PREPOPIK Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Prepopik, and when can generic versions of Prepopik launch?
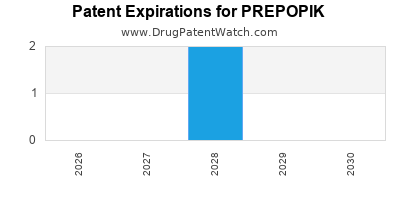
Prepopik is a drug marketed by Ferring Pharms Inc and is included in one NDA. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has thirty-two patent family members in twenty-five countries.
The generic ingredient in PREPOPIK is citric acid; magnesium oxide; sodium picosulfate. There are five drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the citric acid; magnesium oxide; sodium picosulfate profile page.
DrugPatentWatch® Generic Entry Outlook for Prepopik
There have been seven patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There is one tentative approval for the generic drug (citric acid; magnesium oxide; sodium picosulfate), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for PREPOPIK?
- What are the global sales for PREPOPIK?
- What is Average Wholesale Price for PREPOPIK?
Summary for PREPOPIK
| International Patents: | 32 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 5 |
| Drug Prices: | Drug price information for PREPOPIK |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for PREPOPIK |
| DailyMed Link: | PREPOPIK at DailyMed |
Recent Clinical Trials for PREPOPIK
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Ferring Pharmaceuticals | Phase 4 |
| The Cleveland Clinic | Phase 4 |
| Ferring Pharmaceuticals | Phase 3 |
Paragraph IV (Patent) Challenges for PREPOPIK
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| PREPOPIK | Oral Solution | citric acid; magnesium oxide; sodium picosulfate | 10 mg, 3.5 g, and 12 g | 202535 | 1 | 2014-05-21 |
US Patents and Regulatory Information for PREPOPIK
PREPOPIK is protected by two US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ferring Pharms Inc | PREPOPIK | citric acid; magnesium oxide; sodium picosulfate | FOR SOLUTION;ORAL | 202535-001 | Jul 16, 2012 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Ferring Pharms Inc | PREPOPIK | citric acid; magnesium oxide; sodium picosulfate | FOR SOLUTION;ORAL | 202535-001 | Jul 16, 2012 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for PREPOPIK
When does loss-of-exclusivity occur for PREPOPIK?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 08309287
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0818541
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 02152
Estimated Expiration: ⤷ Start Trial
China
Patent: 1406457
Estimated Expiration: ⤷ Start Trial
Patent: 1820859
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0180215
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 07526
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 07526
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 38174
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 4500
Patent: תהליך לייצור הרכב או הרכב רוקחי על ידי ריסוס ציפוי של שכבת נתרן פיקוסולפאט על גרעין של אשלגן ביקרבונאט (Process for the manufacture of a composition or pharmaceutical composition comprising spray coating a layer of sodium picosulphate on a core of potassium bicarbonate)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 54873
Estimated Expiration: ⤷ Start Trial
Patent: 11500549
Patent: 医薬品を製造する方法
Estimated Expiration: ⤷ Start Trial
Jordan
Patent: 06
Patent: عملية لتصنيع منتج صيدلاني يشتمل على حمض الستريك ،أكسيد ماغنسيوم، بيكربونات بوتاسيوم وبيكو كبريتات صوديوم، تركيبة صيدلانية تشتمل على حبيبات متحصل عليها بواسطة هذه العملية ومنتجات وسيطة منها (PROCESS FOR THE MANUFACURE OF A PHARMACEUTICAL PRODUCT COMPRISING CITRIC ACID, MAGNESIUM OXIDE, POTASSIUM BICARBONATE AND SODIUM PICOSULFATE, PHARMACEUTICAL COMPOSITION COMPRISING GRANULES OBTAINED BY SUCH PROCESS AND INTERMEDIATE PRODUCTS THEREOF)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 07526
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 10003588
Patent: PROCESO PARA LA MANUFACTURA DE UN PRODUCTO FARMACEUTICO. (PROCESS FOR THE MANUFACTURE OF A PHARMACEUTICAL PRODUCT.)
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 3899
Patent: PROCESS FOR THE MANUFACURE OF A PHARMACEUTICAL PRODUCT COMPRISING CITRIC ACID, MAGNESIUM OXIDE, POTASSIUM BICARBONATE AND SODIUM PICOSULFATE, PHARMACEUTICAL COMPOSITION COMPRISING GRANULES OBTAINED BY SUCH PROCESS AND INTERMEDIATE
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 07526
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 07526
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 73332
Patent: СПОСОБ ИЗГОТОВЛЕНИЯ ФАРМАЦЕВТИЧЕСКОГО ПРОДУКТА, СОДЕРЖАЩЕГО ЛИМОННУЮ КИСЛОТУ, ОКСИД МАГНИЯ, БИКАРБОНАТ КАЛИЯ И ПИКОСУЛЬФАТ НАТРИЯ, ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ, СОДЕРЖАЩИЕ ГРАНУЛЫ, ПОЛУЧЕННЫЕ ТАКИМ СПОСОБОМ, И ПРОМЕЖУТОЧНОЕ СОЕДИНЕНИЕ (METHOD OF MANUFACTURING PHARMACEUTICAL PRODUCT, CONTAINING CITRIC ACID, MAGNESIUM OXIDE, POTASSIUM BICARBONATE AND SODIUM PICOSULFATE, PHARMACEUTICAL COMPOSITIONS, CONTAINING THUS OBTAINED GRANULES AND INTERMEDIATE COMPOUND)
Estimated Expiration: ⤷ Start Trial
Patent: 10112483
Patent: СПОСОБ ИЗГОТОВЛЕНИЯ ФАРМАЦЕВТИЧЕСКОГО ПРОДУКТА, СОДЕРЖАЩЕГО ЛИМОННУЮ КИСЛОТУ, ОКСИД МАГНИЯ, БИКАРБОНАТ КАЛИЯ И ПИКОСУЛЬФАТ НАТРИЯ, ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ, СОДЕРЖАЩИЕ ГРАНУЛЫ, ПОЛУЧЕННЫЕ ТАКИМ СПОСОБОМ, И ПРОМЕЖУТОЧНОЕ СОЕДИНЕНИЕ (METHOD OF MANUFACTURING PHARMACEUTICAL PRODUCT, CONTAINING CITRIC ACID, MAGNESIUM OXIDE, POTASSIUM BICARBONATE AND SODIUM PICOSULFATE, PHARMACEUTICAL COMPOSITIONS, CONTAINING THUS OBTAINED GRANULES AND INTERMEDIATE COMPOUND)
Estimated Expiration: ⤷ Start Trial
Saudi Arabia
Patent: 290670
Patent: عملية تصنيع خاصة بمنتج صيدلاني من بيكوكبريتات الصوديوم (Manufacturing Process for a Pharmaceutical Sodium Picosulphate Product)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 07526
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1002131
Patent: PROCESS FOR THE MANUFACTURE OF A PHARMACEUTICAL PRODUCT COMPRISING CITRIC ACID,MAGNESIUM OXIDE,POTASSIUM BICARBONATE AND SODIUM PICOSULFATE,PHARMACEUTICAL COMPOSITION COMPRISING GRANULES OBTAINED BY SUCH PROCESS AND INTERMEDIATE
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1330096
Estimated Expiration: ⤷ Start Trial
Patent: 1381263
Estimated Expiration: ⤷ Start Trial
Patent: 100072020
Patent: PROCESS FOR THE MANUFACTURE OF A PHARMACEUTICAL PRODUCT COMPRISING CITRIC ACID, MAGNESIUM OXIDE, POTASSIUM BICARBONATE AND SODIUM PICOSULFATE, PHARMACEUTICAL COMPOSITION COMPRISING GRANULES OBTAINED BY SUCH PROCESS AND INTERMEDIATE
Estimated Expiration: ⤷ Start Trial
Patent: 130105757
Patent: PROCESS FOR THE MANUFACTURE OF A PHARMACEUTICAL PRODUCT COMPRISING CITRIC ACID, MAGNESIUM OXIDE, POTASSIUM BICARBONATE AND SODIUM PICOSULFATE, PHARMACEUTICAL COMPOSITION COMPRISING GRANULES OBTAINED BY SUCH PROCESS AND INTERMEDIATE
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 59716
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 0924759
Patent: Process for the manufacture of a pharmaceutical product
Estimated Expiration: ⤷ Start Trial
Patent: 18352
Estimated Expiration: ⤷ Start Trial
United Kingdom
Patent: 05953
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering PREPOPIK around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| South Korea | 101381263 | ⤷ Start Trial | |
| South Korea | 20100072020 | PROCESS FOR THE MANUFACTURE OF A PHARMACEUTICAL PRODUCT COMPRISING CITRIC ACID, MAGNESIUM OXIDE, POTASSIUM BICARBONATE AND SODIUM PICOSULFATE, PHARMACEUTICAL COMPOSITION COMPRISING GRANULES OBTAINED BY SUCH PROCESS AND INTERMEDIATE | ⤷ Start Trial |
| Mexico | 2010003588 | PROCESO PARA LA MANUFACTURA DE UN PRODUCTO FARMACEUTICO. (PROCESS FOR THE MANUFACTURE OF A PHARMACEUTICAL PRODUCT.) | ⤷ Start Trial |
| South Korea | 20130105757 | PROCESS FOR THE MANUFACTURE OF A PHARMACEUTICAL PRODUCT COMPRISING CITRIC ACID, MAGNESIUM OXIDE, POTASSIUM BICARBONATE AND SODIUM PICOSULFATE, PHARMACEUTICAL COMPOSITION COMPRISING GRANULES OBTAINED BY SUCH PROCESS AND INTERMEDIATE | ⤷ Start Trial |
| Japan | 5654873 | ⤷ Start Trial | |
| Croatia | P20180215 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
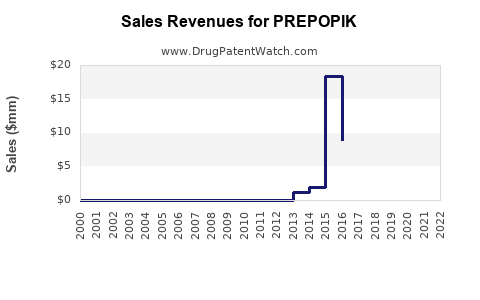
PREPOPIK: Market Dynamics and Financial Trajectory Analysis
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.