World Intellectual Property Organization (WIPO): These 53 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
DrugPatentWatch® Estimated Loss of Exclusivity Dates in World Intellectual Property Organization (WIPO)
Generic Entry Dates in Other Countries
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
Summary: World Intellectual Property Organization (WIPO): These 53 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
| Tradename | Ingredient | Estimated Entry Opportunity Date |
|---|---|---|
| ESBRIET | pirfenidone | 2026-09-22 |
| FARYDAK | panobinostat lactate | 2026-06-12 |
| EPIDUO | adapalene; benzoyl peroxide | 2026-07-13 |
| QVAR REDIHALER | beclomethasone dipropionate | 2027-04-11 |
| OLYSIO | simeprevir sodium | 2026-07-28 |
| TWIRLA | ethinyl estradiol; levonorgestrel | 2027-07-10 |
| >Tradename | >Ingredient | >Estimated Entry Opportunity Date |
Details: World Intellectual Property Organization (WIPO): These 53 Drugs Face Patent Expirations and Generic Entry in 2026 - 2027
When can ESBRIET (pirfenidone) generic drug versions launch?
Generic name: pirfenidone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 22, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,038,315
ESBRIET is a drug marketed by Legacy Pharma. There are twenty patents protecting this drug and two Paragraph IV challenges. One tentatively approved generic is ready to enter the market.
This drug has two hundred and sixty-six patent family members in forty-six countries. There has been litigation on patents covering ESBRIET
See drug price trends for ESBRIET.
The generic ingredient in ESBRIET is pirfenidone. There are twenty-three drug master file entries for this API. Twenty-four suppliers are listed for this generic product. Additional details are available on the pirfenidone profile page.
When can FARYDAK (panobinostat lactate) generic drug versions launch?
Generic name: panobinostat lactate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 12, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,146,716
FARYDAK is a drug marketed by Secura. There are two patents protecting this drug.
This drug has sixty-eight patent family members in forty countries. There has been litigation on patents covering FARYDAK
See drug price trends for FARYDAK.
The generic ingredient in FARYDAK is panobinostat lactate. There is one drug master file entry for this API. Additional details are available on the panobinostat lactate profile page.
When can EPIDUO (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 13, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,006,888
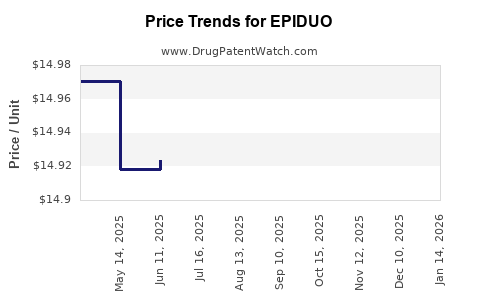
This drug has thirty-five patent family members in twenty-two countries. There has been litigation on patents covering EPIDUO
See drug price trends for EPIDUO.
The generic ingredient in EPIDUO is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can QVAR REDIHALER (beclomethasone dipropionate) generic drug versions launch?
Generic name: beclomethasone dipropionate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: April 11, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,119,552
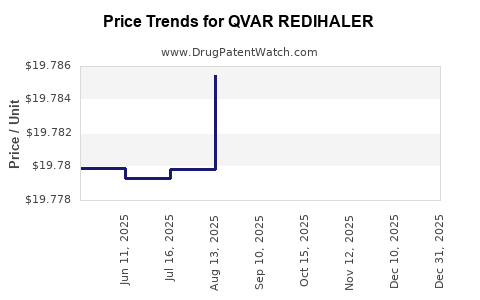
This drug has two hundred and seventy-eight patent family members in twenty-seven countries. There has been litigation on patents covering QVAR REDIHALER
See drug price trends for QVAR REDIHALER.
The generic ingredient in QVAR REDIHALER is beclomethasone dipropionate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the beclomethasone dipropionate profile page.
When can OLYSIO (simeprevir sodium) generic drug versions launch?
Generic name: simeprevir sodium
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 28, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,014,926
OLYSIO is a drug marketed by Janssen Prods. There are eight patents protecting this drug.
This drug has sixty-four patent family members in forty-one countries.
See drug price trends for OLYSIO.
The generic ingredient in OLYSIO is simeprevir sodium. There is one drug master file entry for this API. Additional details are available on the simeprevir sodium profile page.
When can TWIRLA (ethinyl estradiol; levonorgestrel) generic drug versions launch?
Generic name: ethinyl estradiol; levonorgestrel
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 10, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,009,649
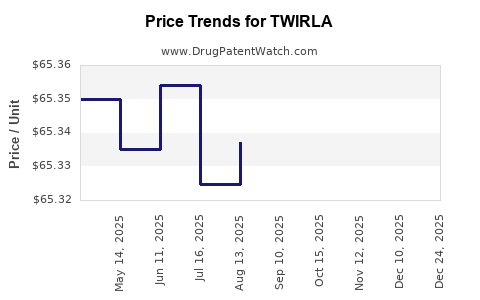
This drug has nineteen patent family members in twelve countries.
See drug price trends for TWIRLA.
The generic ingredient in TWIRLA is ethinyl estradiol; levonorgestrel. There are twenty-six drug master file entries for this API. Twenty-three suppliers are listed for this generic product. Additional details are available on the ethinyl estradiol; levonorgestrel profile page.
When can TWIRLA (ethinyl estradiol; levonorgestrel) generic drug versions launch?
Generic name: ethinyl estradiol; levonorgestrel
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 10, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,011,088,072

This drug has nineteen patent family members in twelve countries.
See drug price trends for TWIRLA.
The generic ingredient in TWIRLA is ethinyl estradiol; levonorgestrel. There are twenty-six drug master file entries for this API. Twenty-three suppliers are listed for this generic product. Additional details are available on the ethinyl estradiol; levonorgestrel profile page.
When can PICATO (ingenol mebutate) generic drug versions launch?
Generic name: ingenol mebutate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 18, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,068,963

This drug has thirty-five patent family members in twenty-one countries. There has been litigation on patents covering PICATO
See drug price trends for PICATO.
The generic ingredient in PICATO is ingenol mebutate. There are three drug master file entries for this API. Additional details are available on the ingenol mebutate profile page.
When can RYTARY (carbidopa; levodopa) generic drug versions launch?
Generic name: carbidopa; levodopa
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 28, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,085,306
RYTARY is a drug marketed by Impax. There are eight patents protecting this drug and two Paragraph IV challenges.
This drug has twenty-five patent family members in twelve countries. There has been litigation on patents covering RYTARY
See drug price trends for RYTARY.
The generic ingredient in RYTARY is carbidopa; levodopa. There are eighteen drug master file entries for this API. Thirty-eight suppliers are listed for this generic product. Additional details are available on the carbidopa; levodopa profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,128,933

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,128,934

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can LETAIRIS (ambrisentan) generic drug versions launch?
Generic name: ambrisentan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 12, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,073,928
LETAIRIS is a drug marketed by Gilead. There are three patents protecting this drug and one Paragraph IV challenge.
This drug has fifty-one patent family members in twenty-six countries. There has been litigation on patents covering LETAIRIS
See drug price trends for LETAIRIS.
The generic ingredient in LETAIRIS is ambrisentan. There are nine drug master file entries for this API. Thirteen suppliers are listed for this generic product. Additional details are available on the ambrisentan profile page.
When can ZEMDRI (plazomicin sulfate) generic drug versions launch?
Generic name: plazomicin sulfate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 21, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,067,692
ZEMDRI is a drug marketed by Cipla Usa. There are four patents protecting this drug.
This drug has twenty-eight patent family members in twenty-one countries. There has been litigation on patents covering ZEMDRI
See drug price trends for ZEMDRI.
The generic ingredient in ZEMDRI is plazomicin sulfate. Three suppliers are listed for this generic product. Additional details are available on the plazomicin sulfate profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 15, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,033,523
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can VANTRELA ER (hydrocodone bitartrate) generic drug versions launch?
Generic name: hydrocodone bitartrate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 15, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,035,474
VANTRELA ER is a drug marketed by Teva Branded Pharm. There are three patents protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has thirty-three patent family members in thirteen countries. There has been litigation on patents covering VANTRELA ER
The generic ingredient in VANTRELA ER is hydrocodone bitartrate. There are twenty-three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the hydrocodone bitartrate profile page.
When can BRILINTA (ticagrelor) generic drug versions launch?
Generic name: ticagrelor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 21, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,024,045
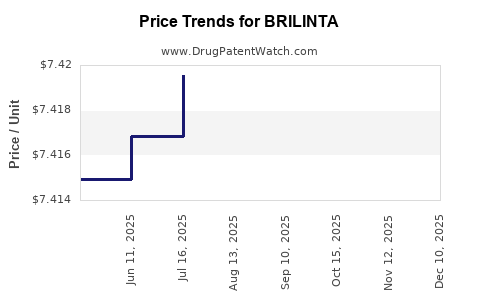
This drug has sixty-two patent family members in thirty-five countries. There has been litigation on patents covering BRILINTA
See drug price trends for BRILINTA.
The generic ingredient in BRILINTA is ticagrelor. There are twenty-one drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ticagrelor profile page.
When can PREPOPIK (citric acid; magnesium oxide; sodium picosulfate) generic drug versions launch?
Generic name: citric acid; magnesium oxide; sodium picosulfate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 12, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,047,633
PREPOPIK is a drug marketed by Ferring Pharms Inc. There are two patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has thirty-two patent family members in twenty-five countries. There has been litigation on patents covering PREPOPIK
See drug price trends for PREPOPIK.
The generic ingredient in PREPOPIK is citric acid; magnesium oxide; sodium picosulfate. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the citric acid; magnesium oxide; sodium picosulfate profile page.
When can EPIDUO FORTE (adapalene; benzoyl peroxide) generic drug versions launch?
Generic name: adapalene; benzoyl peroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 13, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,006,888
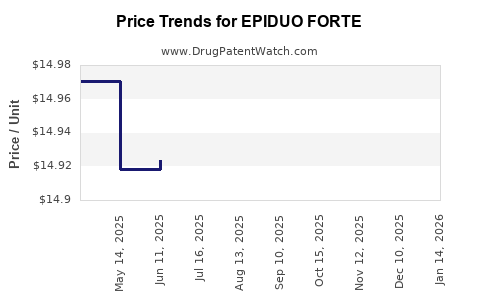
See drug price trends for EPIDUO FORTE.
The generic ingredient in EPIDUO FORTE is adapalene; benzoyl peroxide. There are twelve drug master file entries for this API. Twelve suppliers are listed for this generic product. Additional details are available on the adapalene; benzoyl peroxide profile page.
When can CRESEMBA (isavuconazonium sulfate) generic drug versions launch?
Generic name: isavuconazonium sulfate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 25, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,037,359
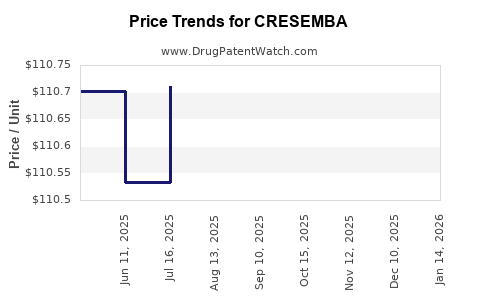
This drug has thirty-two patent family members in nineteen countries. There has been litigation on patents covering CRESEMBA
See drug price trends for CRESEMBA.
The generic ingredient in CRESEMBA is isavuconazonium sulfate. One supplier is listed for this generic product. Additional details are available on the isavuconazonium sulfate profile page.
When can INTRAROSA (prasterone) generic drug versions launch?
Generic name: prasterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 10, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,021,323
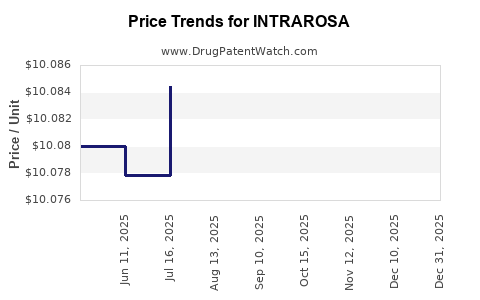
This drug has fifty-nine patent family members in thirty-two countries.
See drug price trends for INTRAROSA.
The generic ingredient in INTRAROSA is prasterone. There are seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the prasterone profile page.
When can MYRBETRIQ (mirabegron) generic drug versions launch?
Generic name: mirabegron
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 02, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,057,685
MYRBETRIQ is a drug marketed by Apgdi. There are six patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has fifty-three patent family members in twenty-five countries. There has been litigation on patents covering MYRBETRIQ
See drug price trends for MYRBETRIQ.
The generic ingredient in MYRBETRIQ is mirabegron. There are nineteen drug master file entries for this API. Eight suppliers are listed for this generic product. Additional details are available on the mirabegron profile page.
When can WINLEVI (clascoterone) generic drug versions launch?
Generic name: clascoterone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 03, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,019,138

This drug has ninety-three patent family members in twenty-nine countries. There has been litigation on patents covering WINLEVI
See drug price trends for WINLEVI.
The generic ingredient in WINLEVI is clascoterone. One supplier is listed for this generic product. Additional details are available on the clascoterone profile page.
When can SALONPAS (menthol; methyl salicylate) generic drug versions launch?
Generic name: menthol; methyl salicylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 04, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,016,077
SALONPAS is a drug marketed by Hisamitsu Pharm Co. There are two patents protecting this drug.
This drug has twenty patent family members in sixteen countries.
The generic ingredient in SALONPAS is menthol; methyl salicylate. There are eighteen drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the menthol; methyl salicylate profile page.
When can PROMACTA (eltrombopag olamine) generic drug versions launch?
Generic name: eltrombopag olamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: May 03, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,136,843
PROMACTA is a drug marketed by Novartis. There are six patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and thirty-five patent family members in forty-one countries. There has been litigation on patents covering PROMACTA
See drug price trends for PROMACTA.
The generic ingredient in PROMACTA is eltrombopag olamine. There are three drug master file entries for this API. Eleven suppliers are listed for this generic product. Additional details are available on the eltrombopag olamine profile page.
When can EDARBI (azilsartan kamedoxomil) generic drug versions launch?
Generic name: azilsartan kamedoxomil
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 28, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,123,536
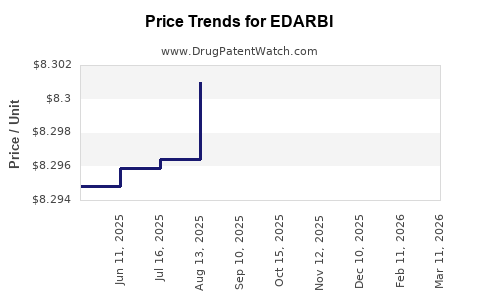
This drug has twenty-one patent family members in seventeen countries. There has been litigation on patents covering EDARBI
See drug price trends for EDARBI.
The generic ingredient in EDARBI is azilsartan kamedoxomil. There are six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the azilsartan kamedoxomil profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 15, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,020,259
CREON is a drug marketed by
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can CREON (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 15, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,020,260
CREON is a drug marketed by
See drug price trends for CREON.
The generic ingredient in CREON is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can SIGNIFOR LAR (pasireotide pamoate) generic drug versions launch?
Generic name: pasireotide pamoate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: May 24, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,142,153
SIGNIFOR LAR is a drug marketed by Recordati Rare. There are three patents protecting this drug.
See drug price trends for SIGNIFOR LAR.
The generic ingredient in SIGNIFOR LAR is pasireotide pamoate. One supplier is listed for this generic product. Additional details are available on the pasireotide pamoate profile page.
When can VELPHORO (sucroferric oxyhydroxide) generic drug versions launch?
Generic name: sucroferric oxyhydroxide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 16, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,062,993
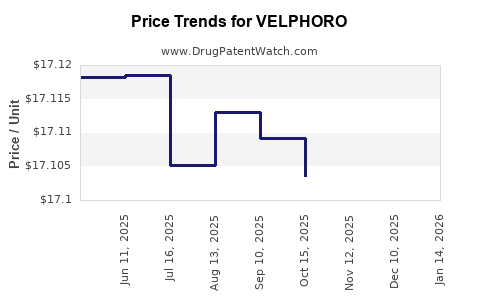
This drug has one hundred and thirty-five patent family members in thirty-six countries. There has been litigation on patents covering VELPHORO
See drug price trends for VELPHORO.
The generic ingredient in VELPHORO is sucroferric oxyhydroxide. There are four drug master file entries for this API. Additional details are available on the sucroferric oxyhydroxide profile page.
When can ZTALMY (ganaxolone) generic drug versions launch?
Generic name: ganaxolone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 28, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,062,266
ZTALMY is a drug marketed by Immedica Pharma. There are eleven patents protecting this drug.
This drug has forty-nine patent family members in sixteen countries. There has been litigation on patents covering ZTALMY
See drug price trends for ZTALMY.
The generic ingredient in ZTALMY is ganaxolone. One supplier is listed for this generic product. Additional details are available on the ganaxolone profile page.
When can ZUNVEYL (benzgalantamine gluconate) generic drug versions launch?
Generic name: benzgalantamine gluconate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 22, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,039,138
ZUNVEYL is a drug marketed by Alpha Cognition. There are four patents protecting this drug.
This drug has twenty-six patent family members in seventeen countries. There has been litigation on patents covering ZUNVEYL
The generic ingredient in ZUNVEYL is benzgalantamine gluconate. One supplier is listed for this generic product. Additional details are available on the benzgalantamine gluconate profile page.
When can VYKAT XR (diazoxide choline) generic drug versions launch?
Generic name: diazoxide choline
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 20, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,081,521
VYKAT XR is a drug marketed by Soleno Therap. There are six patents protecting this drug.
This drug has seventy-eight patent family members in twenty-two countries.
The generic ingredient in VYKAT XR is diazoxide choline. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the diazoxide choline profile page.
When can KYBELLA (deoxycholic acid) generic drug versions launch?
Generic name: deoxycholic acid
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,157,635
KYBELLA is a drug marketed by Abbvie. There are fourteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and forty-six patent family members in thirty-eight countries. There has been litigation on patents covering KYBELLA
The generic ingredient in KYBELLA is deoxycholic acid. There are thirty-three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the deoxycholic acid profile page.
When can KORSUVA (difelikefalin acetate) generic drug versions launch?
Generic name: difelikefalin acetate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 10, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,057,608
KORSUVA is a drug marketed by Vifor Intl. There are twelve patents protecting this drug.
This drug has fifty-three patent family members in twenty-seven countries. There has been litigation on patents covering KORSUVA
See drug price trends for KORSUVA.
The generic ingredient in KORSUVA is difelikefalin acetate. One supplier is listed for this generic product. Additional details are available on the difelikefalin acetate profile page.
When can TRINTELLIX (vortioxetine hydrobromide) generic drug versions launch?
Generic name: vortioxetine hydrobromide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 16, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,113,359
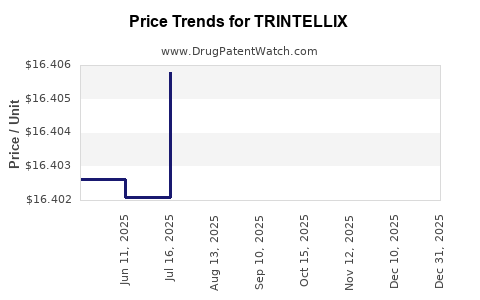
This drug has two hundred and seventeen patent family members in forty-two countries. There has been litigation on patents covering TRINTELLIX
See drug price trends for TRINTELLIX.
The generic ingredient in TRINTELLIX is vortioxetine hydrobromide. There are sixteen drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the vortioxetine hydrobromide profile page.
When can TYMLOS (abaloparatide) generic drug versions launch?
Generic name: abaloparatide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 03, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,063,279

This drug has fifty-six patent family members in twenty-nine countries. There has been litigation on patents covering TYMLOS
See drug price trends for TYMLOS.
The generic ingredient in TYMLOS is abaloparatide. One supplier is listed for this generic product. Additional details are available on the abaloparatide profile page.
When can TYMLOS (abaloparatide) generic drug versions launch?
Generic name: abaloparatide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 03, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,137,093

This drug has fifty-six patent family members in twenty-nine countries. There has been litigation on patents covering TYMLOS
See drug price trends for TYMLOS.
The generic ingredient in TYMLOS is abaloparatide. One supplier is listed for this generic product. Additional details are available on the abaloparatide profile page.
When can TRIPTODUR KIT (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 06, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,149,320
TRIPTODUR KIT is a drug marketed by Azurity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
The generic ingredient in TRIPTODUR KIT is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can TRELSTAR (triptorelin pamoate) generic drug versions launch?
Generic name: triptorelin pamoate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 06, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,149,320
TRELSTAR is a drug marketed by Verity. There is one patent protecting this drug.
This drug has forty-seven patent family members in thirty-two countries.
See drug price trends for TRELSTAR.
The generic ingredient in TRELSTAR is triptorelin pamoate. There are three drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the triptorelin pamoate profile page.
When can LIVDELZI (seladelpar lysine) generic drug versions launch?
Generic name: seladelpar lysine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: September 13, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,033,231
LIVDELZI is a drug marketed by Gilead Sciences Inc. There are six patents protecting this drug.
This drug has one hundred and thirty-eight patent family members in forty-six countries.
The generic ingredient in LIVDELZI is seladelpar lysine. One supplier is listed for this generic product. Additional details are available on the seladelpar lysine profile page.
When can SOFDRA (sofpironium bromide) generic drug versions launch?
Generic name: sofpironium bromide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 13, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,058,971
SOFDRA is a drug marketed by Botanix Sb. There are twenty patents protecting this drug.
This drug has one hundred and sixty patent family members in twenty-six countries. There has been litigation on patents covering SOFDRA
The generic ingredient in SOFDRA is sofpironium bromide. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the sofpironium bromide profile page.
When can SOFDRA (sofpironium bromide) generic drug versions launch?
Generic name: sofpironium bromide
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 13, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,059,021
SOFDRA is a drug marketed by Botanix Sb. There are twenty patents protecting this drug.
This drug has one hundred and sixty patent family members in twenty-six countries. There has been litigation on patents covering SOFDRA
The generic ingredient in SOFDRA is sofpironium bromide. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the sofpironium bromide profile page.
When can CARDAMYST (etripamil) generic drug versions launch?
Generic name: etripamil
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 20, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,156,820
CARDAMYST is a drug marketed by Milestone Pharms Usa. There are six patents protecting this drug.
This drug has sixty-six patent family members in twenty-nine countries.
The generic ingredient in CARDAMYST is etripamil. One supplier is listed for this generic product. Additional details are available on the etripamil profile page.
When can ORKAMBI (ivacaftor; lumacaftor) generic drug versions launch?
Generic name: ivacaftor; lumacaftor
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 07, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,073,757
ORKAMBI is a drug marketed by Vertex Pharms Inc. There are twenty-three patents protecting this drug.
This drug has four hundred and sixty-one patent family members in thirty-six countries. There has been litigation on patents covering ORKAMBI
See drug price trends for ORKAMBI.
The generic ingredient in ORKAMBI is ivacaftor; lumacaftor. There are three drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the ivacaftor; lumacaftor profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 08, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,073,687
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 08, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,032,668
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 08, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,032,703
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can XERMELO (telotristat etiprate) generic drug versions launch?
Generic name: telotristat etiprate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 12, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,073,933
XERMELO is a drug marketed by Tersera. There are five patents protecting this drug.
This drug has seventy patent family members in twenty-nine countries.
See drug price trends for XERMELO.
The generic ingredient in XERMELO is telotristat etiprate. One supplier is listed for this generic product. Additional details are available on the telotristat etiprate profile page.
When can BEVYXXA (betrixaban) generic drug versions launch?
Generic name: betrixaban
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 07, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,056,517
BEVYXXA is a drug marketed by Portola Pharms Inc. There are five patents protecting this drug.
This drug has sixty-seven patent family members in twenty-six countries. There has been litigation on patents covering BEVYXXA
See drug price trends for BEVYXXA.
The generic ingredient in BEVYXXA is betrixaban. Additional details are available on the betrixaban profile page.
When can ZYKADIA (ceritinib) generic drug versions launch?
Generic name: ceritinib
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 08, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,032,694
ZYKADIA is a drug marketed by Novartis. There are eight patents protecting this drug.
This drug has three hundred and twenty-two patent family members in fifty-six countries.
See drug price trends for ZYKADIA.
The generic ingredient in ZYKADIA is ceritinib. There is one drug master file entry for this API. One supplier is listed for this generic product. Additional details are available on the ceritinib profile page.
When can FARXIGA (dapagliflozin) generic drug versions launch?
Generic name: dapagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,002,824
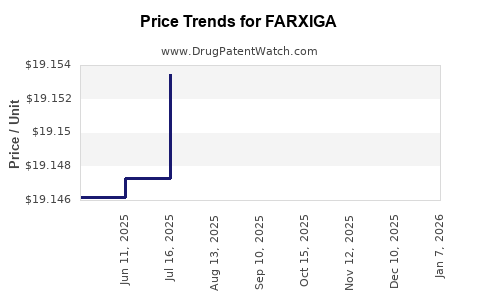
This drug has four hundred and twenty-five patent family members in fifty-two countries. There has been litigation on patents covering FARXIGA
See drug price trends for FARXIGA.
The generic ingredient in FARXIGA is dapagliflozin. There are twenty-six drug master file entries for this API. Twenty-two suppliers are listed for this generic product. Additional details are available on the dapagliflozin profile page.
When can QTERN (dapagliflozin; saxagliptin hydrochloride) generic drug versions launch?
Generic name: dapagliflozin; saxagliptin hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,002,824
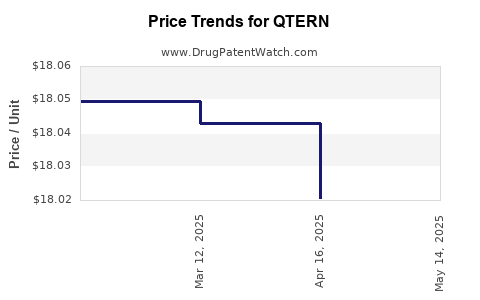
This drug has two hundred and fifty-six patent family members in forty-eight countries. There has been litigation on patents covering QTERN
See drug price trends for QTERN.
The generic ingredient in QTERN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
When can XADAGO (safinamide mesylate) generic drug versions launch?
Generic name: safinamide mesylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,147,491
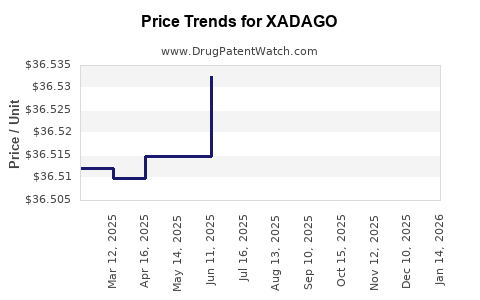
This drug has ninety-seven patent family members in thirty-one countries. There has been litigation on patents covering XADAGO
See drug price trends for XADAGO.
The generic ingredient in XADAGO is safinamide mesylate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the safinamide mesylate profile page.
When can BYSANTI (milsaperidone) generic drug versions launch?
Generic name: milsaperidone
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 29, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,121,899
BYSANTI is a drug marketed by Vanda Pharms Inc. There are nine patents protecting this drug.
This drug has forty-three patent family members in twelve countries. There has been litigation on patents covering BYSANTI
The generic ingredient in BYSANTI is milsaperidone. Additional details are available on the milsaperidone profile page.
When can EMBEDA (morphine sulfate; naltrexone hydrochloride) generic drug versions launch?
Generic name: morphine sulfate; naltrexone hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 19, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,007,149,438
EMBEDA is a drug marketed by Alpharma Pharms. There are seven patents protecting this drug and four Paragraph IV challenges.
This drug has seventy-four patent family members in twenty-three countries.
See drug price trends for EMBEDA.
The generic ingredient in EMBEDA is morphine sulfate; naltrexone hydrochloride. There are twenty-three drug master file entries for this API. Additional details are available on the morphine sulfate; naltrexone hydrochloride profile page.
When can ONPATTRO (patisiran sodium) generic drug versions launch?
Generic name: patisiran sodium
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 27, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,082,817
ONPATTRO is a drug marketed by Alnylam Pharms Inc. There are thirteen patents protecting this drug.
This drug has two hundred and fifty patent family members in thirty-one countries. There has been litigation on patents covering ONPATTRO
See drug price trends for ONPATTRO.
The generic ingredient in ONPATTRO is patisiran sodium. One supplier is listed for this generic product. Additional details are available on the patisiran sodium profile page.
When can ONPATTRO (patisiran sodium) generic drug versions launch?
Generic name: patisiran sodium
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 27, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,127,060
ONPATTRO is a drug marketed by Alnylam Pharms Inc. There are thirteen patents protecting this drug.
This drug has two hundred and fifty patent family members in thirty-one countries. There has been litigation on patents covering ONPATTRO
See drug price trends for ONPATTRO.
The generic ingredient in ONPATTRO is patisiran sodium. One supplier is listed for this generic product. Additional details are available on the patisiran sodium profile page.
When can PANCREAZE (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,102,264
PANCREAZE is a drug marketed by
See drug price trends for PANCREAZE.
The generic ingredient in PANCREAZE is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can ZENPEP (pancrelipase (amylase;lipase;protease)) generic drug versions launch?
Generic name: pancrelipase (amylase;lipase;protease)
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: February 20, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,102,264
ZENPEP is a drug marketed by
See drug price trends for ZENPEP.
The generic ingredient in ZENPEP is pancrelipase (amylase;lipase;protease). There are six drug master file entries for this API. Additional details are available on the pancrelipase (amylase;lipase;protease) profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 07, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,010,921
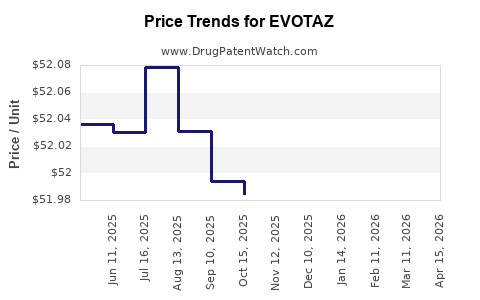
This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 07, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,103,949

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can EVOTAZ (atazanavir sulfate; cobicistat) generic drug versions launch?
Generic name: atazanavir sulfate; cobicistat
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 07, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,008,989

This drug has three hundred and six patent family members in forty-one countries. There has been litigation on patents covering EVOTAZ
See drug price trends for EVOTAZ.
The generic ingredient in EVOTAZ is atazanavir sulfate; cobicistat. There are twenty-five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the atazanavir sulfate; cobicistat profile page.
When can TWIRLA (ethinyl estradiol; levonorgestrel) generic drug versions launch?
Generic name: ethinyl estradiol; levonorgestrel
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 10, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,009,651

This drug has nineteen patent family members in twelve countries.
See drug price trends for TWIRLA.
The generic ingredient in TWIRLA is ethinyl estradiol; levonorgestrel. There are twenty-six drug master file entries for this API. Twenty-three suppliers are listed for this generic product. Additional details are available on the ethinyl estradiol; levonorgestrel profile page.
When can DUZALLO (allopurinol; lesinurad) generic drug versions launch?
Generic name: allopurinol; lesinurad
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 27, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,070,740

This drug has one hundred and forty-four patent family members in thirty-nine countries. There has been litigation on patents covering DUZALLO
See drug price trends for DUZALLO.
The generic ingredient in DUZALLO is allopurinol; lesinurad. There are twenty-two drug master file entries for this API. Additional details are available on the allopurinol; lesinurad profile page.
When can DUZALLO (allopurinol; lesinurad) generic drug versions launch?
Generic name: allopurinol; lesinurad
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 27, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,010,028,189

This drug has one hundred and forty-four patent family members in thirty-nine countries. There has been litigation on patents covering DUZALLO
See drug price trends for DUZALLO.
The generic ingredient in DUZALLO is allopurinol; lesinurad. There are twenty-two drug master file entries for this API. Additional details are available on the allopurinol; lesinurad profile page.
When can DUZALLO (allopurinol; lesinurad) generic drug versions launch?
Generic name: allopurinol; lesinurad
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 27, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,010,028,190

This drug has one hundred and forty-four patent family members in thirty-nine countries. There has been litigation on patents covering DUZALLO
See drug price trends for DUZALLO.
The generic ingredient in DUZALLO is allopurinol; lesinurad. There are twenty-two drug master file entries for this API. Additional details are available on the allopurinol; lesinurad profile page.
When can ZURAMPIC (lesinurad) generic drug versions launch?
Generic name: lesinurad
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 27, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,070,740
ZURAMPIC is a drug marketed by Ironwood Pharms Inc. There are eight patents protecting this drug.
This drug has one hundred and forty-four patent family members in thirty-nine countries. There has been litigation on patents covering ZURAMPIC
See drug price trends for ZURAMPIC.
The generic ingredient in ZURAMPIC is lesinurad. Additional details are available on the lesinurad profile page.
When can ZURAMPIC (lesinurad) generic drug versions launch?
Generic name: lesinurad
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 27, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,010,028,189
ZURAMPIC is a drug marketed by Ironwood Pharms Inc. There are eight patents protecting this drug.
This drug has one hundred and forty-four patent family members in thirty-nine countries. There has been litigation on patents covering ZURAMPIC
See drug price trends for ZURAMPIC.
The generic ingredient in ZURAMPIC is lesinurad. Additional details are available on the lesinurad profile page.
When can ZURAMPIC (lesinurad) generic drug versions launch?
Generic name: lesinurad
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: November 27, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,010,028,190
ZURAMPIC is a drug marketed by Ironwood Pharms Inc. There are eight patents protecting this drug.
This drug has one hundred and forty-four patent family members in thirty-nine countries. There has been litigation on patents covering ZURAMPIC
See drug price trends for ZURAMPIC.
The generic ingredient in ZURAMPIC is lesinurad. Additional details are available on the lesinurad profile page.
When can SEYSARA (sarecycline hydrochloride) generic drug versions launch?
Generic name: sarecycline hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 21, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,079,363
SEYSARA is a drug marketed by Almirall. There are four patents protecting this drug.
This drug has seventy-two patent family members in twenty countries.
See drug price trends for SEYSARA.
The generic ingredient in SEYSARA is sarecycline hydrochloride. One supplier is listed for this generic product. Additional details are available on the sarecycline hydrochloride profile page.
When can OJEMDA (tovorafenib) generic drug versions launch?
Generic name: tovorafenib
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 29, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,006,389
OJEMDA is a drug marketed by Day One Biopharms. There are two patents protecting this drug.
This drug has fifty patent family members in twenty-seven countries.
The generic ingredient in OJEMDA is tovorafenib. One supplier is listed for this generic product. Additional details are available on the tovorafenib profile page.
When can ALIQOPA (copanlisib dihydrochloride) generic drug versions launch?
Generic name: copanlisib dihydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 05, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,070,150
ALIQOPA is a drug marketed by Bayer Healthcare. There are three patents protecting this drug.
This drug has one hundred and five patent family members in forty-eight countries.
See drug price trends for ALIQOPA.
The generic ingredient in ALIQOPA is copanlisib dihydrochloride. Additional details are available on the copanlisib dihydrochloride profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,054,914

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,128,932

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can XIIDRA (lifitegrast) generic drug versions launch?
Generic name: lifitegrast
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: October 19, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,139,817

This drug has one hundred and thirty-five patent family members in twenty-four countries. There has been litigation on patents covering XIIDRA
See drug price trends for XIIDRA.
The generic ingredient in XIIDRA is lifitegrast. Three suppliers are listed for this generic product. Additional details are available on the lifitegrast profile page.
When can DAKLINZA (daclatasvir dihydrochloride) generic drug versions launch?
Generic name: daclatasvir dihydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 11, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,021,927

This drug has ninety-three patent family members in thirty-one countries. There has been litigation on patents covering DAKLINZA
See drug price trends for DAKLINZA.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this API. Additional details are available on the daclatasvir dihydrochloride profile page.
When can VARUBI (rolapitant hydrochloride) generic drug versions launch?
Generic name: rolapitant hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: March 22, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,118,331
VARUBI is a drug marketed by Tersera. There are eight patents protecting this drug.
This drug has one hundred and fifty-eight patent family members in thirty-five countries.
The generic ingredient in VARUBI is rolapitant hydrochloride. One supplier is listed for this generic product. Additional details are available on the rolapitant hydrochloride profile page.
When can EXFORGE HCT (amlodipine besylate; hydrochlorothiazide; valsartan) generic drug versions launch?
Generic name: amlodipine besylate; hydrochlorothiazide; valsartan
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 27, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,002,905

See drug price trends for EXFORGE HCT.
The generic ingredient in EXFORGE HCT is amlodipine besylate; hydrochlorothiazide; valsartan. There are fifty drug master file entries for this API. Five suppliers are listed for this generic product. Additional details are available on the amlodipine besylate; hydrochlorothiazide; valsartan profile page.
When can ZUBSOLV (buprenorphine hydrochloride; naloxone hydrochloride) generic drug versions launch?
Generic name: buprenorphine hydrochloride; naloxone hydrochloride
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 04, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,068,471
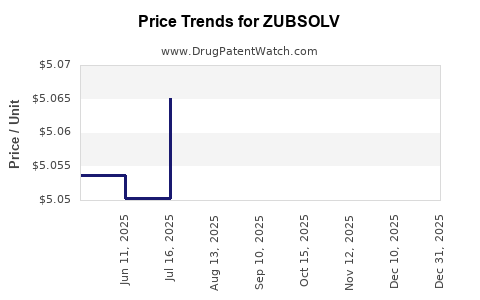
This drug has sixty-two patent family members in thirty-two countries. There has been litigation on patents covering ZUBSOLV
See drug price trends for ZUBSOLV.
The generic ingredient in ZUBSOLV is buprenorphine hydrochloride; naloxone hydrochloride. There are twenty-nine drug master file entries for this API. Twenty-seven suppliers are listed for this generic product. Additional details are available on the buprenorphine hydrochloride; naloxone hydrochloride profile page.
When can BYDUREON (exenatide synthetic) generic drug versions launch?
Generic name: exenatide synthetic
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: June 28, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,002,824

This drug has three hundred and seven patent family members in forty-eight countries. There has been litigation on patents covering BYDUREON
See drug price trends for BYDUREON.
The generic ingredient in BYDUREON is exenatide synthetic. There are seven drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the exenatide synthetic profile page.
When can ORENITRAM (treprostinil diolamine) generic drug versions launch?
Generic name: treprostinil diolamine
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: December 17, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,078,965
ORENITRAM is a drug marketed by United Therap. There are nine patents protecting this drug and two Paragraph IV challenges.
This drug has fifty-nine patent family members in eight countries. There has been litigation on patents covering ORENITRAM
See drug price trends for ORENITRAM.
The generic ingredient in ORENITRAM is treprostinil diolamine. There are nineteen drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the treprostinil diolamine profile page.
When can BYFAVO (remimazolam besylate) generic drug versions launch?
Generic name: remimazolam besylate
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: July 10, 2026
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,008,007,071
BYFAVO is a drug marketed by Acacia. There are eleven patents protecting this drug.
This drug has fifty-three patent family members in twenty-four countries. There has been litigation on patents covering BYFAVO
See drug price trends for BYFAVO.
The generic ingredient in BYFAVO is remimazolam besylate. One supplier is listed for this generic product. Additional details are available on the remimazolam besylate profile page.
When can JARDIANCE (empagliflozin) generic drug versions launch?
Generic name: empagliflozin
DrugPatentWatch® Estimated Patent Expiration / Generic Entry Opportunity Date: August 16, 2027
Generic Entry Controlled by: World Intellectual Property Organization (WIPO) Patent 2,009,022,007