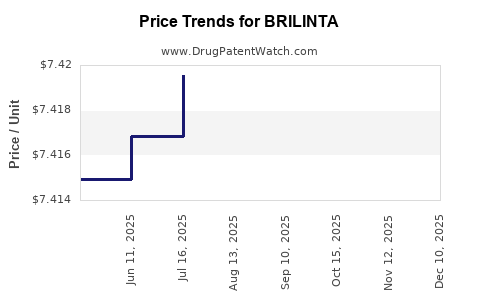
This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can EDARBI (azilsartan kamedoxomil) generic drug versions launch?
Generic name: azilsartan kamedoxomil
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 28, 2027
Generic Entry Controlled by: Canada Patent 2,681,143
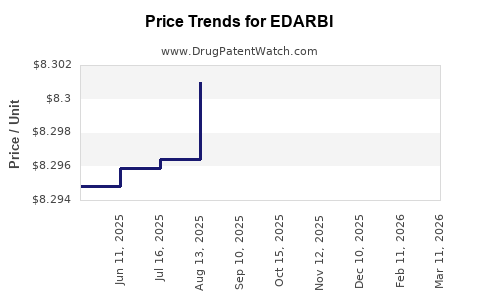
This drug has twenty-one patent family members in seventeen countries. There has been litigation on patents covering EDARBI
See drug price trends for EDARBI.
The generic ingredient in EDARBI is azilsartan kamedoxomil. There are six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the azilsartan kamedoxomil profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,653,344
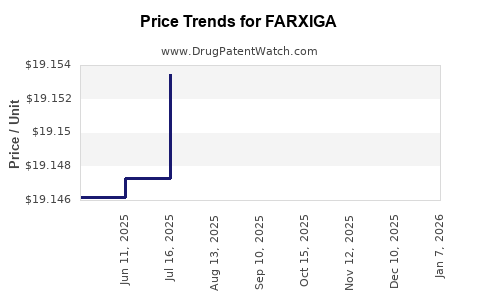
This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,924,318

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can INLYTA (axitinib) generic drug versions launch?
Generic name: axitinib
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: April 05, 2027
Generic Entry Controlled by: Canada Patent 2,682,859
INLYTA is a drug marketed by Pf Prism Cv. There are four patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has ninety-seven patent family members in thirty countries. There has been litigation on patents covering INLYTA
See drug price trends for INLYTA.
The generic ingredient in INLYTA is axitinib. There are four drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the axitinib profile page.
When can JARDIANCE (empagliflozin) generic drug versions launch?
Generic name: empagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 16, 2027
Generic Entry Controlled by: Canada Patent 2,696,558

This drug has four hundred and twenty-eight patent family members in forty-six countries. There has been litigation on patents covering JARDIANCE
See drug price trends for JARDIANCE.
The generic ingredient in JARDIANCE is empagliflozin. There are twenty-two drug master file entries for this API. Four suppliers are listed for this generic product. Additional details are available on the empagliflozin profile page.
When can KAZANO (alogliptin benzoate; metformin hydrochloride) generic drug versions launch?
Generic name: alogliptin benzoate; metformin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 19, 2027
Generic Entry Controlled by: Canada Patent 2,694,620

This drug has ninety patent family members in forty-one countries. There has been litigation on patents covering KAZANO
See drug price trends for KAZANO.
The generic ingredient in KAZANO is alogliptin benzoate; metformin hydrochloride. There are ten drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate; metformin hydrochloride profile page.
When can LETAIRIS (ambrisentan) generic drug versions launch?
Generic name: ambrisentan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 12, 2026
Generic Entry Controlled by: Canada Patent 2,669,536
LETAIRIS is a drug marketed by Gilead. There are three patents protecting this drug and one Paragraph IV challenge.
This drug has fifty-one patent family members in twenty-six countries. There has been litigation on patents covering LETAIRIS
See drug price trends for LETAIRIS.
The generic ingredient in LETAIRIS is ambrisentan. There are nine drug master file entries for this API. Thirteen suppliers are listed for this generic product. Additional details are available on the ambrisentan profile page.
When can NESINA (alogliptin benzoate) generic drug versions launch?
Generic name: alogliptin benzoate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 01, 2027
Generic Entry Controlled by: Canada Patent 2,677,193

This drug has sixty-nine patent family members in thirty-seven countries. There has been litigation on patents covering NESINA
See drug price trends for NESINA.
The generic ingredient in NESINA is alogliptin benzoate. There are ten drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate profile page.
When can OPSUMIT (macitentan) generic drug versions launch?
Generic name: macitentan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 29, 2026
Generic Entry Controlled by: Canada Patent 2,659,770
OPSUMIT is a drug marketed by Actelion. There are five patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has one hundred and one patent family members in thirty-five countries. There has been litigation on patents covering OPSUMIT
See drug price trends for OPSUMIT.
The generic ingredient in OPSUMIT is macitentan. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the macitentan profile page.
When can OSENI (alogliptin benzoate; pioglitazone hydrochloride) generic drug versions launch?
Generic name: alogliptin benzoate; pioglitazone hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 01, 2027
Generic Entry Controlled by: Canada Patent 2,677,201

This drug has one hundred and one patent family members in forty-two countries. There has been litigation on patents covering OSENI
See drug price trends for OSENI.
The generic ingredient in OSENI is alogliptin benzoate; pioglitazone hydrochloride. There are ten drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the alogliptin benzoate; pioglitazone hydrochloride profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: May 03, 2027
Generic Entry Controlled by: Canada Patent 2,685,831
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,653,344
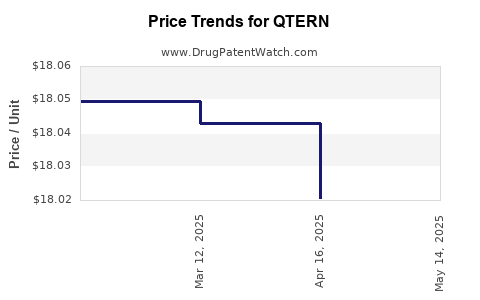
This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,924,318

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can SIRTURO (bedaquiline fumarate) generic drug versions launch?
Generic name: bedaquiline fumarate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 05, 2026
Generic Entry Controlled by: Canada Patent 2,668,512
SIRTURO is a drug marketed by Janssen Therap. There are two patents protecting this drug.
This drug has ninety-seven patent family members in thirty-nine countries.
See drug price trends for SIRTURO.
The generic ingredient in SIRTURO is bedaquiline fumarate. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the bedaquiline fumarate profile page.
When can TRADJENTA (linagliptin) generic drug versions launch?
Generic name: linagliptin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: April 30, 2027
Generic Entry Controlled by: Canada Patent 2,649,922
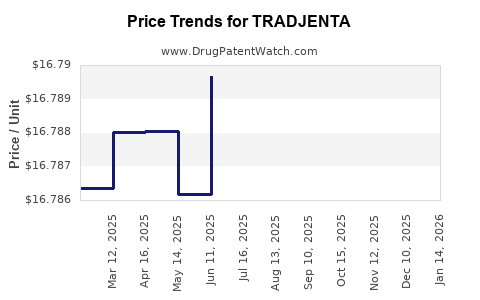
This drug has three hundred and eighty-one patent family members in forty-one countries. There has been litigation on patents covering TRADJENTA
See drug price trends for TRADJENTA.
The generic ingredient in TRADJENTA is linagliptin. There are nineteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the linagliptin profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 15, 2027
Generic Entry Controlled by: Canada Patent 2,655,212
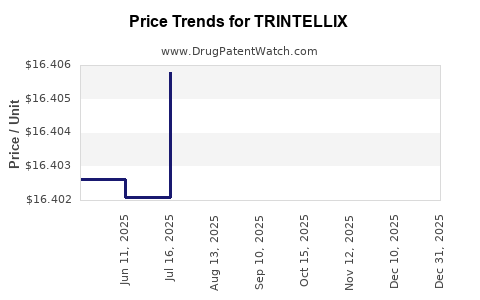
This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 16, 2026
Generic Entry Controlled by: Canada Patent 2,684,571

This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can VARUBI (rolapitant hydrochloride) generic drug versions launch?
Generic name: rolapitant hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 22, 2027
Generic Entry Controlled by: Canada Patent 2,681,465
VARUBI is a drug marketed by Tersera. There are eight patents protecting this drug.
This drug has one hundred and fifty-eight patent family members in thirty-five countries.
The generic ingredient in VARUBI is rolapitant hydrochloride. One supplier is listed for this generic product. Additional details are available on the rolapitant hydrochloride profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: Canada Patent 2,653,012
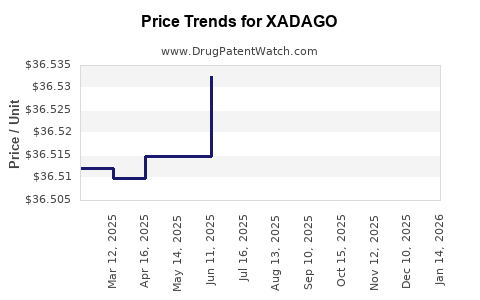
This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can XENLETA (lefamulin acetate) generic drug versions launch?
Generic name: lefamulin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 20, 2027
Generic Entry Controlled by: Canada Patent 2,678,795
XENLETA is a drug marketed by Hong Kong. There are four patents protecting this drug.
This drug has one hundred and twenty patent family members in thirty-six countries. There has been litigation on patents covering XENLETA
See drug price trends for XENLETA.
The generic ingredient in XENLETA is lefamulin acetate. Additional details are available on the lefamulin acetate profile page.
When can BYFAVO (remimazolam besylate) generic drug versions launch?
Generic name: remimazolam besylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 10, 2026
Generic Entry Controlled by: Canada Patent 2,657,347
BYFAVO is a drug marketed by Acacia. There are eleven patents protecting this drug.
This drug has fifty-three patent family members in twenty-four countries. There has been litigation on patents covering BYFAVO
See drug price trends for BYFAVO.
The generic ingredient in BYFAVO is remimazolam besylate. One supplier is listed for this generic product. Additional details are available on the remimazolam besylate profile page.
When can TRELSTAR (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 06, 2027
Generic Entry Controlled by: Canada Patent 2,688,478
TRELSTAR is a drug marketed by Verity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
See drug price trends for TRELSTAR.
The generic ingredient in TRELSTAR is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can TRIFERIC AVNU (ferric pyrophosphate citrate) generic drug versions launch?
Generic name: ferric pyrophosphate citrate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 20, 2027
Generic Entry Controlled by: Canada Patent 2,638,081
TRIFERIC AVNU is a drug marketed by Rockwell Medical Inc. There is one patent protecting this drug.
This drug has twelve patent family members in eleven countries.
See drug price trends for TRIFERIC AVNU.
The generic ingredient in TRIFERIC AVNU is ferric pyrophosphate citrate. There are twenty drug master file entries for this API. Additional details are available on the ferric pyrophosphate citrate profile page.
When can TWIRLA (ethinyl estradiol; levonorgestrel) generic drug versions launch?
Generic name: ethinyl estradiol; levonorgestrel
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 10, 2027
Generic Entry Controlled by: Canada Patent 2,692,884
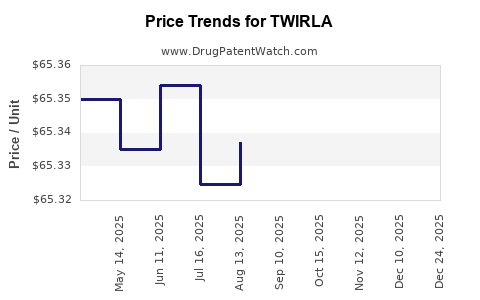
This drug has nineteen patent family members in twelve countries.
See drug price trends for TWIRLA.
The generic ingredient in TWIRLA is ethinyl estradiol; levonorgestrel. There are twenty-six drug master file entries for this API. Twenty-three suppliers are listed for this generic product. Additional details are available on the ethinyl estradiol; levonorgestrel profile page.
When can WINLEVI (clascoterone) generic drug versions launch?
Generic name: clascoterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 03, 2027
Generic Entry Controlled by: Canada Patent 2,691,445

This drug has ninety-three patent family members in twenty-nine countries. There has been litigation on patents covering WINLEVI
See drug price trends for WINLEVI.
The generic ingredient in WINLEVI is clascoterone. One supplier is listed for this generic product. Additional details are available on the clascoterone profile page.
When can WINLEVI (clascoterone) generic drug versions launch?
Generic name: clascoterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 03, 2027
Generic Entry Controlled by: Canada Patent 2,871,039

This drug has ninety-three patent family members in twenty-nine countries. There has been litigation on patents covering WINLEVI
See drug price trends for WINLEVI.
The generic ingredient in WINLEVI is clascoterone. One supplier is listed for this generic product. Additional details are available on the clascoterone profile page.
When can TEPMETKO (tepotinib hydrochloride) generic drug versions launch?
Generic name: tepotinib hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 12, 2027
Generic Entry Controlled by: Canada Patent 2,692,867
TEPMETKO is a drug marketed by Emd Serono Inc. There are eight patents protecting this drug.
This drug has seventy-nine patent family members in thirty-six countries. There has been litigation on patents covering TEPMETKO
See drug price trends for TEPMETKO.
The generic ingredient in TEPMETKO is tepotinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the tepotinib hydrochloride profile page.
When can TEPMETKO (tepotinib hydrochloride) generic drug versions launch?
Generic name: tepotinib hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 12, 2027
Generic Entry Controlled by: Canada Patent 2,693,600
TEPMETKO is a drug marketed by Emd Serono Inc. There are eight patents protecting this drug.
This drug has seventy-nine patent family members in thirty-six countries. There has been litigation on patents covering TEPMETKO
See drug price trends for TEPMETKO.
The generic ingredient in TEPMETKO is tepotinib hydrochloride. One supplier is listed for this generic product. Additional details are available on the tepotinib hydrochloride profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 10, 2026
Generic Entry Controlled by: Canada Patent 2,667,155
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 10, 2026
Generic Entry Controlled by: Canada Patent 2,667,460
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 10, 2026
Generic Entry Controlled by: Canada Patent 2,898,514
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,985,797

This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,985,797

This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 12, 2026
Generic Entry Controlled by: Canada Patent 2,650,263
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 13, 2026
Generic Entry Controlled by: Canada Patent 2,656,456
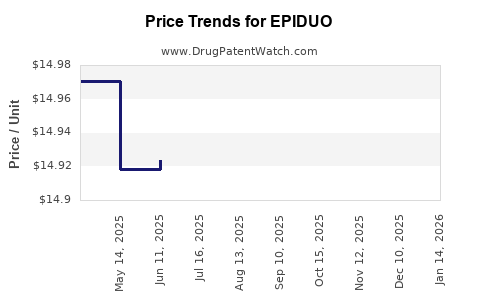
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: Canada Patent 3,105,972

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can PREPOPIK (citric acid; magnesium oxide; sodium picosulfate) generic drug versions launch?
Generic name: citric acid; magnesium oxide; sodium picosulfate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 12, 2027
Generic Entry Controlled by: Canada Patent 2,702,152
PREPOPIK is a drug marketed by Ferring Pharms Inc. There are two patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering PREPOPIK
See drug price trends for PREPOPIK.
The generic ingredient in PREPOPIK is citric acid; magnesium oxide; sodium picosulfate. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the citric acid; magnesium oxide; sodium picosulfate profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 13, 2026
Generic Entry Controlled by: Canada Patent 2,656,456
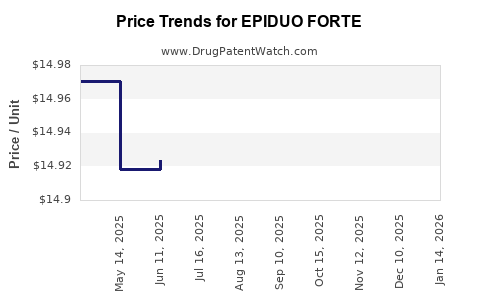
See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can EXFORGE HCT (amlodipine besylate; hydrochlorothiazide; valsartan) generic drug versions launch?
Generic name: amlodipine besylate; hydrochlorothiazide; valsartan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 27, 2026
Generic Entry Controlled by: Canada Patent 2,654,986

See drug price trends for EXFORGE HCT.
The generic ingredient in EXFORGE HCT is amlodipine besylate; hydrochlorothiazide; valsartan. There are fifty drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the amlodipine besylate; hydrochlorothiazide; valsartan profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: Canada Patent 2,985,797

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can TEKTURNA HCT (aliskiren hemifumarate; hydrochlorothiazide) generic drug versions launch?
Generic name: aliskiren hemifumarate; hydrochlorothiazide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 23, 2026
Generic Entry Controlled by: Canada Patent 2,654,872

This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering TEKTURNA HCT
See drug price trends for TEKTURNA HCT.
The generic ingredient in TEKTURNA HCT is aliskiren hemifumarate; hydrochlorothiazide. There are four drug master file entries for this API. Additional details are available on the aliskiren hemifumarate; hydrochlorothiazide profile page.
When can RASUVO (methotrexate) generic drug versions launch?
Generic name: methotrexate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 21, 2026
Generic Entry Controlled by: Canada Patent 2,869,671

This drug has twenty-nine patent family members in twenty-one countries. There has been litigation on patents covering RASUVO
See drug price trends for RASUVO.
The generic ingredient in RASUVO is methotrexate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the methotrexate profile page.
When can XERMELO (telotristat etiprate) generic drug versions launch?
Generic name: telotristat etiprate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 12, 2026
Generic Entry Controlled by: Canada Patent 2,672,233
XERMELO is a drug marketed by Tersera. There are five patents protecting this drug.
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for XERMELO.
The generic ingredient in XERMELO is telotristat etiprate. One supplier is listed for this generic product. Additional details are available on the telotristat etiprate profile page.
When can LOCAMETZ (gallium ga-68 gozetotide) generic drug versions launch?
Generic name: gallium ga-68 gozetotide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 17, 2027
Generic Entry Controlled by: Canada Patent 2,696,627
LOCAMETZ is a drug marketed by Novartis. There are two patents protecting this drug.
This drug has fifty-five patent family members in sixteen countries.
See drug price trends for LOCAMETZ.
The generic ingredient in LOCAMETZ is gallium ga-68 gozetotide. There are sixteen drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the gallium ga-68 gozetotide profile page.
When can ENTRESTO SPRINKLE (sacubitril; valsartan) generic drug versions launch?
Generic name: sacubitril; valsartan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 08, 2026
Generic Entry Controlled by: Canada Patent 2,590,511
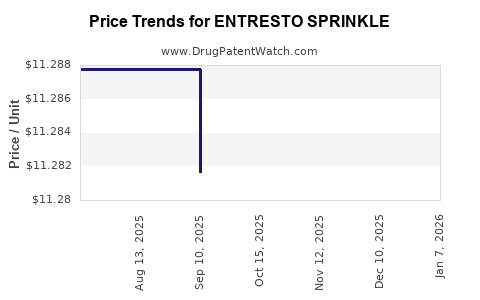
This drug has one hundred and eight patent family members in thirty-nine countries. There has been litigation on patents covering ENTRESTO SPRINKLE
See drug price trends for ENTRESTO SPRINKLE.
The generic ingredient in ENTRESTO SPRINKLE is sacubitril; valsartan. There are eleven drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the sacubitril; valsartan profile page.
When can ZUNVEYL (benzgalantamine gluconate) generic drug versions launch?
Generic name: benzgalantamine gluconate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 22, 2026
Generic Entry Controlled by: Canada Patent 2,623,114
ZUNVEYL is a drug marketed by Alpha Cognition. There are four patents protecting this drug.
This drug has twenty-six patent family members in seventeen countries. There has been litigation on patents covering ZUNVEYL
The generic ingredient in ZUNVEYL is benzgalantamine gluconate. One supplier is listed for this generic product. Additional details are available on the benzgalantamine gluconate profile page.
When can TRIPTODUR KIT (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 06, 2027
Generic Entry Controlled by: Canada Patent 2,688,478
TRIPTODUR KIT is a drug marketed by Azurity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
The generic ingredient in TRIPTODUR KIT is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can FINACEA (azelaic acid) generic drug versions launch?
Generic name: azelaic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 07, 2026
Generic Entry Controlled by: Canada Patent 2,611,577

This drug has one hundred and thirty-seven patent family members in twenty countries. There has been litigation on patents covering FINACEA
See drug price trends for FINACEA .
The generic ingredient in FINACEA is azelaic acid. There are eight drug master file entries for this API. Eight suppliers are listed for this generic product. Additional details are available on the azelaic acid profile page.
When can REVLIMID (lenalidomide) generic drug versions launch?
Generic name: lenalidomide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 03, 2026
Generic Entry Controlled by: Canada Patent 2,570,755
REVLIMID is a drug marketed by Bristol Myers Squibb. There are two patents protecting this drug and three Paragraph IV challenges.
This drug has three hundred and thirty-one patent family members in forty-one countries. There has been litigation on patents covering REVLIMID
See drug price trends for REVLIMID.
The generic ingredient in REVLIMID is lenalidomide. There are fourteen drug master file entries for this API. Nineteen suppliers are listed for this generic product. Additional details are available on the lenalidomide profile page.
When can REVLIMID (lenalidomide) generic drug versions launch?
Generic name: lenalidomide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 03, 2026
Generic Entry Controlled by: Canada Patent 2,972,299
REVLIMID is a drug marketed by Bristol Myers Squibb. There are two patents protecting this drug and three Paragraph IV challenges.
This drug has three hundred and thirty-one patent family members in forty-one countries. There has been litigation on patents covering REVLIMID
See drug price trends for REVLIMID.
The generic ingredient in REVLIMID is lenalidomide. There are fourteen drug master file entries for this API. Nineteen suppliers are listed for this generic product. Additional details are available on the lenalidomide profile page.
When can XTANDI (enzalutamide) generic drug versions launch?
Generic name: enzalutamide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 29, 2027
Generic Entry Controlled by: Canada Patent 2,648,139
XTANDI is a drug marketed by Astellas. There are seven patents protecting this drug and one Paragraph IV challenge. Three tentatively approved generics are ready to enter the market.
This drug has one hundred and ninety-two patent family members in thirty-six countries. There has been litigation on patents covering XTANDI
See drug price trends for XTANDI.
The generic ingredient in XTANDI is enzalutamide. There are nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the enzalutamide profile page.
When can EUCRISA (crisaborole) generic drug versions launch?
Generic name: crisaborole
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 16, 2026
Generic Entry Controlled by: Canada Patent 2,635,680
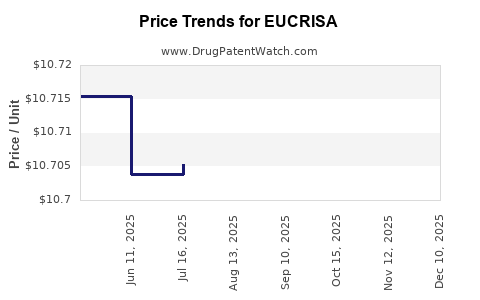
This drug has one hundred and forty-eight patent family members in twenty-eight countries. There has been litigation on patents covering EUCRISA
See drug price trends for EUCRISA.
The generic ingredient in EUCRISA is crisaborole. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the crisaborole profile page.
When can OMONTYS (peginesatide acetate) generic drug versions launch?
Generic name: peginesatide acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 05, 2026
Generic Entry Controlled by: Canada Patent 2,609,401
OMONTYS is a drug marketed by Takeda Pharms Usa. There are two patents protecting this drug.
This drug has twenty-seven patent family members in eighteen countries.
The generic ingredient in OMONTYS is peginesatide acetate. Additional details are available on the peginesatide acetate profile page.
When can KERYDIN (tavaborole) generic drug versions launch?
Generic name: tavaborole
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 16, 2026
Generic Entry Controlled by: Canada Patent 2,635,680

See drug price trends for KERYDIN.
The generic ingredient in KERYDIN is tavaborole. There are six drug master file entries for this API. Seven suppliers are listed for this generic product. Additional details are available on the tavaborole profile page.
When can APLENZIN (bupropion hydrobromide) generic drug versions launch?
Generic name: bupropion hydrobromide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 27, 2026
Generic Entry Controlled by: Canada Patent 2,578,626
APLENZIN is a drug marketed by Bausch. There are eight patents protecting this drug and three Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has fifty-two patent family members in eighteen countries. There has been litigation on patents covering APLENZIN
See drug price trends for APLENZIN.
The generic ingredient in APLENZIN is bupropion hydrobromide. There are thirty-eight drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the bupropion hydrobromide profile page.
When can APLENZIN (bupropion hydrobromide) generic drug versions launch?
Generic name: bupropion hydrobromide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 26, 2027
Generic Entry Controlled by: Canada Patent 2,655,596
APLENZIN is a drug marketed by Bausch. There are eight patents protecting this drug and three Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has fifty-two patent family members in eighteen countries. There has been litigation on patents covering APLENZIN
See drug price trends for APLENZIN.
The generic ingredient in APLENZIN is bupropion hydrobromide. There are thirty-eight drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the bupropion hydrobromide profile page.